PERIOPERATIVE SAFETY Microsoft Clipart Table of Contents 3
PERIOPERATIVE SAFETY Microsoft Clipart

Table of Contents 3 Laser 16 Electrosurgery 49 Universal Protocol 62 Sentinel Events 64 Counts 81 Sharps Safety 83 Hypothermia 88 NPSG 91 Radiation safety 100 Chemical Hazards 109 Specimens

Laser Safety LASER is an acronym for Light Amplification by Stimulated Emission of Radiation LASER consists of: an optical cavity, a pumping system, and an appropriate lasing medium ©images. MD, 2006

Laser Safety Microsoft Clipart optical cavity: media excited with mirrors to redirect the produced photons back along the same path pumping system: uses photons from another source as a xenon gas flash tube to transfer energy to media, electrical discharge within the pure gas or gas mixture media, thus binding energy released in chemical reactions to raise the media to a “lasing” state. laser medium can be a solid (state), gas, dye (in liquid), or semiconductor

Laser Types Solid state lasers have lasing material distributed in a solid matrix (ie. YAG laser) Gas lasers (helium and helium-neon, He. Ne) are the most common gas lasers CO 2 lasers are also utilized. Excimer lasers (the name is derived from the terms excited and dimers) use reactive gases such as chlorine and fluorine mixed with inert gases such as argon, krypton, or xenon, then produces ultraviolet light.

Laser Safety Eye Injury: unprotected laser exposure may result in eye injuries including retinal burns, cataracts, and permanent blindness. Lasers produce invisible ultraviolet, or other radiation, both employees and visitors should use appropriate eye protection at all times. Signs must be posted whenever a laser is in use ANSIZ 136. 3 - Procedural & Equipment Controls for Class IV Laser System www. osha. gov

Laser Safety Selection of laser protection depend upon the laser in use. Workers with exposure to laser beams must be furnished suitable laser protection. Eyes are the most critical organ for damage during laser procedures. Account for 69. 8% of Laser accidents. www. osha. gov

Laser Safety ANSI recommends baseline eye exams Laser goggles should be checked for ◦ ◦ ◦ Cracking Pitting Discoloration Frame condition Light leaks

Laser Safety Maximum Permissible Exposure (MPE): amount of laser radiation to which individuals may be safely exposed without a hazardous effect Lasers should be placed on “Standby” mode when not in use Sterile water should be kept readily available in case of fire

Laser Safety ü ü ü ü Laser Log documentation should at minimum include the following: Patient identification information Type of laser Model number/Biomed number Procedure Names of individuals in the room Information that protective glasses were offered to and worn by all (or list exceptions) Completed laser checklist Number of joules used Wattage ENT cases document Laser Time on and off Mode Delivery System Wavelength

AORN Standards and Recommended Practice and Guidelines for Laser Safety Recommended Practice I: “A Laser Safety should be established for private practitioners and/or health care facilities” Recommended Practice II: “ Personnel working in laser environments should demonstrate competency commensurate with their responsibilities. Education programs should be specific to laser systems used and procedures performed in the facility. ”

AORN Standards and Recommended Practice and Guidelines for Laser Safety Recommended Practice III: “All people should know where lasers are being used, and access to these areas should be controlled. ” Recommended Practice IV: “All people in the laser treatment area should be protected from unintentional laser beam exposure. ” The laser operator managing the laser equipment should have no competing responsibilities that would require leaving the laser unattended during active use.

AORN Standards and Recommended Practice and Guidelines for Laser Safety Recommended Practice V: “All people in the nominal zone should wear appropriate eye wear approved by the laser safety officer. ” Recommended Practice VI: “ Personnel working in the laser environment should avoid exposure to laser plume generated during laser surgery. ” Smoke plume evacuation should be implemented High filtration masks are used during laser procedures

AORN Standards and Recommended Practice and Guidelines for Laser Safety Recommended Practice VII: “ All people in the laser treatment area should be protected from electrical hazards associated with laser use. ” Recommended Practice VIII: “All people should be protected from flammability hazards with laser use. ” Class 4 laser has potential for fire with anesthetic gases, solutions, ointments and plastic resins. A fire extinguisher should be in close proximity to the laser procedure room

AORN Standards and Recommended Practice and Guidelines for Laser Safety Recommended Practice IX: “Policies and Procedures for laser safety should be with regard to individual practice settings, applicable to standards, federal and state regulations.

