Peripheral Vascular Intervention PVI Device Utilization and Patient














- Slides: 16

Peripheral Vascular Intervention (PVI) Device Utilization and Patient Outcomes Lillian Camino, RPVI, RVT; Melissa Easterday BS; Gary Lemmon, MD Indiana University Health Methodist Hospital Indianapolis, Indiana

Disclosures We do not have any actual or potential conflict of interest in relation to this presentation.

SCAI Consensus Guideline for Device Selection in Femoral‐Popliteal Arterial Interventions In 2018, ACC/AHA/SCAI/SIR/SVM released a publication regarding appropriate use criteria for Peripheral Artery Intervention. – Journal of the American College of Cardiology Dec 2018, 25614; DOI: 10. 1016/j. jacc. 2018. 10. 002 Green: Class 1 strong recommendation These are DES and DCB

Paclitaxel‐Coated Balloons and Stents in the Femoropopliteal Artery of the Leg In 2018 JAHA released an article indicating increased risk of death following application of paclitaxel‐coated balloons and stents in the femoropopliteal artery of the lower limbs. – In 2018, Risk of Death Following Application of Paclitaxel‐Coated Balloons and Stents in the Femoropopliteal Artery of the Leg: A Systematic Review and Meta‐Analysis of Randomized Controlled – – Ie “Konstantinos Katsanos study” Journal of the American Heart Association Dec 18, 2018, Vol 7, Issue 24 https: //doi. org/10. 1161/JAHA. 118. 011245 In 2019 FDA released a series or letters to Health Care Providers regarding Paclitaxel Coated balloons and stents in PVI – https: //www. fda. gov/medical‐devices/letters‐health‐care‐ providers/update‐treatment‐peripheral‐arterial‐disease‐paclitaxel‐ coated‐balloons‐and‐paclitaxel‐eluting

Global Vascular Guidelines (GVG) for patients with critical limb‐threatening Recently in 2019 JVS and ESVS copublished Global Vascular Guidelines (GVG) for patients with critical limb-threatening ischemia – Representation of • Society for Vascular Surgery (SVS), • European Society for Vascular Surgery (ESVS), and • World Federation of Vascular Societies (WFVS), – https: //doi. org/10. 1016/j. jvs. 2019. 04. 469 Just Released

Goal Indiana University Health (IUH) Methodist Hospital (MH) : Review device specific procedural outcomes for LE PVI – Analyze the amount and type of devices used in Femoral‐popliteal atherosclerotic disease treatment. • Immediate and long term outcomes of the procedure • Determine efficacy of treatment at follow up timeframes

Method Type: Retrospective analysis Software: – VQI PVI Registry Database – Microsoft Excel Timeframe: 2012‐ 2018 Pathology: Atherosclerotic occlusive disease only (ASO) Segments (native vessels only): – Superficial Femoral (SFA) – Popliteal (Pop) – “SFA/Pop” Cases presented are PER Segment AND SIDE treated – If one case has R‐SFA and L‐Pop, it will count as 2 cases Symptoms and outcomes adjusted by laterality – right segments were matched only with right sided data (R‐ segments w R‐sided symptoms and R‐sided amputations, etc)

Method Definitions: Non-critical limb ischemia Target Lesion Revascularization (TLR): (NCLI) : – asymptomatic – claudicant Critical limb ischemia (CLI): – – rest pain ulcers non‐healing amputations acute ischemia cases – percutaneous PVI* – open bypass graft* *(related to the area treated) Major Amputations: – above the knee amputation (AKA) – below the knee amputation (BKA) * Ipsilateral to area treated

Method A = Atherectomy – – Directional Orbital Excisional Laser SG = Stentgrafts – Covered stents ST = Stents – Balloon expanded – Self expanded – Drug eluting stents SB = Special Balloons – Drug coated balloons – Cryoplasty – Cutting Balloons PTA = Simple Balloons – Uncoated balloons Order of entry of devices (when more devices used than registry capacity to collect): – 1 st Atherectomies – 2 nd Stentgrafts – 3 rd Stents – 4 th Special Balloons – 5 th Plain Balloons

Results We were able to collect and analyze robust amount of data We will present here the highlights of the data

Results 1012 Patients treated – 1004 Patients with only 1 limb treated – 8 Patients with bilateral limbs treated R≈L 1108 segments treated – 901 with 1 segment treated – 102 with 2 segments treated – 1 with 3 segments treated Symptoms at index: – NCLI: 553/1108 – CLI: 540/1108 – Unknown: 15/1108 PTA Alone was performed on 245/1108 (22% of all cases)

