PGT 120 Engineering Materials Lecture 2 Atom Bonding

PGT 120 Engineering Materials Lecture 2: Atom & Bonding
ATOMIC STRUCTURE § Atoms are the structural unit of all engineering materials penicilin
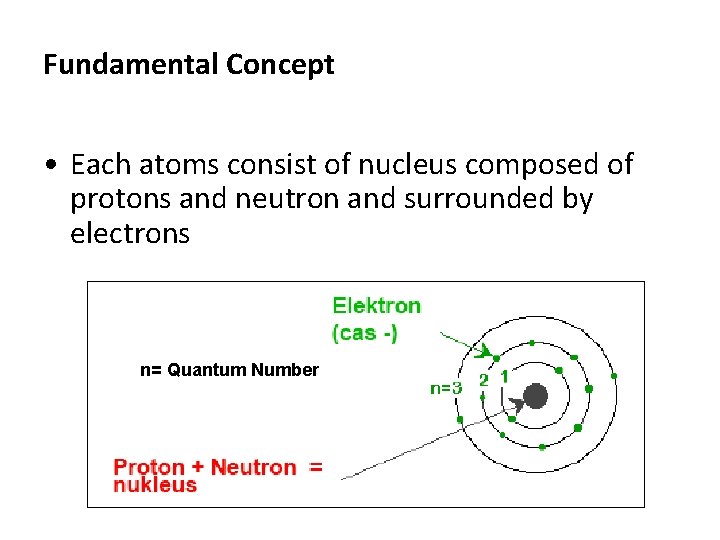
Fundamental Concept • Each atoms consist of nucleus composed of protons and neutron and surrounded by electrons n= Quantum Number
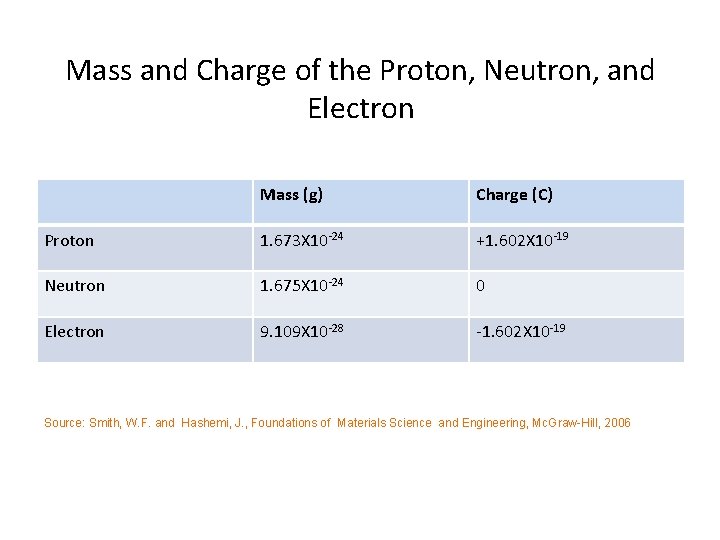
Mass and Charge of the Proton, Neutron, and Electron Mass (g) Charge (C) Proton 1. 673 X 10 -24 +1. 602 X 10 -19 Neutron 1. 675 X 10 -24 0 Electron 9. 109 X 10 -28 -1. 602 X 10 -19 Source: Smith, W. F. and Hashemi, J. , Foundations of Materials Science and Engineering, Mc. Graw-Hill, 2006

• Atomic number, Z - Number of protons (p). In a neutral atom the atomic number is equal to the number of electrons (Z=e). • Atomic mass, A - Total mass of proton and neutron in the nucleus ( A=Z+N ). • Isotope - atoms that have two or more atomic mass. Same number of proton but different number of neutron. • 1 atomic mass unit (a. m. u) = 1/12 of the atomic mass of carbon • 1 mole= 6. 023 x 1023 atoms ( Avogadro’s number NA ).

Atomic Number and Atomic Mass • Atomic Number = Number of Protons in the nucleus • Unique to an element Ø Example : - Hydrogen = 1, Uranium = 92 • Relative atomic mass = Mass in grams of 6. 203 x 1023 ( Avagadro Number) Atoms. Ø Example : - Carbon has 6 Protons and 6 Neutrons. Atomic Mass = 12. • • One Atomic Mass unit is 1/12 th of mass of carbon atom. One gram mole = Gram atomic mass of an element. Example : - One gram Mole of Carbon 2 -3 12 Grams Of Carbon 6. 023 x 1023 Carbon Atoms

