Petrucci Harwood Herring Madura GENERAL CHEMISTRY Ninth Edition







![Weak Acids Acetic Acid [CH 3 CO 2 -][H 3 O+] K a= = Weak Acids Acetic Acid [CH 3 CO 2 -][H 3 O+] K a= =](https://slidetodoc.com/presentation_image_h/ca60ed8dc67f23e79fa2c1cdc4b4901e/image-18.jpg)
![Weak Bases [CH 3 NH 3+][HO-] K b= = 4. 3 10 -4 [CH Weak Bases [CH 3 NH 3+][HO-] K b= = 4. 3 10 -4 [CH](https://slidetodoc.com/presentation_image_h/ca60ed8dc67f23e79fa2c1cdc4b4901e/image-19.jpg)



![Percent Ionization Ka = [H 3 O+][A-] [HA] n H O n. A 1 Percent Ionization Ka = [H 3 O+][A-] [HA] n H O n. A 1](https://slidetodoc.com/presentation_image_h/ca60ed8dc67f23e79fa2c1cdc4b4901e/image-25.jpg)









- Slides: 41

Petrucci • Harwood • Herring • Madura GENERAL CHEMISTRY Ninth Edition Principles and Modern Applications Chapter 16: Acids and Bases Philip Dutton University of Windsor, Canada Prentice-Hall © 2007 1 General Chemistry: Chapter 16 Prentice-Hall © 2007

Contents 16 -1 16 -2 16 -3 16 -4 16 -5 16 -6 16 -7 16 -8 2 The Arrhenius Theory: A Brief Review Brønsted-Lowry Theory of Acids and Bases The Self-Ionization of Water and the p. H Scale Strong Acids and Strong Bases Weak Acids and Weak Bases Polyprotic Acids Ions as Acids and Bases Molecular Structure and Acid-Base Behaviour General Chemistry: Chapter 16 Prentice-Hall © 2007

16 -1 The Arrhenius Theory: A Brief Review H 2 O HCl(g) → H+(aq) + Cl-(aq) H 2 O Na. OH(s) → Na+(aq) + OH-(aq) + H+(aq) + Cl-(aq) → H 2 O(l) + Na+(aq) + Cl-(aq) H+(aq) + OH-(aq) → H 2 O(l) Arrhenius theory did not handle non OHbases such as ammonia very well. 3 General Chemistry: Chapter 16 Prentice-Hall © 2007

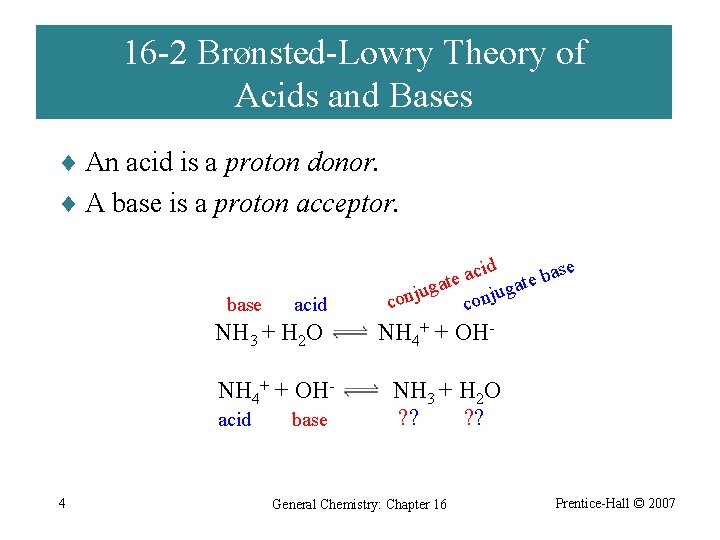
16 -2 Brønsted-Lowry Theory of Acids and Bases ¨ An acid is a proton donor. ¨ A base is a proton acceptor. base acid NH 3 + H 2 O NH 4+ + OHacid 4 base cid ase a b e e t at ga g u u j j n co con NH 4+ + OHNH 3 + H 2 O ? ? General Chemistry: Chapter 16 Prentice-Hall © 2007

The Solvated Proton 5 General Chemistry: Chapter 16 Prentice-Hall © 2007

Base Ionization Constant base acid NH 3 + H 2 O conjugate base acid NH 4+ + OH- [NH 4+][OH-] K c= [NH 3][H 2 O] Kb= Kc[H 2 O] = 6 [NH 4+][OH-] [NH 3] = 1. 8 10 -5 General Chemistry: Chapter 16 Prentice-Hall © 2007