Steps to be taken in Management of an Airway Explosion According to ANSI Standard C 9. 4 Order of Priority is as follows: ◦ Stop Ventilation ◦ Disconnect oxygen source, flood with saline if needed ◦ Remove tracheal tube/endoscope ◦ Mask ventilate and reintubate ◦ Diagnosis of injury (bronchoscopy) ◦ Monitor patient 24 hours ◦ Administration of short term steroids ◦ Administer antibiotics ◦ Ventilatory support

Electrosurgery Terms Active Electrode: an accessory that directs current flow to the surgical site (pencils, tips) ©Conmed, Used by permission

Electrosurgery Terms Generator: machine that produces radio frequency waves ©Conmed, Used by permission Dispersive electrode: accessory that directs electrical current flow from the patient back to the surgical generator (called “grounding pad”, “plate”, “return electrode”) Dispersive electrode gel pad on bed ©Conmed, Used by permission

Electrosurgery Terms Monopolar electrosurgery: only the active electrode is in the surgical wound, electrical current is then directed through the patient’s body, received by the dispersive pad and transferred back to the generator. Bipolar electrosurgery: current flows between two tips of bipolar forceps which is positioned around the tissue to create hemostasis and then returns to the electrosurgical unit.

Electrosurgery Terms Electrosurgery is on most surgical procedures. As current passes through the tissue, heat is generated to produce cutting/coagulation.

Electrosurgical Components Generator ©Conmed, Used by permission Active Electrode Dispersive Electrode ©Conmed, Used by permission

Electrosurgery History 1920’s Dr. William Bovie developed the first spark gap vacuum tube generator that produced cutting with hemostasis This was the basis until the 1970’s, when solid state electrosurgical units were introduced. (Referred to as “Bovie”) These early ESU presented significant risk for burns or shock injury. Today, generators are solid state and isolated systems which dramatically reduce burns and shock injury risk.

Generators Designed to deliver current that will cut, coagulate or combine the two ◦ Cut: continuous frequency waveform causes cutting. Tissue is severed by intense heat from the active electrode. Active electrode is held slightly above the tissue. ◦ Coagulation: interrupted frequency waveform causes this to occur. Active electrode is in direct contact with tissue, ends of small to moderate size vessels are seared and bleeding is stopped.

Generators ◦ Combination of cut and coagulation: occurs simultaneously • Power and type of current are regulated by controls on the ESU generator and active electrode • Selection of the type of current and amount of power is selected by the surgeon, which is determined by the procedure and preference.

Active Electrode Delivers current from the generator to the surgical site. Most are disposable handheld devices. Active electrodes come with different tips (blade, ball, loop, hook, needle) Activated by hand or foot pedal ©Conmed, Used by permission

Active Electrode Bipolar electrodes are shaped like forceps with tips. One tip acts as the active electrode, the other tip acts as the return or dispersive electrode. Current flows from generator to forceps through tissue between forcep tips returned to generator through the other tip of the forceps. Since current does not disperse through the dispersive pad, no dispersive electrode (grounding pad) is needed.

Dispersive Electrode Referred to as “Bovie pad”, “patient plate”, “return electrode”. Current is dissipated through the hand piece to the patient and returned to generator via the dispersive electrode ©Conmed, Used by permission

Dispersive Electrode Dispersive electrode is much larger than active electrode, current density is low, therefore burns usually do not occur May be reusable or disposable Reusable made from metal have been replaced with a pressure reduction pad and have current limiting abilities to protect the patient from pad site burns (ie. Megadyne) Pad is placed on OR table to maximize contact with the patient. A cord connects pad to the generator

Dispersive Electrode Most commonly used is the disposable adhesive foil pad covered with foam and impregnated with electrolyte gel. This pad comes in direct contact with the patients skin Pad should have uniform contact with patients skin Newer generators identify potential current concentration that may cause a burn, if this occurs the machine will alarm and deactivate (scenario: if pad loosens from patient skin, “tents” etc. )

Dispersive Electrode Application Check return electrode for appropriate size, integrity, conductive gel and expiration date. Document on OR record. Prep site. Remove hair if necessary. Clean site of any lotions/gels/grease. Do not cut or fold electrode. (must be large enough to prevent condition of current at return site.