Z X A Ex: Determine the number of electron (e), neutron (N) in fluorin atom Ans: A=p+N= Z=p=e= 9 So, N=A-Z=10 9 F 19
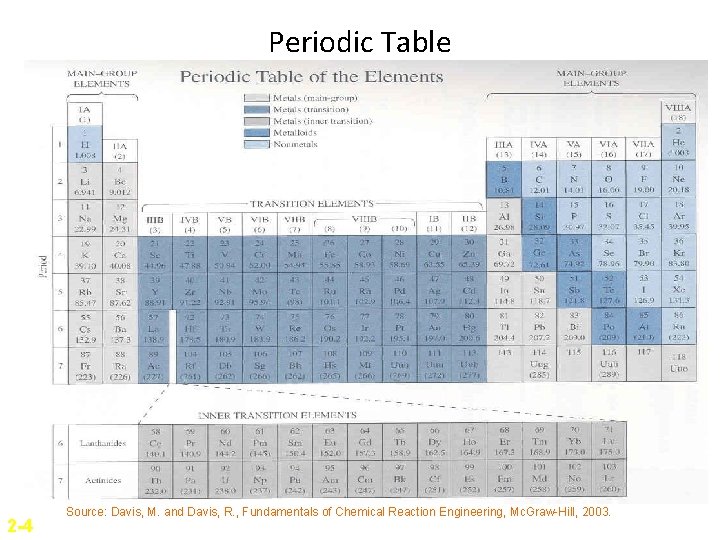
Periodic Table 2 -4 Source: Davis, M. and Davis, R. , Fundamentals of Chemical Reaction Engineering, Mc. Graw-Hill, 2003.

Example: 1 mole aluminium have mass of 26. 98 g and 6. 023 x 1023 atoms. 1) What is the mass in grams of 1 atom of aluminium (A=26. 98 g/mol) Ans:

2) How many atom of Coper (Cu) in 1 gram Of Coper ? (A=63. 54 g/mol) Ans:

Ans 1: 1 mol = 6. 023 x 1023 atom mass 1 mol Al = 26. 98 g mass (g) in 1 atom Al= 26. 98 g 6. 023 x 1023 Ans 2: 1 mol Cu= 63. 54 g 1 mol Cu= 6. 023 x 1023 atom Number of Atom Cu in 1 gram Cu = 6. 023 x 1023 atom 63. 54

• The Cladding (outside layers) of the US quarter coin consists of an alloy of 75 wt % copper and 25 wt % nickel. What are the atomic percent of Cu and Ni of this materials?

• Solutions: Using the basis of 100 g of the 75 wt % Cu and 25 wt % Ni alloy, there are 75 g Cu and 25 g Ni. Thus, the number of gram-moles of copper and nickel is No. of gram-moles of Cu= No. of gram moles of Ni = Total gram-moles=1. 6063 mol Atomic % Cu= Atomic % Ni=
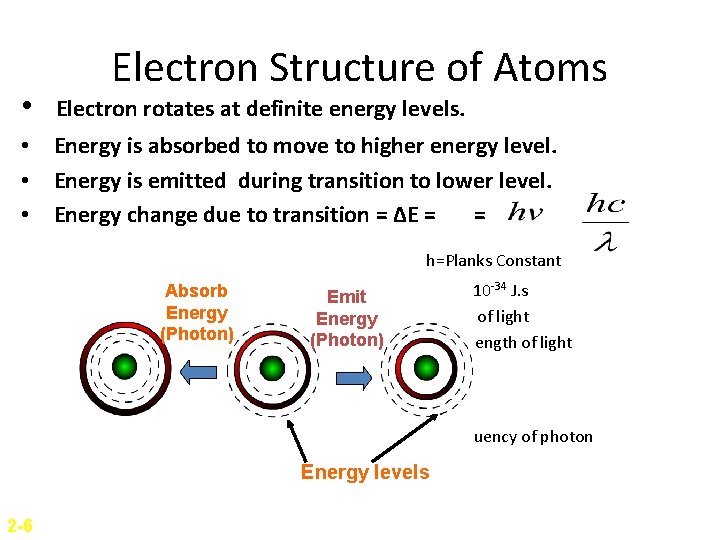
• Electron Structure of Atoms Electron rotates at definite energy levels. • Energy is absorbed to move to higher energy level. • Energy is emitted during transition to lower level. • Energy change due to transition = ΔE = = h=Planks Constant Absorb Energy (Photon) Emit Energy (Photon) = 6. 63 x 10 -34 J. s c = Speed of light λ = Wavelength of light v=frequency of photon Energy levels 2 -6