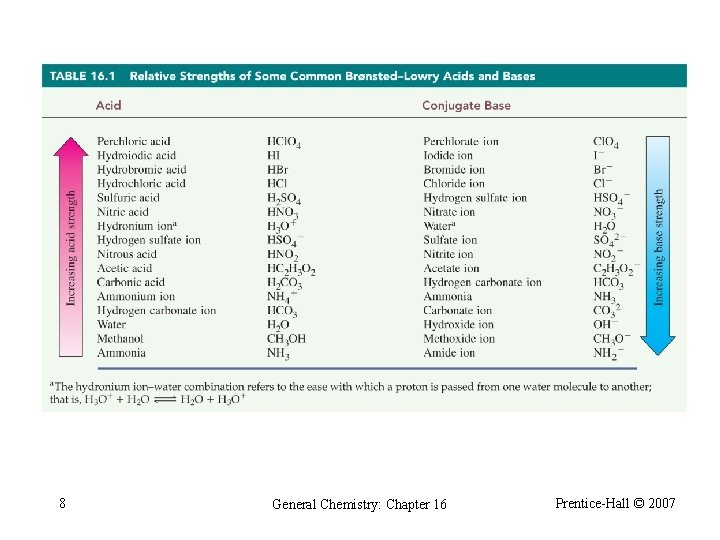
Acid Ionization Constant acid base CH 3 CO 2 H + H 2 O conjugate base conjugate acid CH 3 CO 2 - + H 3 O+ [CH 3 CO 2 -][H 3 O+] K c= [CH 3 CO 2 H][H 2 O] [CH 3 CO 2 -][H 3 O+] Ka= Kc[H 2 O] = = 1. 8 10 -5 [CH 3 CO 2 H] 7 General Chemistry: Chapter 16 Prentice-Hall © 2007

8 General Chemistry: Chapter 16 Prentice-Hall © 2007

A Weak Base 9 General Chemistry: Chapter 16 Prentice-Hall © 2007

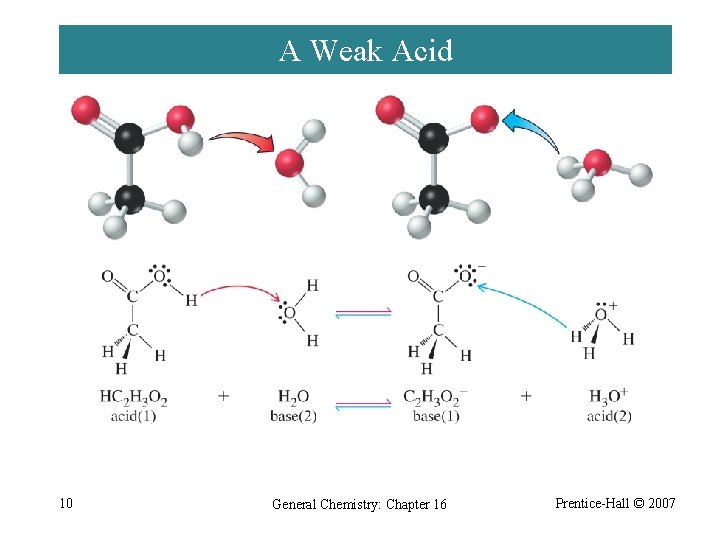
A Weak Acid 10 General Chemistry: Chapter 16 Prentice-Hall © 2007

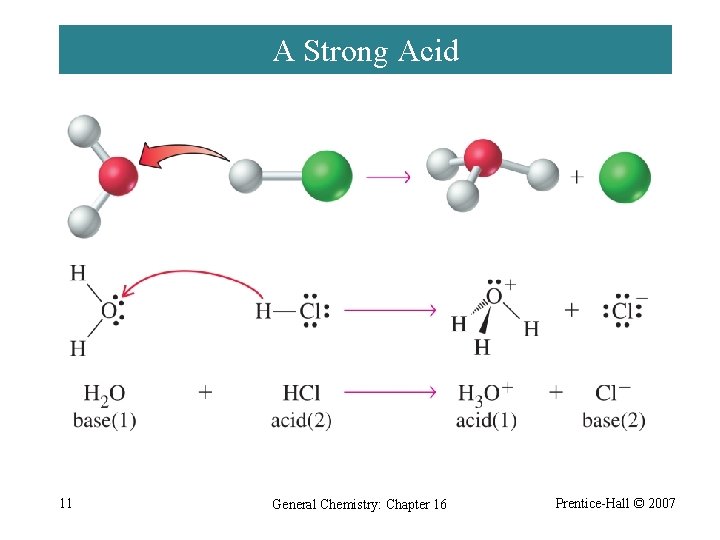
A Strong Acid 11 General Chemistry: Chapter 16 Prentice-Hall © 2007