Dispersive Electrode Application Place the on clean dry skin, close to the surgical site, over a LARGE MASS OF WELL PERFUSED MUSCLE. DO NOT place on bony prominences, scar tissue, hairy surfaces, distal to tourniquets or other pressure points. DO NOT place over an implant metal prosthesis. DO NOT place over a tattoo. Place after final positioning, to prevent buckling and ensure good skin contact is maintained

Dispersive Electrode Application Keep from pooling fluids. Fluids can interfere with pad-skin contact. Make sure patient is not in contact with any metal objects Remove jewelry. Tape over the object is not adequate If multiple ESU’s are in use, use a separate pad for each ESU Place pad on same side as surgical site

Hazards of Electrosurgery Alternate site burns Return site burns Plume Explosion and Fire Electric Shock “Other” burns Electromagnetic interference

Hazards of Electrosurgery Alternate site burns: current may bypass intended circuit and seek ground through path of less resistance (ie. EKG electrode, stirrup, towel clip, OR table, ring) Return site burns: if isolated ESU is utilized, ground seeking current is not a risk. Return burns may still occur if there is poor electrical contact between patient’s skin and return electrode. Poor contact can be caused between electrode and skin or result of poor patient prep or pad placement.

Hazards of Electrosurgery Plume: contains hazardous substances that may be inhaled by patient or surgical staff. Explosion and fire: intestinal explosions may occur as a result of ESU sparks which ignites hydrogen or methane gas in a poorly prepared colon. Fire is always a risk since the OR environment is so oxygen rich. An ESU spark in a oxygen rich environment can cause a fire.

Hazards of Electrosurgery Electric Shock: a faulty generator may deliver lower frequency current, and may result in electric shock. “Other Burns”: ◦ Insulation failure: active electrode is compromised, allowing current to escape at unintended tissue sites ◦ Direct coupling: current flows from a noninsulated metal instrument to a non-target tissue

Hazards of Electrosurgery ◦ Capacitive coupling: current passes through intact insulation to non-target tissue. • ESU power setting excessively high • Wrong type of active electrode used • Incorrect application of active electrode to the tissue • Unintentional activation of active electrode • Electromagnetic interference: can cause outdated pacemakers to increase their rate of firing, leading to fibrillation

Argon Enhanced Coagulation (AEC) Form of electrosurgery utilizing Argon gas to increase effectiveness of therapeutic current When argon is delivered to the active electrode, this creates a highly conductive stream of ionized gas to carry current to tissue. Known as “Argon Beam Coagulator”

Nursing Interventions for use of ESU Pre-Operative: √ ESU for damage, integrity of wires, plugs and connections √ Active electrode cord and hand piece √ Return electrode ESU compatibility, appropriate size, integrity, conductive gel and expiration date ─ Choose and prepare electrode site ─ Site choice. On surgical side. Close to surgical site. Over smooth skin, well perfused muscle ─ Prep site. Remove hair if necessary. Clean skin of lotion/grease with non flammable agent

Nursing Interventions for use of ESU Intraoperative: ◦ Place return electrode (grounding pad) on patient after final positioning is made ◦ Check to make sure patient is not in contact with any metal object ◦ Keep active electrode (pencil) in holster when not in use ◦ Clean active electrode frequently ◦ Keep return electrode (grounding pad) dry. Protect from pooling fluids.