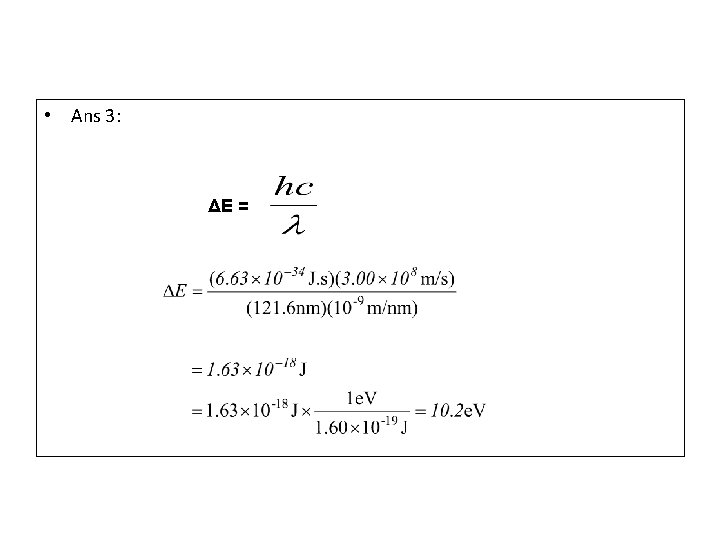
• Example 3) Calculate the energy in joules (J) and electron volts (e. V) of the photon whose wave length is 121. 6 nm. (Given 1. 00 e. V=1. 60 X 10 -19 J; h= 6. 63 X 10 -34 J. s)

Energy in Hydrogen Atom • • Hydrogen atom has one proton and one electron Energy of hydrogen atoms for different energy levels is given by (n=1, 2…. . )principal quantum numbers • Example: - If an electron undergoes transition from n=3 state to n=2 state, the energy of photon emitted is • Energy required to completely remove an electron from hydrogen atom is known as ionization energy 2 -7

• A hydrogen atom exists with its electron in the n= 3 state. The electron undergoes a transition to the n=2 state. Calculate (a) the energy of the photon emitted, (b) its frequency, and (c) its wavelength. Ans: (a) Energy of the photon emited is

b) The frequency of the photon is c) The wavelength of the photon is

Quantum Numbers of Electrons of Atoms Principal Quantum Number (n) • Represents main energy levels. • Range 1 to 7. • Larger the ‘n’ higher the energy. Subsidiary Quantum Number l • Represents sub energy levels (orbital). • Range 0…n-1. • Represented by letters s, p, d and f. n=1 n=2 n=3 2 -8 n=1 n=2 s orbital (l=0) p Orbital (l=1)

Quantum Numbers of Electrons of Atoms (Cont. . ) Magnetic Quantum Number ml. Represents spatial • orientation of single atomic orbital. • Permissible values are –l to +l. • Example: - if l=1, ml = -1, 0, +1. I. e. 2 l+1 allowed values. • No effect on energy. 2 -9 Electron spin quantum number ms. • Specifies two directions of electron spin. • Directions are clockwise or anticlockwise. • Values are +1/2 or – 1/2. • Two electrons on same orbital have opposite spins. • No effect on energy.

Electron Structure of Multielectron Atom • Maximum number of electrons in each atomic shell is given by 2 n 2. • Atomic size (radius) increases with addition of shells. • Electron Configuration lists the arrangement of electrons in orbitals. Ø Example : - Number of Electrons Orbital letters 1 s 2 2 p 6 3 s 2 Principal Quantum Numbers Ø For Iron, (Z=26), Electronic configuration is 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6 4 s 2 2 -10

Stable electron configurations. . . • have complete s and p subshells • tend to be unreactive.

• Most elements: Electron configuration not stable. • Why? Valence (outer) shell usually not filled completely.

Example • Write the electron configuration for the following atoms by using conventional spdf notation. • a) Fe atom (Z=26) and the Fe 2+ and Fe 3+ ions

remember !!!!! 7 s 7 p 7 d 6 s 6 p 6 d 6 f 5 s 5 p 5 d 5 f 4 s 4 p 4 d 4 f 3 s 3 p 3 d 2 s 2 p 1 s

Electrons fill up in this order The order for writing the orbitals

Assignment 1 • Using the spdf notation, write the electron configuration for atom and ions below • Magnesium –(Mg, Mg 2+) • Nikel (Ni, Ni 2+) • Cobalt (Co, Co 2+, Co 3+) • Silicon-(Si) • Tin-(Sn) • Lead-(Pb) – Submit by Monday –last 5 pm – Blok 9, Room 09 -00 -0 A or PPK Mikroe office.