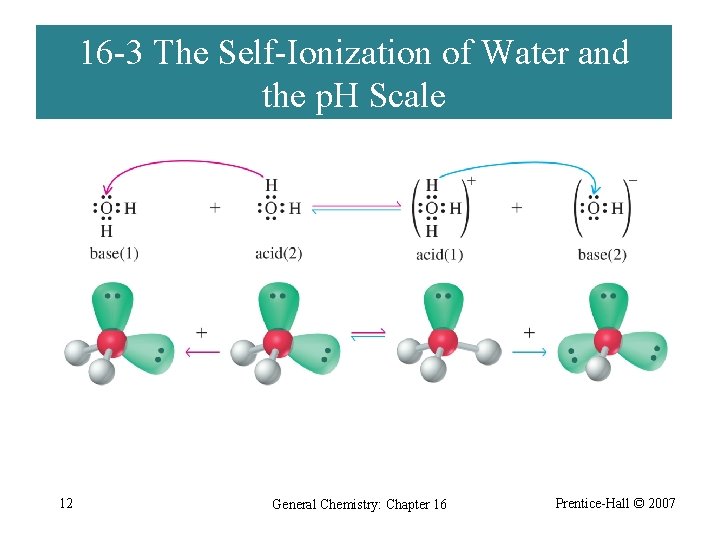
16 -3 The Self-Ionization of Water and the p. H Scale 12 General Chemistry: Chapter 16 Prentice-Hall © 2007

Ion Product of Water base acid H 2 O + H 2 O K c= conjugate base acid H 3 O+ + OH- [H 3 O+][OH-] [H 2 O] KW= Kc[H 2 O] = [H 3 O+][OH-] = 1. 0 10 -14 13 General Chemistry: Chapter 16 Prentice-Hall © 2007

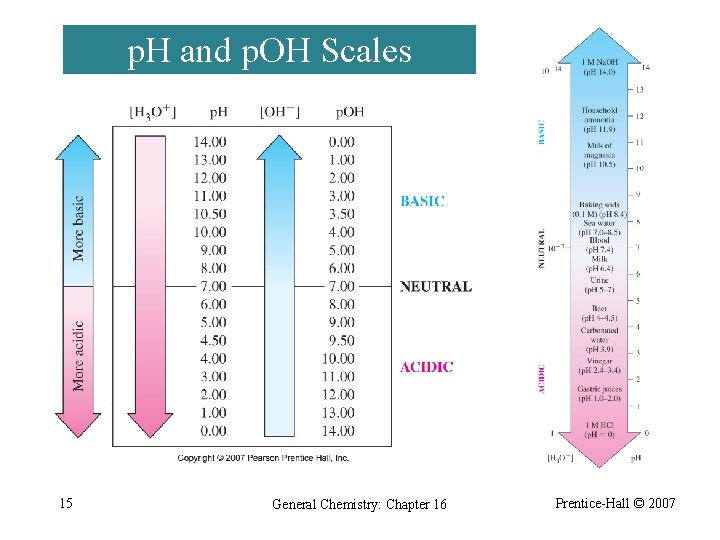
p. H and p. OH ¨ The potential of the hydrogen ion was defined in 1909 as the negative of the logarithm of [H+]. p. H = -log[H 3 O+] p. OH = -log[OH-] KW = [H 3 O+][OH-]= 1. 0 10 -14 -log. KW = -log[H 3 O+]-log[OH-]= -log(1. 0 10 -14) p. KW = p. H + p. OH= -(-14) p. KW = p. H + p. OH = 14 14 General Chemistry: Chapter 16 Prentice-Hall © 2007

p. H and p. OH Scales 15 General Chemistry: Chapter 16 Prentice-Hall © 2007

16 -4 Strong Acids and Bases HCl CH 3 CO 2 H Thymol Blue Indicator p. H < 1. 2 < p. H < 2. 8 < p. H 16 General Chemistry: Chapter 16 Prentice-Hall © 2007

