PETROLEUM Liquid Hydrocarbons Petroleum along with coal and

PETROLEUM Liquid Hydrocarbons • Petroleum, along with coal and natural gas, is classified as a fossil fuel. • Petroleum is a complicated liquid mixture of organic compounds. The main components of petroleum are hydrocarbons. • Petroleum also has varying proportions of sulfur-, nitrogen, and oxygen- containing compounds. 1

How do petroleum find ? • Petroleum in its natural form when first collected is usually named crude oil, oil and can be clear, green or black and may be either thin like gasoline or thick like tar. • Petroleum deposits nearly always have associated natural gas, part or all of which is sent through a separation process. 2

Petroleum formation • Millions of years ago various creatures roamed the surface of this earth. Some of these creatures have become extinct, while there are some which still exist today. • Some of them made their way to the ocean after their dead and sunk thousands of feet below to finally settle on the seabed. Both plants and animals settled on the seabed after their death. 3

How does the extraction and refining of petroleum take place? • The process of extraction of petroleum begins with the identifying where exactly the petroleum is present. • Once the petroleum presence has been noted and recorded, a well is drilled to reach the underground reservoir of petroleum. 4
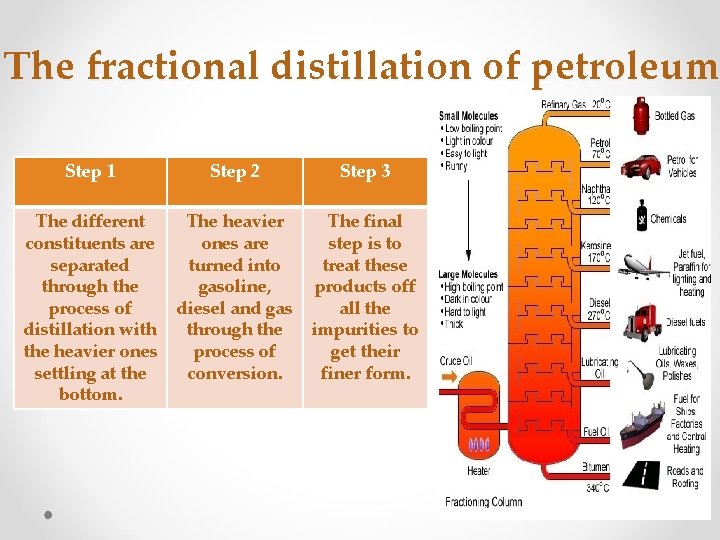
The fractional distillation of petroleum Step 1 Step 2 Step 3 The different constituents are separated through the process of distillation with the heavier ones settling at the bottom. The heavier ones are turned into gasoline, diesel and gas through the process of conversion. The final step is to treat these products off all the impurities to get their finer form. 5
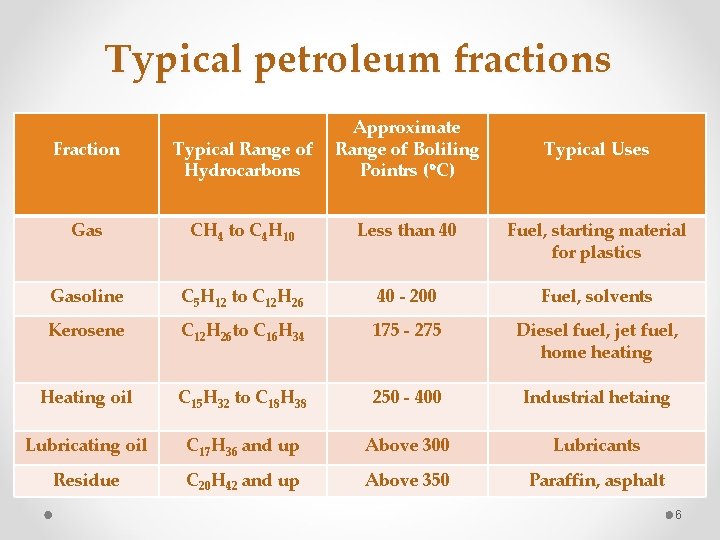
Typical petroleum fractions Fraction Typical Range of Hydrocarbons Approximate Range of Boliling Pointrs ( C) Gas CH 4 to C 4 H 10 Less than 40 Fuel, starting material for plastics Gasoline C 5 H 12 to C 12 H 26 40 - 200 Fuel, solvents Kerosene C 12 H 26 to C 16 H 34 175 - 275 Diesel fuel, jet fuel, home heating Heating oil C 15 H 32 to C 18 H 38 250 - 400 Industrial hetaing Lubricating oil C 17 H 36 and up Above 300 Lubricants Residue C 20 H 42 and up Above 350 Paraffin, asphalt Typical Uses 6
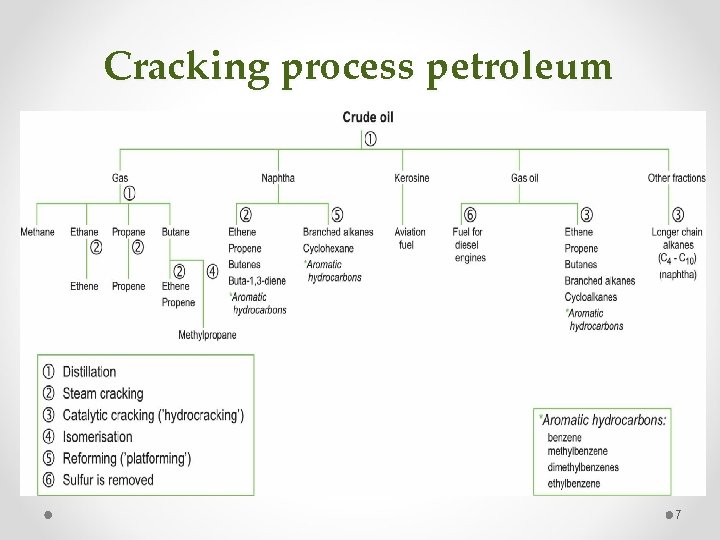
Cracking process petroleum 7