Electron Structure and Chemical Activity (Cont. . ) • Electronegative elements accept electrons during chemical reaction. • Some elements behave as both electronegative and electropositive. • Electronegativity is the degree to which the atom attracts electrons to itself Ø Measured on a scale of 0 to 4. 1 Ø Example : - Electronegativity of Fluorine is 4. 1 Electronegativity of Sodium is 1. Te Na Electropositive 0 2 -12 K 1 W 2 H N Se 3 O Fl 4 Electronegative

Atomic and Molecular Bonds • Ionic bonds : - Strong atomic bonds due to transfer of electrons • Covalent bonds : - Large interactive force due to sharing of electrons • Metallic bonds : - Non-directional bonds formed by sharing of electrons • Permanent Dipole bonds : - Weak intermolecular bonds due to attraction between the ends of permanent dipoles. • Fluctuating Dipole bonds : - Very weak electric dipole bonds due to asymmetric distribution of electron densities. 2 -12
Ionic Bonding
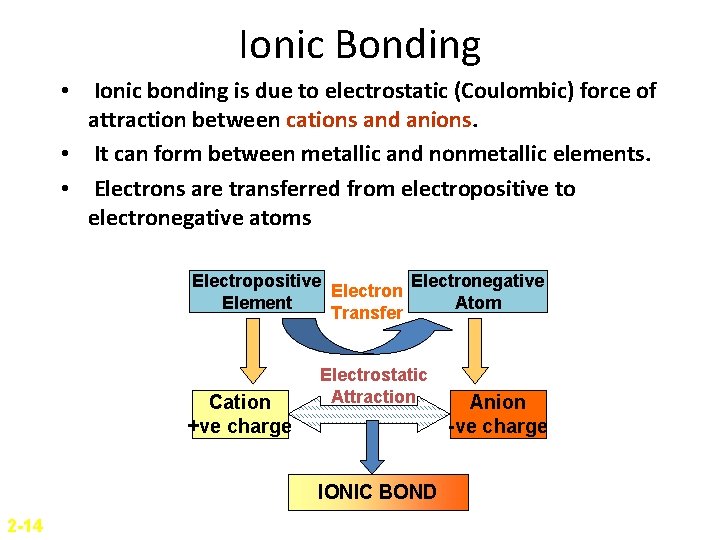
Ionic Bonding • Ionic bonding is due to electrostatic (Coulombic) force of attraction between cations and anions. • It can form between metallic and nonmetallic elements. • Electrons are transferred from electropositive to electronegative atoms Electropositive Electronegative Electron Element Atom Transfer Cation +ve charge Electrostatic Attraction IONIC BOND 2 -14 Anion -ve charge
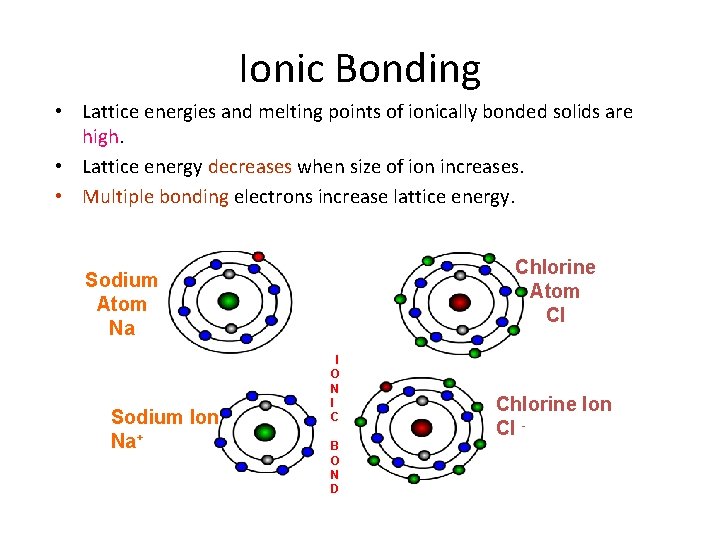
Ionic Bonding • Lattice energies and melting points of ionically bonded solids are high. • Lattice energy decreases when size of ion increases. • Multiple bonding electrons increase lattice energy. Chlorine Atom Cl Sodium Atom Na Sodium Ion Na+ I O N I C B O N D Chlorine Ion Cl -

Ionic Force for Ion Pair • Nucleus of one ion attracts electron of another ion. • The electron clouds of ion repulse each other when they are sufficiently close. Force versus separation Distance for a pair of oppositely charged ions Figure 2. 11 2 -16

Ion Force for Ion Pair (Cont. . ) Z 1, Z 2 = Number of electrons removed or added during ion formation e = Electron Charge a = Interionic seperation distance ε = Permeability of free space (8. 85 x 10 -12 c 2/Nm 2) (n and b are constants) 2 -17