16 -5 Weak Acids and Bases Acetic Acid Lactic Acid 17 General Chemistry: Chapter 16 Glycine Prentice-Hall©© 2007
![Weak Acids Acetic Acid CH 3 CO 2 H 3 O K a Weak Acids Acetic Acid [CH 3 CO 2 -][H 3 O+] K a= =](https://slidetodoc.com/presentation_image_h/ca60ed8dc67f23e79fa2c1cdc4b4901e/image-18.jpg)
Weak Acids Acetic Acid [CH 3 CO 2 -][H 3 O+] K a= = 1. 8 10 -5 [CH 3 CO 2 H] p. Ka= -log(1. 8 10 -5) = 4. 74 18 General Chemistry: Chapter 16 Prentice-Hall©© 2007
![Weak Bases CH 3 NH 3HO K b 4 3 10 4 CH Weak Bases [CH 3 NH 3+][HO-] K b= = 4. 3 10 -4 [CH](https://slidetodoc.com/presentation_image_h/ca60ed8dc67f23e79fa2c1cdc4b4901e/image-19.jpg)
Weak Bases [CH 3 NH 3+][HO-] K b= = 4. 3 10 -4 [CH 3 NH 2] p. Kb= -log(4. 2 10 -4) = 3. 37 19 General Chemistry: Chapter 16 Prentice-Hall © 2007

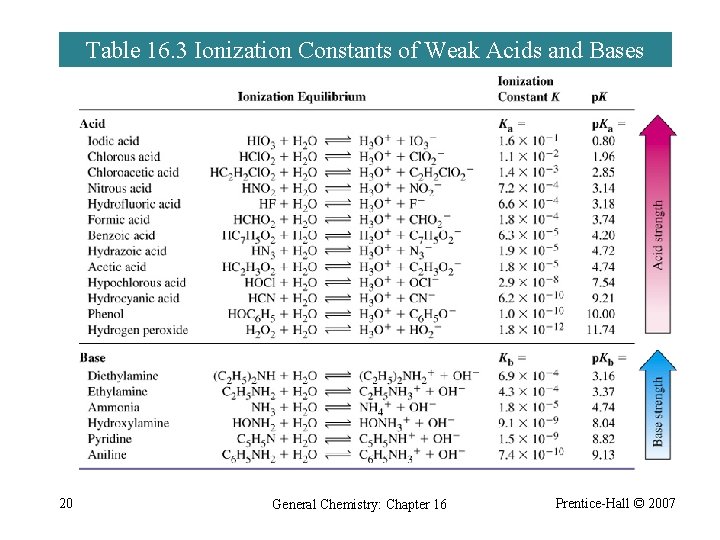
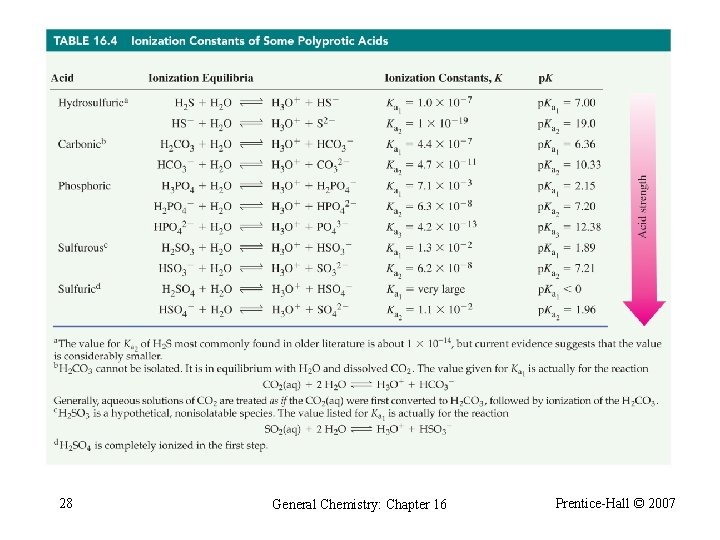
Table 16. 3 Ionization Constants of Weak Acids and Bases 20 General Chemistry: Chapter 16 Prentice-Hall © 2007

