Petrucci Harwood Herring Madura GENERAL CHEMISTRY Ninth Edition























- Slides: 30

Petrucci • Harwood • Herring • Madura GENERAL CHEMISTRY Ninth Edition Principles and Modern Applications Chapter 19: Spontaneous Change: Entropy and Free Energy 1 General Chemistry: Chapter 19 Prentice-Hall © 2007

Contents 19 -1 19 -2 19 -3 19 -4 19 -5 19 -6 19 -7 2 Spontaneity: The Meaning of Spontaneous Change The Concept of Entropy Evaluating Entropy and Entropy Changes Criteria for Spontaneous Change: The Second Law of Thermodynamics Standard Free Energy Change, ΔG° Free Energy Change and Equilibrium ΔG° and Keq as Functions of Temperature General Chemistry: Chapter 19 Prentice-Hall © 2007

19 -1 Spontaneity: The Meaning of Spontaneous Change 3 General Chemistry: Chapter 19 Prentice-Hall © 2007

Spontaneous Process ¨ A process that occurs in a system left to itself. · Once started, no external actions is necessary to make the process continue. ¨ A non-spontaneous process will not occur without external action continuously applied. 4 Fe(s) + 3 O 2(g) → 2 Fe 2 O 3(s) H 2 O(s) 4 H 2 O(l) General Chemistry: Chapter 19 Prentice-Hall © 2007

Spontaneous Process ¨ Potential energy decreases. ¨ For chemical systems the internal energy U is equivalent to potential energy. ¨ Berthelot and Thomsen 1870’s. · Spontaneous change occurs in the direction in which the enthalpy of a system decreases. · Mainly true but there are exceptions. 5 General Chemistry: Chapter 19 Prentice-Hall © 2007

19 -2 The Concept of Entropy ¨ Entropy, S. ΔU = ΔH = 0 · The greater the number of configurations of the microscopic particles among the energy levels in a particular system, the greater the entropy of the system. ΔS > 0 spontaneous 6 General Chemistry: Chapter 19 Prentice-Hall © 2007

The Boltzmann Equation for Entropy S = k ln. W ¨ Entropy, S ¨ States. · The microscopic energy levels available in a system. ¨ Microstates, W. · The particular way in which particles are distributed amongst the states. Number of microstates = W. ¨ The Boltzmann constant, k. · Effectively the gas constant per molecule = R/NA. 7 General Chemistry: Chapter 19 Prentice-Hall © 2007

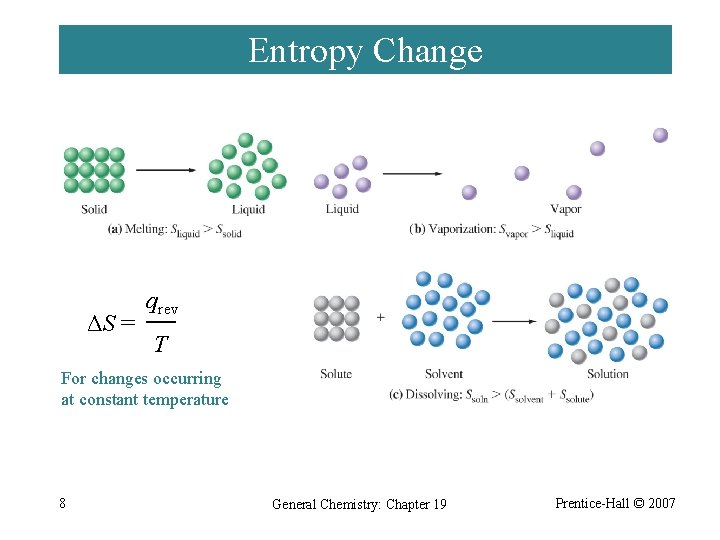
Entropy Change ΔS = qrev T For changes occurring at constant temperature 8 General Chemistry: Chapter 19 Prentice-Hall © 2007

19 -3 Evaluating Entropy and Entropy Changes ¨ Phase transitions. · Exchange of heat can be carried out reversibly. ΔS = H 2 O(s, 1 atm) ΔH Ttr H 2 O(l, 1 atm) ΔHfus = 6. 02 k. J at 273. 15 K ° ΔHfus 6. 02 k. J mol-1 ΔSfus = = = 2. 20 10 -2 k. J mol-1 K-1 Ttr 273. 15 K 9 General Chemistry: Chapter 19 Prentice-Hall © 2007

Trouton’s Rule ΔS = 10 ΔHvap Tbp 87 k. J mol-1 K-1 General Chemistry: Chapter 19 Prentice-Hall © 2007