Nursing Interventions for use of ESU Postoperative: Remove electrode carefully Check return electrode site for injury Check for alternate site burns Remind laparoscopic patient to report signs of internal burn injury immediately ◦ Document condition of pad site ◦ ◦

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice I: “Personnel selecting the electrosurgical unit (ESU) and accessories for purchase or use should make decisions based on safety features to minimize risks to patients and personnel. ” Recommended Practice II: “Personnel should demonstrate competence in use of ESU and accessories. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice III: “The ESU and accessories should be used according to manufacturers’ written instructions. ” Recommended Practice IV: “The ESU system should be used in a manner that minimizes the potential for injuries. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice V: “ The electrical cords and plugs of the ESU should be handled in a manner that minimizes the potential for damage and subsequent patient injuries. ” (Do Not bend cords) Recommended Practice VI: “ The active electrode should be used in a manner that minimizes the potential for injuries. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice VII: “When monopolar electrosurgery is used, a dispersive electrode should be used in a manner that minimizes the potential for injuries. ” Recommended Practice VIII: “Personnel should take special precautions when using the ESU during endoscopic procedures. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice IX: “Personnel should take special precautions when using the ESU with Patients who have pacemakers, internal cardioverter-defibrillators (ICDs) or other electrical implants. ” Recommended Practice X: “Bipolar active electrodes (including vessel occluding devices should be used according to manufacturer’s written instructions. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice XI: “Ultrasonic electrosurgical devices function differently from monopolar active electrodes and should be used according to manufacturers’ written instructions. ” Recommended Practice XII: “Argon enhanced coagulation (AEC) technology poses unique risks to patient and personnel safety and should be used according to manufacturers’ written instructions. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice XIII: “When using the AEC unit with endoscopic procedures, personnel should follow the manufacturer’s instructions and recommendations for use. ” Recommended Practice XV: “Health care facilities’ policies and procedures for electrosurgery must be in compliance with the Safe Medical Devices Act. ”

AORN Standards, Recommended, Practices and Guidelines for Electrosurgery Recommended Practice XVI: “Policies and procedures for electrosurgery should be developed, reviewed periodically, revised as necessary, and readily available in the practice setting. ”

Universal Protocol TM JCAHO Hospital’s National Patient Safety Goal Now called Universal Protocol TM To prevent wrong site, wrong procedure, wrong person surgery TM www. jointcommission. org

Universal Protocol TM Pre-operative verification process with 2 identifiers The Doctor marks the operative site (PA participating in procedure may mark site) ◦ Right/Left distinction, multiple structures, or levels “Time out” immediately before starting the procedure with all team members present.

THE VERIFICATION PROCESS AND TIME OUT INITIATIVE Preventing: Wrong Person Wrong Site Wrong Procedure/Surgery

Verification Protocol and Time Out Initiative Wrong procedure, wrong person surgery can be prevented. The Universal Protocol for preventing wrong site, wrong procedure, wrong person surgery was developed to meet this

Universal Protocol Principles Active involvement and effective communication of all members of the surgical/healthcare team is a must. The patient or legally designated representative MUST be involved in the process. Consistent implementation of a standardized approach using a universal protocol is vital. Microsoft Clipart

Procedures Defined The Universal Protocol is applicable to all operative and invasive procedures that expose patients to more than minimal risk, including procedures done Microsoft Clipart • Operating Room • Emergency Room • Special Procedures • GI Lab • Cardiac Cath Lab • Interventional Radiology • Patient Bedside

Universal Protocol Implementation 3 action steps, taken together, are required for implementation of the Universal Protocol: Pre-procedural Verification Process: (Patient Identification) Marking the Procedure Site: (Only if laterality or level is involved) Time Out with Pause: (Verification with All team present Microsoft Clipart

Preprocedural Verification Preprocedural verification begins with the physician’s discussion of therapy options with the patient or legal representative. The patient should be awake and aware, if possible. Benefits and risks of planned procedures or surgeries are discussed, including restrictions, care requirements, pain management, and expectations for recovery as well as risk of not having the procedure. Informed consent is obtained. The procedure/surgery is scheduled. Microsoft Clipart

Pre-procedural Verification At the time of admission or entry into the facility Microsoft Clipart Anytime responsibility for care is transferred to another caregiver Microsoft Clipart Before the patient leaves the preoperative area or enters the procedure/surgical room Microsoft Clipart

Time Out A “Time Out” must be conducted in the location where the procedure will be done, just before starting the procedure Microsoft Clipart All of the team members must be present at the time when the TIME OUT is called. All must concur. Microsoft Clipart

Time Out The entire procedural/operative team must participate. The phrase “Time Out” must be used to initiate the verification process. Active communication is required by the entire team. Microsoft Clipart