Interionic Force - Example • Force of attraction between Na+ and Cl- ions Z 1 = +1 for Na+, Z 2 = -1 for Cle = 1. 60 x 10 -19 C , ε 0 = 8. 85 x 10 -12 C 2/Nm 2 a 0 = Sum of Radii of Na+ and Cl- ions = 0. 095 nm + 0. 181 nm = 2. 76 x 10 -10 m Cl- Na+ a 0 2 -18

Interionic Energies for Ion Pairs • Net potential energy for a pair of oppositely charged ions = Attraction Repulsion Energy Released Absorbed • Enet is minimum when ions are at equilibrium seperation distance a 0 2 -19

Ion Arrangements in Ionic Solids • Ionic bonds are Non Directional • Geometric arrangements are present in solids to maintain electric neutrality. Ø Example: - in Na. Cl, six Cl- ions pack around central Na+ Ions Ionic packing In Na. Cl and Cs. Cl Figure 2. 13 Cs. Cl Na. Cl • As the ratio of cation to anion radius decreases, fewer anion surround central cation. 2 -20

Bonding Energies • Lattice energies and melting points of ionically bonded solids are high. • Lattice energy decreases when size of ion increases. • Multiple bonding electrons increase lattice energy. Ø Example : - Na. Cl Cs. Cl Ba. O 2 -21 Lattice energy = 766 KJ/mol Melting point = 801 o. C Lattice energy = 649 KJ/mol Melting Point = 646 o. C Lattice energy = 3127 KJ/mol Melting point = 1923 o. C
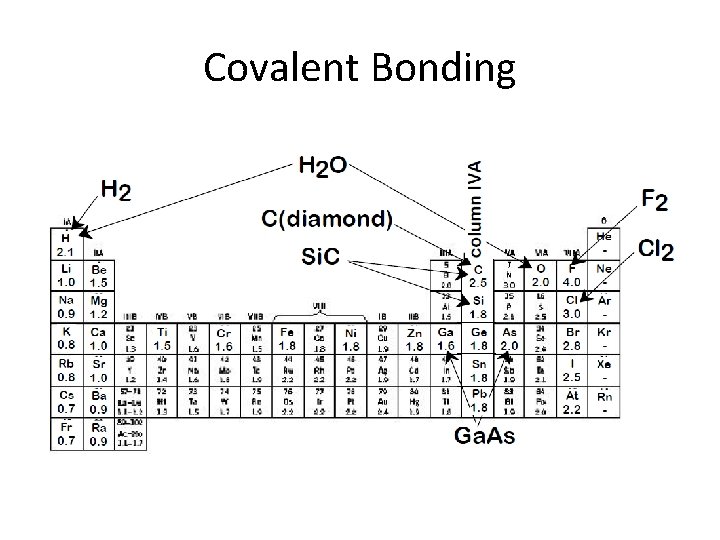
Covalent Bonding
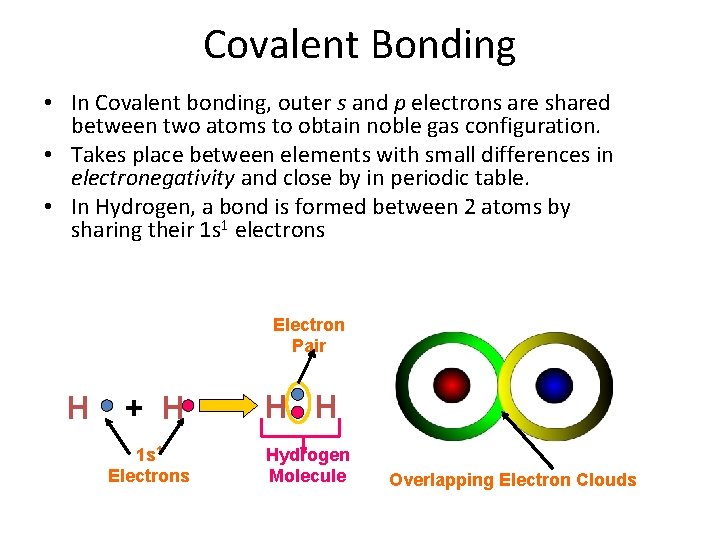