Absolute Entropies ¨ Third law of thermodynamics. · The entropy of a pure perfect crystal at 0 K is zero. ¨ Standard molar entropy. · Tabulated in Appendix D. ΔS = [ p. S°(products) - r. S°(reactants)] 11 General Chemistry: Chapter 19 Prentice-Hall © 2007

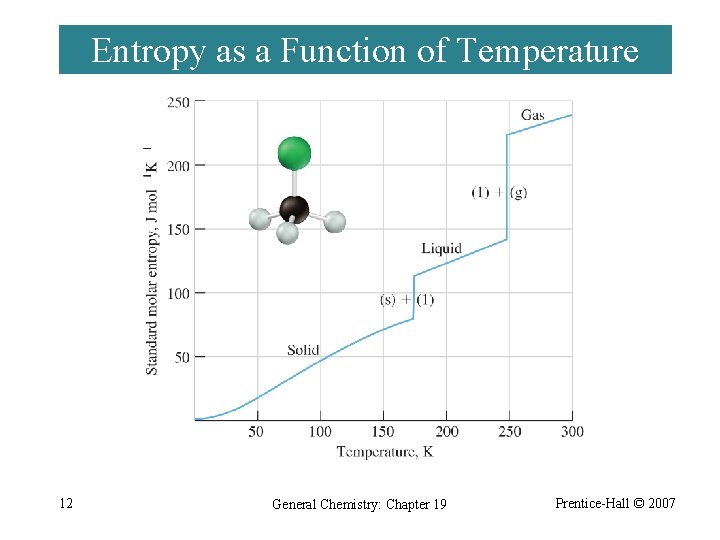
Entropy as a Function of Temperature 12 General Chemistry: Chapter 19 Prentice-Hall © 2007

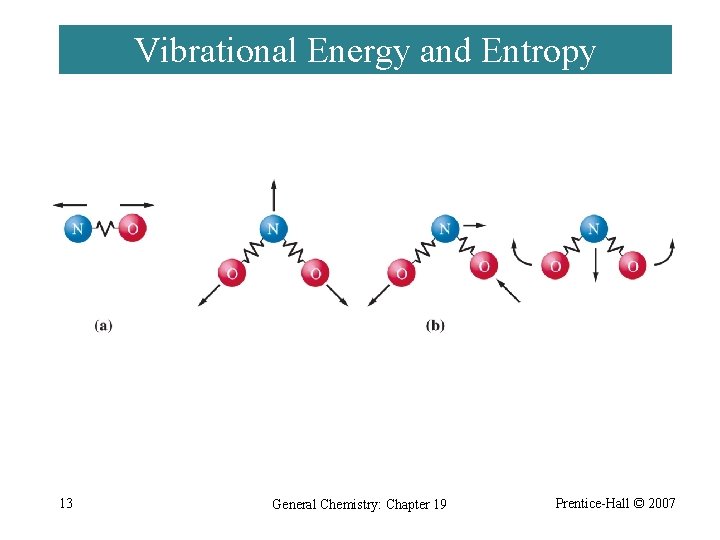
Vibrational Energy and Entropy 13 General Chemistry: Chapter 19 Prentice-Hall © 2007

19 -4 Criteria for Spontaneous Change: The Second Law of Thermodynamics. ΔStotal = ΔSuniverse = ΔSsystem + ΔSsurroundings The Second Law of Thermodynamics: ΔSuniverse = ΔSsystem + ΔSsurroundings > 0 All spontaneous processes produce an increase in the entropy of the universe. 14 General Chemistry: Chapter 19 Prentice-Hall © 2007

Free Energy and Free Energy Change ¨ Hypothetical process: · only pressure-volume work, at constant T and P. qsurroundings = -qp = -ΔHsys ¨ Make the enthalpy change reversible. · large surroundings, infinitesimal change in temperature. ¨ Under these conditions we can calculate entropy. 15 General Chemistry: Chapter 19 Prentice-Hall © 2007

Free Energy and Free Energy Change For the universe: TΔSuniv. = TΔSsys – ΔHsys = -(ΔHsys – TΔSsys) -TΔSuniv. = ΔHsys – TΔSsys For the system: G = H - TS ΔG = ΔH - TΔS ΔGsys = - TΔSuniverse 16 General Chemistry: Chapter 19 Prentice-Hall © 2007

Criteria for Spontaneous Change ΔGsys < 0 (negative), the process is spontaneous. ΔGsys = 0 (zero), the process is at equilibrium. ΔGsys > 0 (positive), the process is non-spontaneous. J. Willard Gibbs 1839 -1903 17 General Chemistry: Chapter 19 Prentice-Hall © 2007