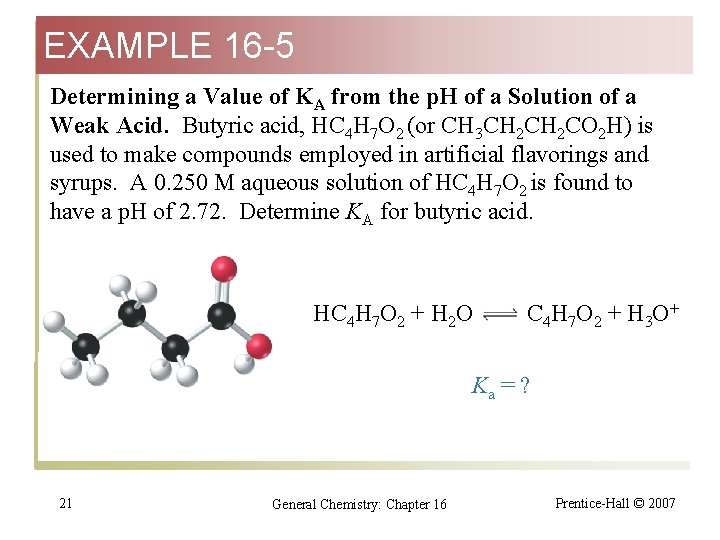
EXAMPLE 16 -5 Determining a Value of KA from the p. H of a Solution of a Weak Acid. Butyric acid, HC 4 H 7 O 2 (or CH 3 CH 2 CO 2 H) is used to make compounds employed in artificial flavorings and syrups. A 0. 250 M aqueous solution of HC 4 H 7 O 2 is found to have a p. H of 2. 72. Determine KA for butyric acid. HC 4 H 7 O 2 + H 2 O C 4 H 7 O 2 + H 3 O+ Ka = ? 21 General Chemistry: Chapter 16 Prentice-Hall © 2007

EXAMPLE 16 -5 Solution: For HC 4 H 7 O 2 KA is likely to be much larger than KW. Therefore assume self-ionization of water is unimportant. HC 4 H 7 O 2 + H 2 O C 4 H 7 O 2 + H 3 O+ Initial conc. 0. 250 M 0 0 Changes -x M +x M Equilibrium Concentration (0. 250 -x) M x. M 22 General Chemistry: Chapter 16 Prentice-Hall © 2007

EXAMPLE 16 -5 HC 4 H 7 O 2 + H 2 O C 4 H 7 O 2 + H 3 O+ Log[H 3 O+] = -p. H = -2. 72 [H 3 O+] = 10 -2. 72 = 1. 9 10 -3 = x K a= [H 3 O+] [C 4 H 7 O 2 -] [HC 4 H 7 O 2] Ka= 1. 5 10 -5 23 = 1. 9 10 -3 · 1. 9 10 -3 (0. 250 – 1. 9 10 -3) Check assumption: Ka >> KW. General Chemistry: Chapter 16 Prentice-Hall © 2007

Percent Ionization HA + H 2 O H 3 O+ + A- Degree of ionization = Percent ionization = 24 [H 3 O+] from HA [HA] originally General Chemistry: Chapter 16 100% Prentice-Hall © 2007
![Percent Ionization Ka H 3 OA HA n H O n A 1 Percent Ionization Ka = [H 3 O+][A-] [HA] n H O n. A 1](https://slidetodoc.com/presentation_image_h/ca60ed8dc67f23e79fa2c1cdc4b4901e/image-25.jpg)
Percent Ionization Ka = [H 3 O+][A-] [HA] n H O n. A 1 Ka = n. HA V 3 25 General Chemistry: Chapter 16 + - Prentice-Hall © 2007

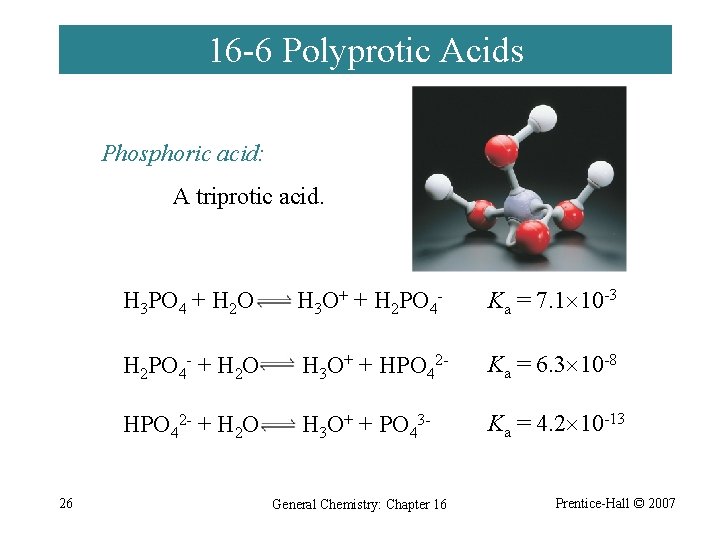
16 -6 Polyprotic Acids Phosphoric acid: A triprotic acid. 26 H 3 PO 4 + H 2 O H 3 O+ + H 2 PO 4 - Ka = 7. 1 10 -3 H 2 PO 4 - + H 2 O H 3 O+ + HPO 42 - Ka = 6. 3 10 -8 HPO 42 - + H 2 O H 3 O+ + PO 43 - Ka = 4. 2 10 -13 General Chemistry: Chapter 16 Prentice-Hall © 2007