When in Doubt…Time Out All questions must be resolved prior to the start of any procedure or surgery. Resolution of all discrepancies must be documented in the medical record. Circulating nurse has the opportunity and responsibility to implement and promote Time Out protocol Microsoft Clipart

AORN Position Statement on Correct Site Surgery “AORN is dedicated to patient safety. Using suggested risk-prevention strategies for identification and verification of correct patient, surgical site, and procedure will reduce the risk of error. AORN endorses JCAHO’s Universal Protocol for Preventing Wrong Site, Wrong Procedure, Wrong Person Surgery…. . ”

Sentinel Event An unexpected occurrence involving death or a serious physical or psychological injury or risk. Serious injury includes loss or a limb or function. Recurrence would carry a significant chance of a serious adverse outcome. A sentinel event signals the need for immediate investigation and response.

Sentinel Event JCAHO requires that the organization submit a root cause analysis, action plan and all sentinel event information through the mail to JCAHO. Four alternatives are available to the organization after either a voluntary report of a sentinel event or response to JCAHO inquiry of a sentinel event.

Surgical Counts Refers to counting sponges, sharps (blades, needles), surgical instruments that are initially opened and all delivered to field during surgery Counts are performed by the scrub person and circulating nurse Facility policies determine what is counted based on procedure and probability of retained item

Surgical Counts Some facilities will use a count sheet or dry erase board where counts are recorded. Preliminary, first closing, and final results are documented on the Perioperative record. Perioperative nurses are responsible for count procedures to be carried out according to policy

Surgical Counts Sponges: placed in a sterile field to absorb blood and fluids. ALL are X-Ray detectable via radiopaque strip or thread ◦ “Lap Sponges”: square or rectangular in shape, used for large amounts of blood, are packaged in units of 5 ©Kimberly Clark, Used by permission

Surgical Counts Raytec/4 x 4’s/Gauze sponges: used where a small amount of blood or fluid is anticipated. These also may be folded, placed on a sponge stick for swabbing an area. Come in packages of 10. ©Kimberly Clark, Used by permission

Surgical Counts “Peanuts”: are small sponges, (the size of a peanut) that are clamped to forceps, used on delicate tissue “Kitner” dissectors: is a small roll of cotton tape that is used for dissection or absorption Tonsil sponges: cotton filled gauze in a ball shape, used to absorb blood or stop blood. Cottonoid sponges/patties/neuro sponges: made of compressed cotton, supplied in a variety of sizes, used on delicate structures on brain and spinal cord Versant Advantage, Inc. , 2006

Surgical Counts Instrument Counts: all instruments in set and any added to the field are counted (to include any parts ex. Wing nuts from retractors etc) Sharp Counts: any needle, ESU/Bovie tips, blades Microsoft clipart

Surgical Counts are performed ALOUD and TOGETHER All items must be visible so both persons can verify the count Counts are performed ◦ Before surgery begins ◦ When additional items (sponges, sharps, instruments) are added to the sterile field during the procedure Microsoft clipart

Surgical Counts During the procedure, scrub person discards the sponges off the sterile field into a “kick bucket” As five to ten sponges accumulate the circulator places them in a impervious bag Soiled sponges are only handled with an instrument or gloved hands ©Pedigo, Used by permission

Surgical Counts At the end of case, soiled sponges will be placed in sponge counter bags to view the sponges all in one place to verify the correct sponge count ALL SPONGES, SHARPS & INSTRUMENTS ARE TO REMAIN IN THE ROOM UNTIL THE PROCEDURE IS FINISHED & THE PATIENT HAS LEFT THE ROOM

Surgical Counts A second count is performed as wound closure begins. The count should be aloud and together. Counting should be consistent: start at the surgical field, Mayo stand, back table, discarded sponges in kick bucket, sponge counter bag Surgical team is informed out loud of results of count (sponge and needle count correct)

Surgical Counts Third count is started as skin closure begins Results are reported and documented If a count is incorrect: ◦ Surgical team notified ◦ Anesthesia keeps patient asleep ◦ Thorough search of wound, room (including trash, under OR table) for missing item ◦ OR Supervisor is notified ◦ If not found, an X-ray is ordered ◦ Complete an incident report