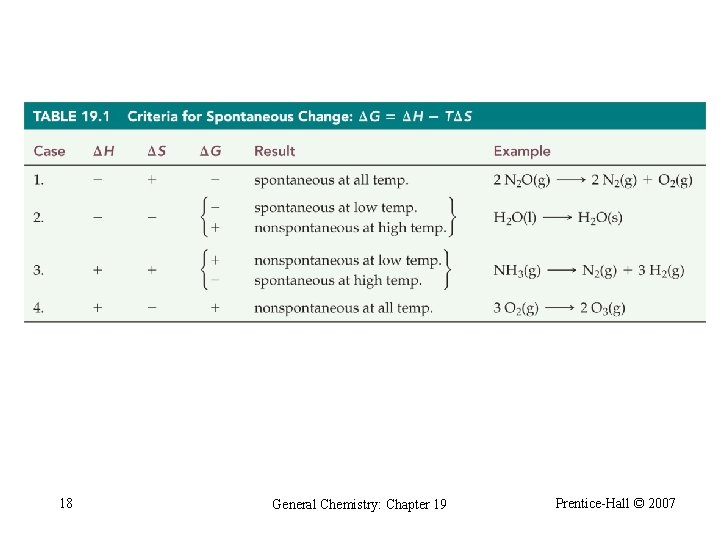
Table 19. 1 Criteria for Spontaneous Change 18 General Chemistry: Chapter 19 Prentice-Hall © 2007

19 -5 Standard Free Energy Change, ΔG° ¨ The standard free energy of formation, ΔGf°. · The change in free energy for a reaction in which a substance in its standard state is formed from its elements in reference forms in their standard states. ¨ The standard free energy of reaction, ΔG° = [ p ΔGf°(products) - r ΔGf°(reactants)] 19 General Chemistry: Chapter 19 Prentice-Hall © 2007

Free Energy Change and Equilibrium Condensation 20 Equilibrium General Chemistry: Chapter 19 Vaporization Prentice-Hall © 2007

Relationship of ΔG° to ΔG for Non-standard Conditions 2 N 2(g) + 3 H 2(g) ΔG = ΔH - TΔS 2 NH 3(g) ΔG° = ΔH° - TΔS° For ideal gases ΔH = ΔH° ΔG = ΔH° - TΔS 21 General Chemistry: Chapter 19 Prentice-Hall © 2007

Relationship Between S and S° qrev = -w = RT ln Vf Vi qrev Vf = R ln ΔS = T Vi Pf Pi Vf = R ln = -R ln ΔS = Sf – Si = R ln Pi Pf Vi P P = S° - R ln S = S° - R ln 1 P° 22 General Chemistry: Chapter 19 Prentice-Hall © 2007

N 2(g) + 3 H 2(g) SN 2 = SN 2 – Rln PN 2 2 NH 3(g) SH 2 = SH 2 – Rln PH 2 SNH 3 = SNH 3 – Rln PNH 3 ΔSrxn = 2 (SNH 3 – Rln PNH 3) – (SN 2 – Rln PN 2) – 3 (SH 2 – Rln PH 2) ΔSrxn = 2 SNH 3 – SN 2 – 3 SH 2+ Rln ΔSrxn = ΔS°rxn + Rln 23 PN 2 PH 3 2 2 PNH 3 General Chemistry: Chapter 19 Prentice-Hall © 2007

ΔG Under Non-standard Conditions ΔG = ΔH° - TΔS ΔSrxn = ΔS°rxn + Rln ΔG = ΔH° - TΔS°rxn – TR ln PN 22 PH 3 2 2 PNH 3 2 ΔG = ΔG° + RT ln PNH 3 PN 22 PH 3 2 ΔG = ΔG° + RT ln Q 24 General Chemistry: Chapter 19 Prentice-Hall © 2007

ΔG and the Equilibrium Constant Keq ΔG = ΔG° + RT ln Q If the reaction is at equilibrium then: ΔG = ΔG° + RT ln Keq= 0 ΔG° = -RT ln Keq 25 General Chemistry: Chapter 19 Prentice-Hall © 2007