Phosphoric Acid ¨ Ka 1 >> Ka 2 ◦ All H 3 O+ is formed in the first ionization step. ¨ H 2 PO 4 - essentially does not ionize further. ◦ Assume [H 2 PO 4 -] = [H 3 O+]. ¨ [HPO 42 -] Ka 2 regardless of solution molarity. 27 General Chemistry: Chapter 16 Prentice-Hall © 2007

28 General Chemistry: Chapter 16 Prentice-Hall © 2007

EXAMPLE 16 -9 Calculating Ion Concentrations in a Polyprotic Acid Solution. For a 3. 0 M H 3 PO 4 solution, calculate: (a) [H 3 O+]; (b) [H 2 PO 4 -]; (c) [HPO 42 -] H 3 PO 4 + H 2 O (d) [PO 43 -] H 2 PO 4 - + H 3 O+ Initial conc. 3. 0 M 0 0 Changes -x M +x M Equilibrium Concentration (3. 0 -x) M x. M 29 General Chemistry: Chapter 16 Prentice-Hall © 2007

EXAMPLE 16 -9 H 3 PO 4 + K a= H 2 O [H 3 O+] [H 2 PO 4 -] [H 3 PO 4] H 2 PO 4 - + H 3 O+ = x·x (3. 0 – x) = 7. 1 10 -3 Assume that x << 3. 0 x 2 = (3. 0)(7. 1 10 -3) x = 0. 14 M [H 2 PO 4 -] = [H 3 O+] = 0. 14 M 30 General Chemistry: Chapter 16 Prentice-Hall © 2007

EXAMPLE 16 -9 H 2 PO 4 - + H 2 O HPO 42 - + H 3 O+ Initial conc. 0. 14 M 0 0. 14 M Changes -y M +y M Equilibrium Concentration (0. 14 - y) M y. M (0. 14 +y) M K a= [H 3 O+] [HPO 42 -] [H 2 PO 4 -] y << 0. 14 M 31 = y · (0. 14 + y) (0. 14 - y) = 6. 3 10 -8 y = [HPO 42 -] = 6. 3 10 -8 General Chemistry: Chapter 16 Prentice-Hall © 2007

EXAMPLE 16 -9 HPO 4 - + K a= H 2 O [H 3 O+] [HPO 42 -] [H 2 PO 4 -] PO 43 - + H 3 O+ = (0. 14)[PO 43 -] 6. 3 10 -8 = 4. 2 10 -13 M [PO 43 -] = 1. 9 10 -19 M 32 General Chemistry: Chapter 16 Prentice-Hall © 2007

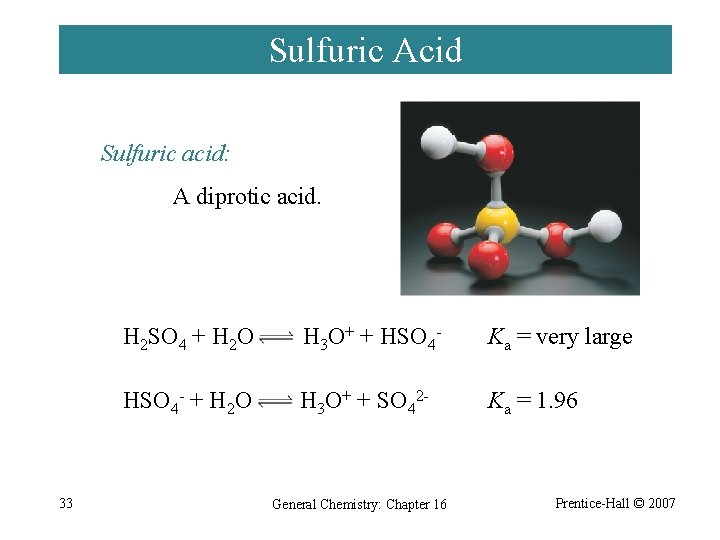
Sulfuric Acid Sulfuric acid: A diprotic acid. 33 H 2 SO 4 + H 2 O H 3 O+ + HSO 4 - Ka = very large HSO 4 - + H 2 O H 3 O+ + SO 42 - Ka = 1. 96 General Chemistry: Chapter 16 Prentice-Hall © 2007