Surgical Counts Facts If the surgical wound is packed and left in, a x-ray detectible sponge shall NOT be used If a needle or blade breaks, it should be isolated, placed in a designated area from the sterile field and accounted for in entirety Following surgical procedure, dispose of sharps in a OSHA Biohazard sharp box

Surgical Counts Facts Instrument counts are done TWICE not three times as sponges and sharps Instrument sets are standardized and numbers are kept to minimum and match the count sheet for that set Minimally invasive surgery cases require instrument counts for standard instruments incase the procedure becomes open

Surgical Counts Documentation What was counted ◦ Sponges ◦ Sharps ◦ Instruments Results of count Surgeons notified of results Names, titles, signatures of persons performing the count Any retained item Action taken if count is incorrect Reason if count was omitted (emergency) Action taken if count was omitted (x-ray) Microsoft clipart

AORN Recommended Practices for Sponge, Sharp and Instrument Counts Recommended Practice I: “Sponges should be counted on all procedures in which the possibility exists that a sponge could be retained. ” Recommended Practice II: “Sharps and other miscellaneous items should be counted on all procedures. ” ©Kimberly Clark, used by permission

AORN Recommended Practices for Sponge, Sharp and Instrument Counts Recommended Practice III: “Instruments should be counted for all procedures in which the likelihood exists that an instrument could be retained. ” Recommended Practice IV: “Additional measures for investigation, reconciliation, documentation, and prevention of retained surgical items should be taken. ” Microsoft Clipart

AORN Recommended Practices for Sponge, Sharp and Instrument Counts Recommended Practice V: “Sponge, sharp and instrument counts should be documented on the patient’s intra-operative record by the RN circulator. ” Recommended Practice VI: “Policies and procedures for sponges, sharps and instrument counts should be developed, reviewed periodically, revised as necessary and readily available in the practice setting. ”

Sharps Safety Hands free transfer of sharps should occur through creation of a “neutral zone” After use, surgeon places the contaminated sharp in a “neutral zone” in the field. It is there that the scrub person will retrieve it Microsoft clipart

Hypothermia in the Perioperative Setting Causes of Hypothermia: ◦ Anesthetic agents dilate blood vessels ◦ Inhibits temperature regulating mechanism in hypothalamus ◦ Exposure to cold irrigating solutions ◦ Lying on cold OR beds ◦ Evaporative heat loss from open body cavities ◦ Reduction in metabolic heat production ◦ Room temperature IV fluids

Hypothermia in the Perioperative Setting Temperature monitored during surgery via: ◦ External patch thermometer ◦ Internal esophageal ◦ Rectal probe
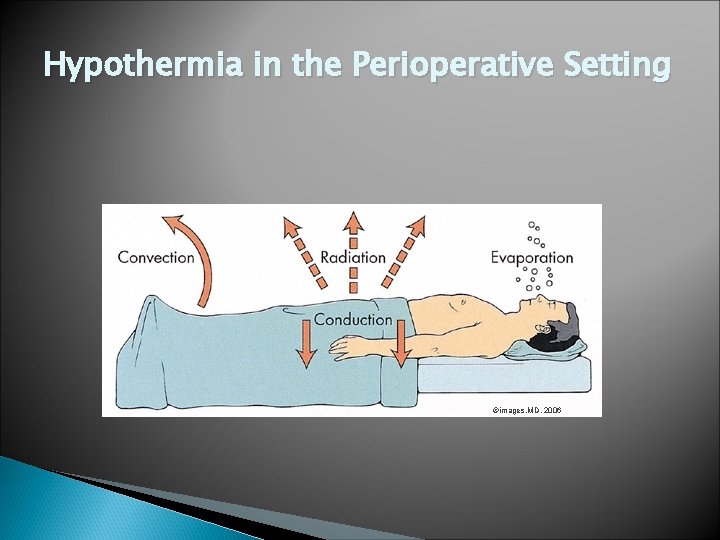
Hypothermia in the Perioperative Setting ©images. MD, 2006