Covalent Bonding • In Covalent bonding, outer s and p electrons are shared between two atoms to obtain noble gas configuration. • Takes place between elements with small differences in electronegativity and close by in periodic table. • In Hydrogen, a bond is formed between 2 atoms by sharing their 1 s 1 electrons Electron Pair H + H 1 s 1 Electrons H H Hydrogen Molecule Overlapping Electron Clouds
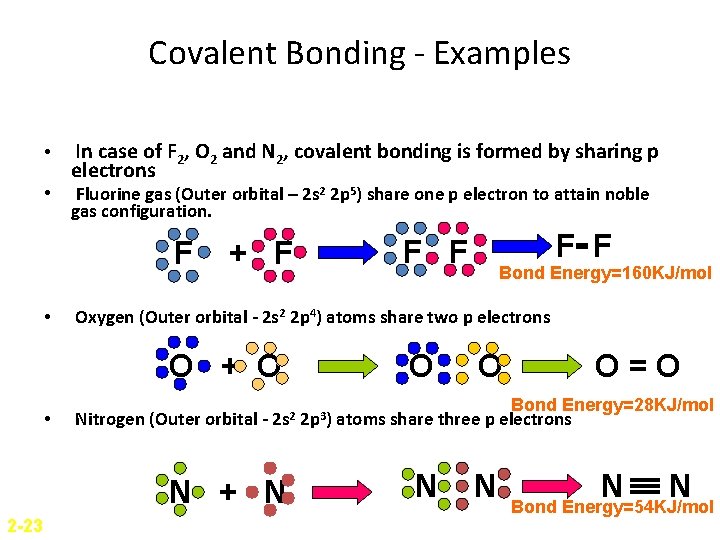
Covalent Bonding - Examples • • In case of F 2, O 2 and N 2, covalent bonding is formed by sharing p electrons Fluorine gas (Outer orbital – 2 s 2 2 p 5) share one p electron to attain noble gas configuration. F • + F H Nitrogen (Outer orbital - 2 s 2 HH N + N 2 -23 Bond Energy=160 KJ/mol Oxygen (Outer orbital - 2 s 2 2 p 4) atoms share two p electrons O + O • F F O 2 p 3) O O=O Bond Energy=28 KJ/mol atoms share three p electrons N N Bond Energy=54 KJ/mol
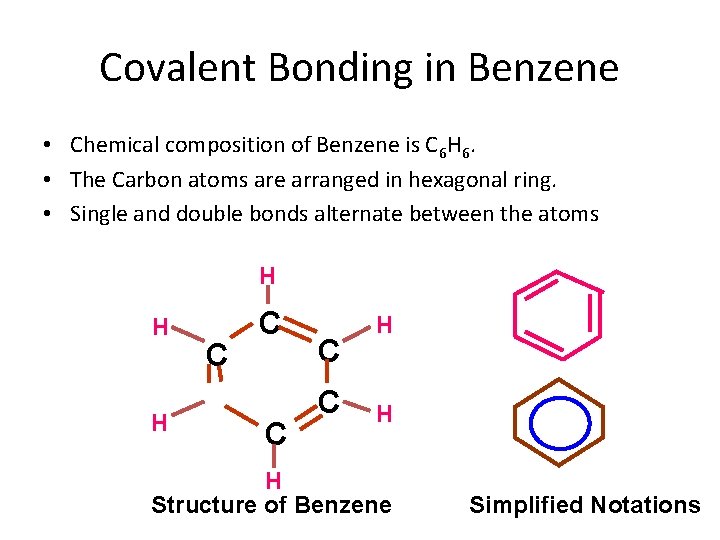
Covalent Bonding in Benzene • Chemical composition of Benzene is C 6 H 6. • The Carbon atoms are arranged in hexagonal ring. • Single and double bonds alternate between the atoms H H H C C C H H H Structure of Benzene Simplified Notations
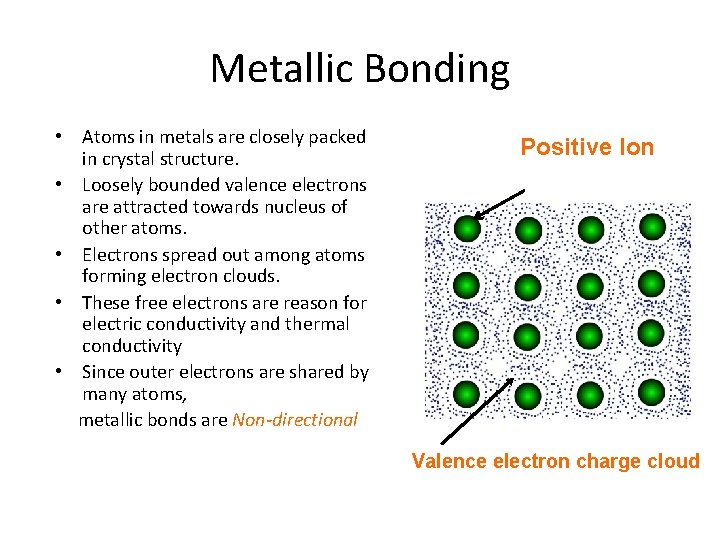
Metallic Bonding • Atoms in metals are closely packed in crystal structure. • Loosely bounded valence electrons are attracted towards nucleus of other atoms. • Electrons spread out among atoms forming electron clouds. • These free electrons are reason for electric conductivity and thermal conductivity • Since outer electrons are shared by many atoms, metallic bonds are Non-directional Positive Ion Valence electron charge cloud
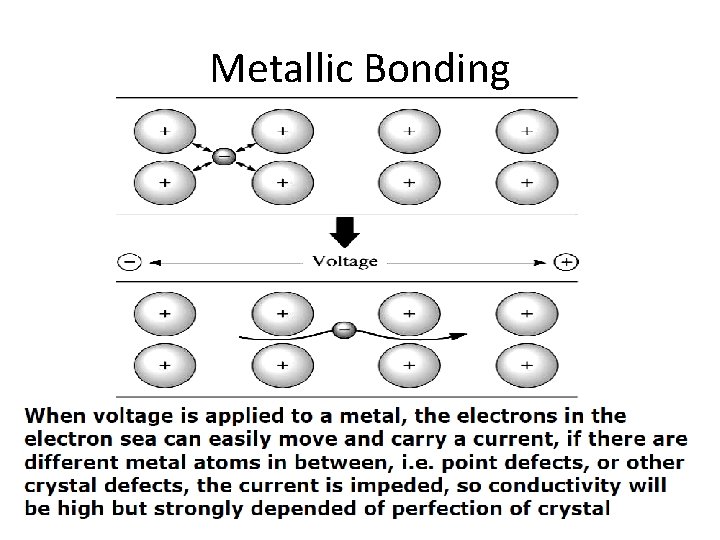
Metallic Bonding