Results Outcomes at LTFU 620 Segments with LTFU data 488 no LTFU IUH-MH averages 64. 8% compliance of LTFU for PVI module at the time of study Unchanged Symptoms Worsening of Symptoms (281/620) (21/620) (75/620) (43/620) • 84. 3% NCLI (237/281) • 100% NCLI (21/21) • 37. 3% NCLI (28/75) • 14. 0% NCLI (6/43) • 61. 3% CLI (46/75) • 86. 0% CLI (37/43) 45. 3% • 15. 7% CLI (44/281) 3. 4% TLR 12. 1% Major Amputation 6. 9%

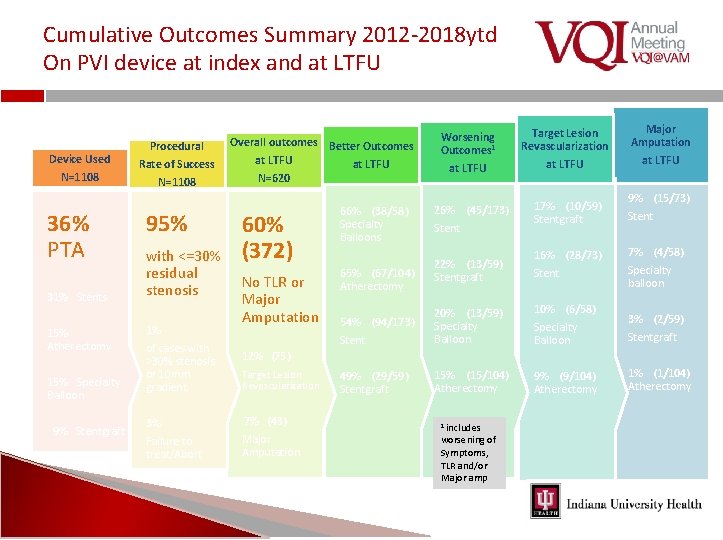
Cumulative Outcomes Summary 2012‐ 2018 ytd On PVI device at index and at LTFU Device Used N=1108 36% PTA 31% Stents 15% Atherectomy 15% Specialty Balloon 9% Stentgraft Procedural Rate of Success N=1108 95% with <=30% residual stenosis 1% of cases with >30% stenosis or 10 mm gradient 3% Failure to treat/Abort Overall outcomes Better Outcomes at LTFU N=620 60% (372) No TLR or Major Amputation 66% (38/58) Specialty Balloons Worsening Outcomes 1 Target Lesion Revascularization at LTFU 26% (45/173) 17% (10/59) Stentgraft Stent Major Amputation at LTFU 9% (15/73) Stent 22% (13/59) Stentgraft 16% (28/73) Stent 7% (4/58) Specialty balloon 54% (94/173) Stent 20% (13/59) Specialty Balloon 10% (6/58) Specialty Balloon 3% (2/59) Stentgraft 49% (29/59) Stentgraft 15% (15/104) Atherectomy 9% (9/104) Atherectomy 1% (1/104) Atherectomy 65% (67/104) Atherectomy 12% (75) Target Lesion Revascularization 7% (43) Major Amputation 1 includes worsening of Symptoms, TLR and/or Major amp

Challenges/Limitations VQI/Pathways Analytic and Reporting Engine unable to produce this type of analysis (Manual Analysis) Limitations in amount of devices captured – Only 2 devices captured 2012‐ 2015 – Only 3 devices captured 2016‐ 2018 – Treatment options may require >>3 devices LTFU void Incomplete/Missing data Specialty balloon descriptors not captured at the time – Drug coated balloons – Cryoplasty – Cutting Balloons Inadequate statistical power – single site WIFI scoring impact on PVI Underestimated time needed to complete this review!!!!

Conclusions Deep dive analysis was possible using VQI PVI raw data We found value in analysis of VQI data – to review internal treatment strategies and patient outcomes. Preconceived notions/bias of device performance could not be reproduced – all devices performed well when compared to each other. The review supports the adoption of current practices to align with the recently published SCAI PVI guidelines. Insights from the initial review will guide future continued tracking New PVI module update may allow future review of Paclitaxel containing devices data review* *Registry limited to 3 or less devices

Thank you