Hypothermia in the Perioperative Setting Studies have shown that warm blankets have shown little effect on patient’s core temperature Studies have shown that forced air preoperatively, throughout the procedure maintain higher temperatures Studies have also shown that mild hypothermia triple infection rate and prolong hospital stays

Hypothermia in the Perioperative Setting Nursing Interventions to prevent hypothermia: ◦ ◦ ◦ Warm prep solutions Forced air warming pre, during and post-op Warm irrigation fluids Warmed IV fluids Head coverings

JCAHO 2016 NATIONAL PATIENT SAFETY GOALS Improve the accuracy of patient identification (Two identifiers) Improve effectiveness of communication among caregivers Improve the safety of using medications Reduce the risk of health care associated infections Prevent mistakes in surgery (Time Out)

JCAHO 2016 NATIONAL PATIENT SAFETY GOALS Accurately and completely reconcile medications across the continuum of care Reduce the risk of patient harm resulting from falls Reduce the risk of surgical fires Eliminate wrong-site, wrong patient, wrong -procedure surgery (Mark site) Improve effectiveness of clinical alarm systems (Alarms must be audible)

Radiation Safety in the OR Fluoroscopy primary source of potential exposure for OR staff All personnel should remain at least 6 feet away from the X-ray tube ©images. MD, 2006

Radiation Safety in the OR Amount of radiation received dependent on the following: ◦ Amount of exposure time ◦ If radiation is emitted at a constant rate

Radiation Safety in the OR Protection consists of: ◦ ◦ ◦ Lead aprons Thyroid shields Leaded gloves Leaded eye wear Portable lead screens

Radiation Safety in the OR Lead aprons: ◦ ◦ Checked regularly by x-ray (usually yearly) Check prior to wearing for visible cracks, punctures Aprons should be hung on a mobile or fixed rack Never fold an apron

Radiation Safety in the OR Radiation Safety Officer assists perioperative personnel in complying with safety regulations ◦ Using shielding, time and distance to limit exposure ◦ Posting radiation warning signs on doors to OR ◦ Documenting all protective measures on perioperative record

AORN Recommended Practices for Reducing Radiological Exposure in the Practice Setting Recommended Practice I: “The patient should be exposed to radiation only if medically indicated. ” Recommended Practice II: “During medically indicated radiological procedure, the patient should be protected from unnecessary radiation exposure. ”

AORN Recommended Practices for Reducing Radiological Exposure in the Practice Setting Recommended Practice III: “Occupational exposure to radiation should be minimized in the practice setting. ” Recommended Practice IV: Shielding devices should be handled carefully and examined periodically to prevent and detect damage that could diminish their effectiveness. ”

AORN Recommended Practices for Reducing Radiological Exposure in the Practice Setting Recommended Practice V: “Individuals should be protected from exposure to patients who have received diagnostic or therapeutic radionuclides that may pose a radiation risk. ” Recommended Practice VI: “Radiation monitoring devices should be worn by personnel who are in frequent proximity to radiation, as determined by the RSO. ”

AORN Recommended Practices for Reducing Radiological Exposure in the Practice Setting Recommended Practice VII: “Appropriate handling of all therapeutic radiation sources in the practice setting should minimize exposure. ” Recommended Practice VIII: “Measures taken to protect patients from direct and indirect radiation exposure should be documented on the perioperative nursing record. ”

Chemical Hazards Methyl Methacrylate (bone cement) Formaldehyde Peracidic acid (Steris sterilant) Waste Anesthetic Gases Latex Considerations

Methyl Methacrylate (Bone Cement) Acrylic cement type compound that polymerizes into a strong plastic Used in orthopedic prosthesis and neurosurgical reconstruction of cranial bone Supplied in separate containers (liquid and powder). Liquid is flammable. Always mix under a suction evacuation bowel or exhaust hood. Liquid should always be poured into powder.