Metallic Bonds (Cont. . ) • Overall energy of individual atoms are lowered by metallic bonds • Minimum energy between atoms exist at equilibrium distance a 0 Fewer the number of valence electrons involved, more • metallic the bond is. Ø Example: - Na Bonding energy 108 KJ/mol, Melting temperature 97. 7 o. C • Higher the number of valence electrons involved, higher is the bonding energy. Ca Bonding energy 177 KJ/mol, Ø Example: Melting temperature 851 o. C 2 -29
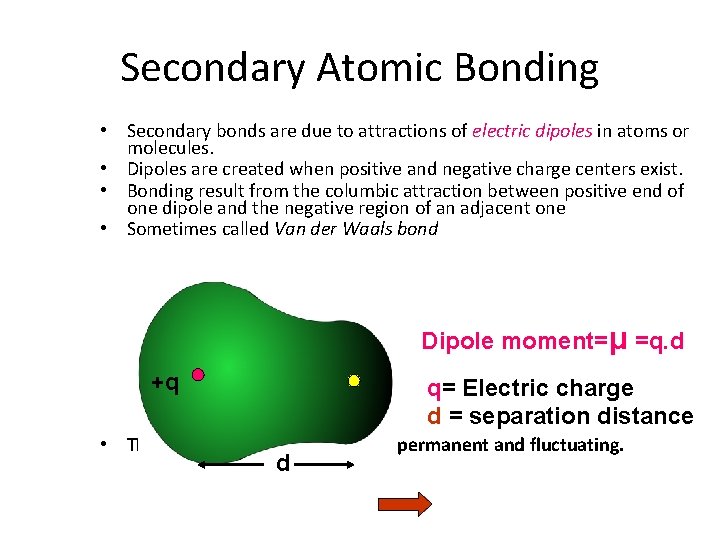
Secondary Atomic Bonding • Secondary bonds are due to attractions of electric dipoles in atoms or molecules. • Dipoles are created when positive and negative charge centers exist. • Bonding result from the columbic attraction between positive end of one dipole and the negative region of an adjacent one • Sometimes called Van der Waals bond Dipole moment=μ =q. d +q q= Electric charge d = separation distance • There two types of bonds d permanent and fluctuating.
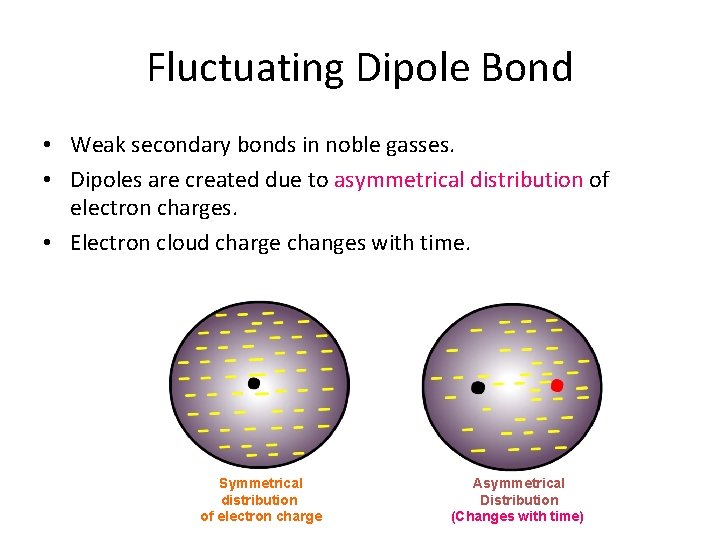