What is the equation for petroleum combustion • Petroleum is largely a mixture of hydrocarbons, and hydrocarbons burn readily. As with natural gas, complete combustion yields mainly carbon dioxide and water. 2 C 8 H 18 + 25 O 2 16 CO 2 + 18 H 2 O • Combustion in air also leads to the formation of nitrogen oxides. Incomplete burning produces carbon monoxide and soot. Petroleum usually contains small amounts of sulfur compounds that produce sulfur dioxide when burned. 8

Environmental effect of petroleum Ø Efficiently burned, petroleum products are rather clean fuels. Ø Fuel oil, used for heating homes or to produce electricity, can be burned efficiently and thus contributes only moderately to air pollution. Ø Gasoline, however, the major fraction of petroleum, is used to power automobiles; because the internalcombustion engines in most automobiles are rather inefficient, the combustion of gasoline contributes heavily to air pollution. 9
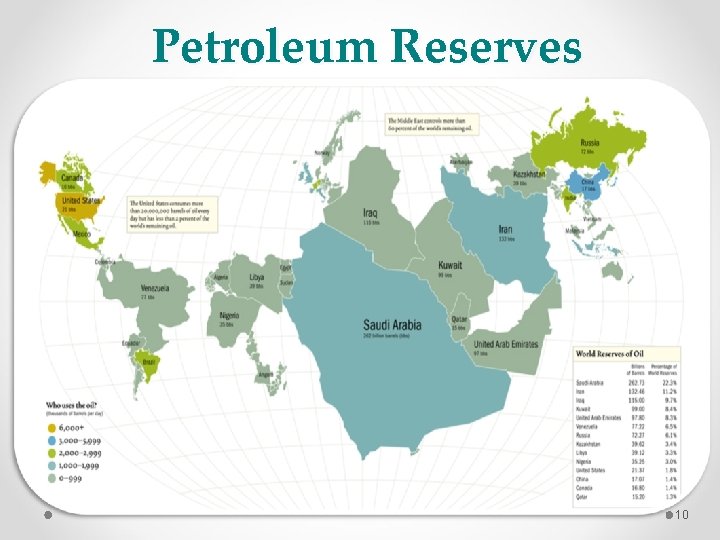
Petroleum Reserves 10

What are petroleum products, and what is petroleum used for? • The most important products and by-products are: petrol, paraffin, diesel, gas oil, and petro-chemical by-products – plastic, detergents, aviation gasoline, neptha, mobil, grease, vaseline, wax, asphalt, etc. • In its thickest form, the almost black petroleum is named bitumen, this is used for paving road, forming the blacktop, it is also an excellent water repellent and is used in roofing. • 11
- Slides: 11