General Approach to Solution Equilibrium Calculations ¨ Identify species present in any significant amounts in solution (excluding H 2 O). ¨ Write equations that include these species. · Number of equations = number of unknowns. ◦ Equilibrium constant expressions. ◦ Material balance equations. ◦ Electroneutrality condition. ¨ Solve the system of equations for the unknowns. 34 General Chemistry: Chapter 16 Prentice-Hall © 2007

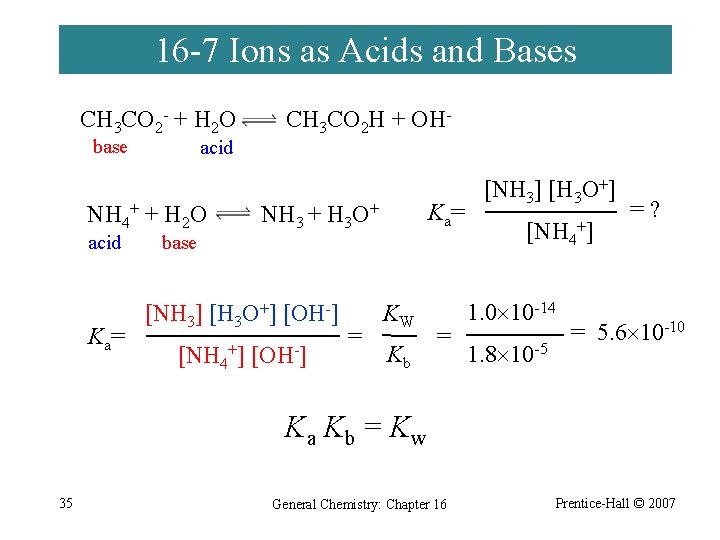
16 -7 Ions as Acids and Bases CH 3 CO 2 - + H 2 O base acid NH 4+ + H 2 O acid K a= base CH 3 CO 2 H + OH- K a= NH 3 + H 3 O+ [NH 3] [H 3 O+] [OH-] [NH 4+] [OH-] = KW Kb = [NH 3] [H 3 O+] [NH 4+] 1. 0 10 -14 1. 8 10 -5 =? = 5. 6 10 -10 Ka Kb = Kw 35 General Chemistry: Chapter 16 Prentice-Hall © 2007

Hydrolysis ¨ Water (hydro) causing cleavage (lysis) of a bond. 36 Na+ + H 2 O → Na+ + H 2 O No reaction Cl- + H 2 O → Cl- + H 2 O No reaction NH 4+ + H 2 O → NH 3 + H 3 O+ Hydrolysis General Chemistry: Chapter 16 Prentice-Hall © 2007

16 -8 Molecular Structure and Acid-Base Behavior ¨ Why is HCl a strong acid, but HF is a weak one? ¨ Why is CH 3 CO 2 H a stronger acid than CH 3 CH 2 OH? ¨ There is a relationship between molecular structure and acid strength. ¨ Bond dissociation energies are measured in the gas phase and not in solution. 37 General Chemistry: Chapter 16 Prentice-Hall © 2007

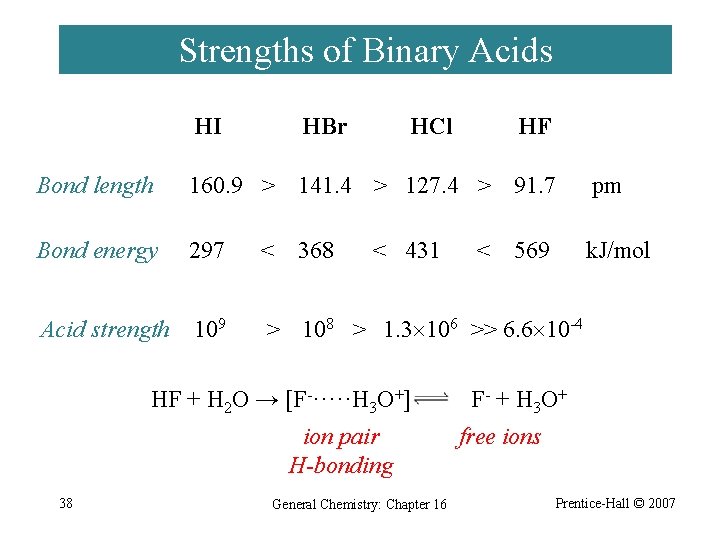
Strengths of Binary Acids HI HBr HCl HF Bond length 160. 9 > 141. 4 > 127. 4 > 91. 7 pm Bond energy 297 < 368 k. J/mol Acid strength 109 > 108 > 1. 3 106 >> 6. 6 10 -4 < 431 HF + H 2 O → [F-·····H 3 O+] ion pair H-bonding 38 General Chemistry: Chapter 16 < 569 F - + H 3 O + free ions Prentice-Hall © 2007