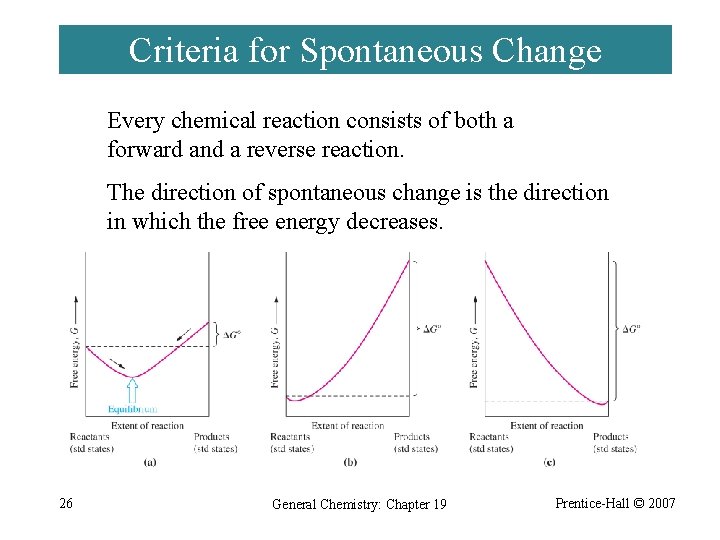
Criteria for Spontaneous Change Every chemical reaction consists of both a forward and a reverse reaction. The direction of spontaneous change is the direction in which the free energy decreases. 26 General Chemistry: Chapter 19 Prentice-Hall © 2007

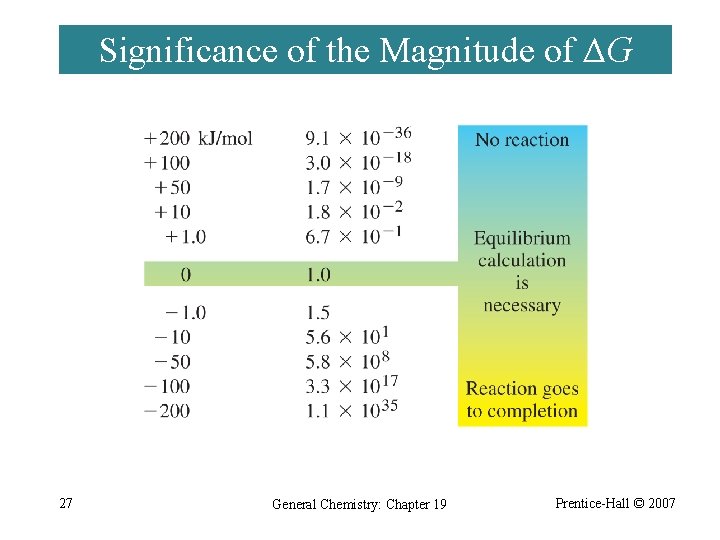
Significance of the Magnitude of ΔG 27 General Chemistry: Chapter 19 Prentice-Hall © 2007

19 -7 ΔG° and Keq as Functions of Temperature ΔG° = ΔH° -TΔS° ΔG° = -RT ln Keq -ΔG° -ΔH° TΔS° ln Keq = = + RT RT RT -ΔH° ΔS° ln Keq = + RT R 28 General Chemistry: Chapter 19 Prentice-Hall © 2007

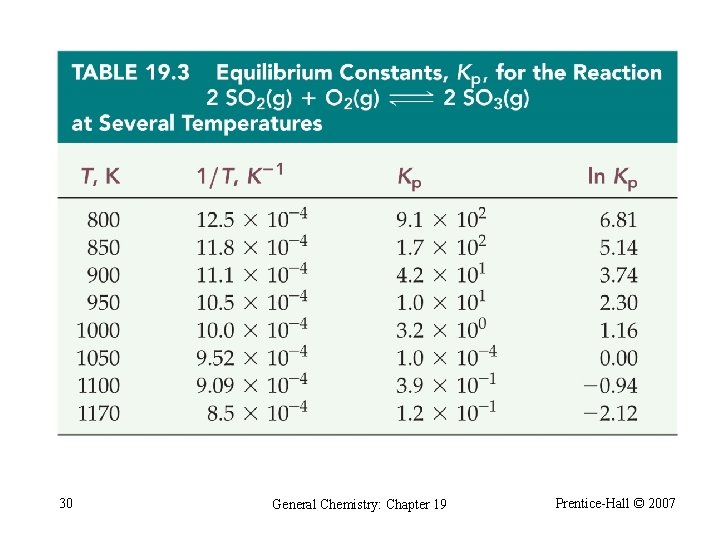
Van’t Hoff Equation If we evaluate this equation for a change in temperature: ln Keq 2 Keq 1 -ΔH° ΔS° = + + RT 2 R RT 1 R ln 29 Keq 2 Keq 1 1 -ΔH° 1 = R T 2 T 1 General Chemistry: Chapter 19 Prentice-Hall © 2007

30 General Chemistry: Chapter 19 Prentice-Hall © 2007