Fluctuating Dipole Bond • Weak secondary bonds in noble gasses. • Dipoles are created due to asymmetrical distribution of electron charges. • Electron cloud charge changes with time. Symmetrical distribution of electron charge Asymmetrical Distribution (Changes with time)
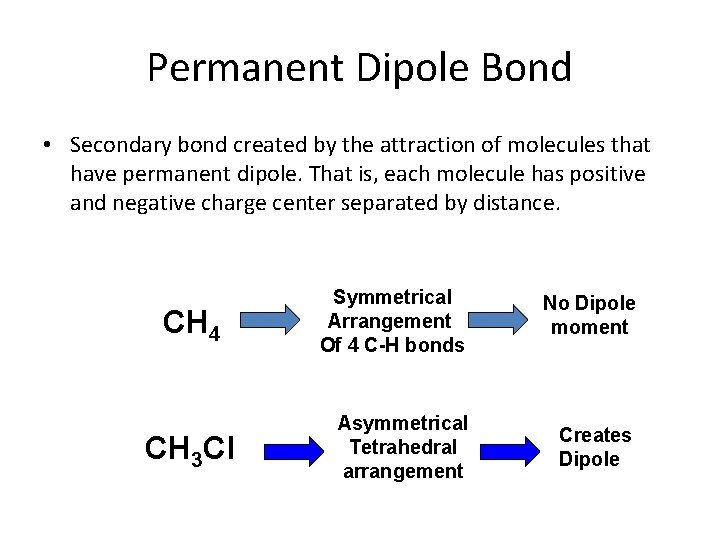
Permanent Dipole Bond • Secondary bond created by the attraction of molecules that have permanent dipole. That is, each molecule has positive and negative charge center separated by distance. CH 4 CH 3 Cl Symmetrical Arrangement Of 4 C-H bonds No Dipole moment Asymmetrical Tetrahedral arrangement Creates Dipole
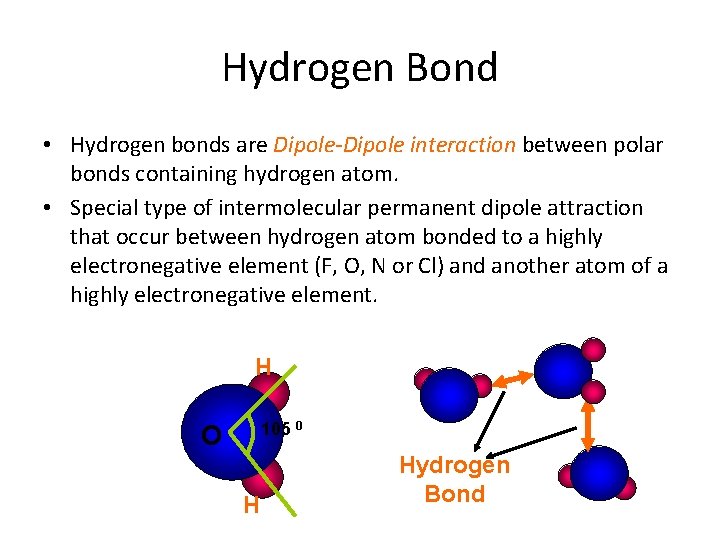
Hydrogen Bond • Hydrogen bonds are Dipole-Dipole interaction between polar bonds containing hydrogen atom. • Special type of intermolecular permanent dipole attraction that occur between hydrogen atom bonded to a highly electronegative element (F, O, N or Cl) and another atom of a highly electronegative element. H 105 0 O H Hydrogen Bond

Mixed Bonding • Chemical bonding of atoms or ions can involve more than one type of primary bond and can also involve secondary dipole bond. 1. 2. 3. 4. Ionic - covalent Metallic – ionic Ionic – covalent - metallic
- Slides: 51