Strengths of Oxoacids ¨ Factors promoting electron withdrawal from the OH bond to the oxygen atom: · High electronegativity (EN) of the central atom. · A large number of terminal O atoms in the molecule. 39 H-O-Cl H-O-Br ENCl = 3. 0 ENBr= 2. 8 Ka = 2. 9 10 -8 Ka = 2. 1 10 -9 General Chemistry: Chapter 16 Prentice-Hall © 2007

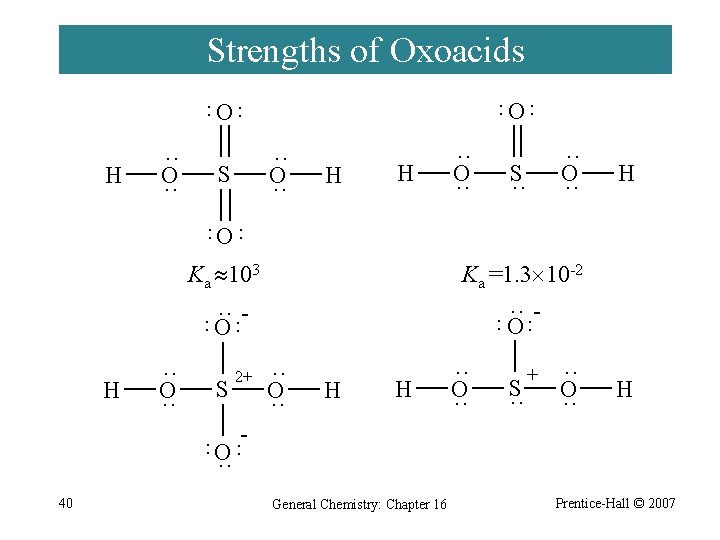
Strengths of Oxoacids O ·· ·· O H ·· O ·· S H H ·· O ·· S ·· H ·· ·· O Ka 103 Ka =1. 3 10 -2 ·· O ·· ·· ·· O H ·· O ·· S ·· O ·· H H ·· O ·· S ·· + ·· O ·· H - ·· ·· O ·· 2+ 40 General Chemistry: Chapter 16 Prentice-Hall © 2007

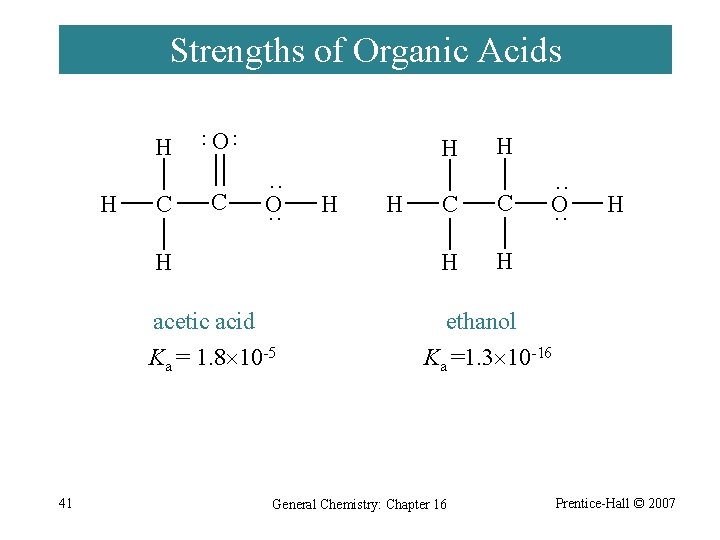
Strengths of Organic Acids H C O ·· ·· H C H ·· O ·· H acetic acid Ka = 1. 8 10 -5 41 H H H C C H H ·· O ·· H ethanol Ka =1. 3 10 -16 General Chemistry: Chapter 16 Prentice-Hall © 2007