Methyl Methacrylate (Bone Cement) Precautions: ◦ Double gloving is recommended when mixing ◦ Contact lenses should not be worn when working with bone cement (vapors can cause damage to lens of eye) ◦ Can cause skin rashes, itching, watering eyes, headache and respiratory irritation ◦ Electronic equipment should not be used until bone cement is set and supplies have been discarded

Formaldehyde Used in the operating room to preserve surgical specimens Can be combined with water and methanol to make formalin Affects nose and upper respiratory tract Strong pungent odor- can cause respiratory irritation and watery eyes

Formaldehyde Precautions to take: ◦ ◦ Wear gloves Ensure there is adequate ventilation Should NOT be taken into OR Suite Label container

Peracidic Acid Sterilant for Steris 1 E is very irritating When disposing of chemical, have good ventilation Use PPE, gown, gloves, mask, goggles Flush down sink with copious amounts of running water

Waste Anesthetic Gases: ◦ Nitrous Oxide ◦ Halothane ◦ Forane ◦ Ultane ◦ Suprane ◦ Ethrane Exposure to waste gases may lead to neurologic problems or blood abnormalities

Waste Anesthetic Gases Recommended permissible exposure level for nitrous is 25 ppm Personal dosimeters are available to measure level in OR Each OR is required to have a Scavenger system in place so waste gases will not escape into the OR Routine leak testing should be performed to make sure the scavenger system is functioning properly

MSDS (Material Safety Data Sheet) Every employee must be informed of potential hazards of chemicals in the work place MSDS (Material Safety Data Sheet) should be readily available Manufacturers of chemical must provide an MSDS about the product Staff should know the location of MSDS and first-aid measure with chemical hazards worked with

MSDS (Material Safety Data Sheet) All employees should wear PPE (personal protective equipment) when working with chemical hazards Label instructions on chemical container should be followed

Care and Handling of Surgical Specimens Proper handling and care is of utmost importance for a SAFE patient outcome Responsibility of circulating nurse to: ◦ ◦ ◦ Identify Document Proper care Labeling Timely delivery to lab

Care and Handling of Surgical Specimens Examples of specimens: ◦ ◦ ◦ Blood Tissue Bone Body fluids Foreign bodies

Care and Handling of Surgical Specimens Labeling: ◦ Must ensure patient name and origin of specimen ◦ Date, time, and initials of person preparing specimen go on label ◦ Provide descriptive information (from surgeon) (ie. Suture tag on breast biopsy is at 9 o’clock) ◦ All specimens must be documented on Perioperative record ◦ Specimen will be entered in the specimen log and placed in the pick up bin

Care and Handling of Surgical Specimens All tissue should be kept moist and sent to the lab ASAP ◦ Fresh frozen specimen should be sent to pathology immediately with NO formalin on it ◦ If specimen is a “permanent” then formalin is placed on it, labeled and will NOT be taken to the laboratory immediately

Care and Handling of Surgical Specimens Frozen section: ◦ When immediate tissue identification or identification of a malignancy under a microscope in the lab is needed. ◦ Place on a moist towel, telfa or in a dry specimen container and sent to lab immediately ◦ Always wear gloves when handling specimens ◦ Place a patient label with the specimen ◦ Complete the pathology specimen form and send with the pathologist ◦ Send at least 4 extra patient labels

AORN Recommended Practices for the Care and Handling of Specimens in the Perioperative Environment Recommended Practice I: “Assessment of specimen collection and special handling needs should begin when the procedure is scheduled. ” Recommended Practice II: “Procedures for correct patient and specimen identification should be implemented, including confirmation of consistent and accurate patient information on labels and forms. ”

AORN Recommended Practices for the Care and Handling of Specimens in the Perioperative Environment Recommended Practice III: “Collection and handling of a specimen should be completed in a manner that protects and secures the specimen and prevents contamination of personnel handling the specimen. ” Recommended Practice IV: “specimen containers should be labeled to communicate patient, specimen, preservative and biohazard information. ”

AORN Recommended Practices for the Care and Handling of Specimens in the Perioperative Environment Recommended Practice V: “Tissue specimens should be designated for routine pathological exam, gross identification only, or disposal according to health care organizational policies. ” Recommended Practice VI: “Health care organizations should establish communication and documentation methods regarding specimen accountability and accurate documentation. ”

AORN Recommended Practices for the Care and Handling of Specimens in the Perioperative Environment Recommended Practice VII: “Explanted defective medical devices must be reported to the manufacturer. ” Recommended Practice VIII: “Transfer and transport methods and process should be established by the health care organization according to the specimen type and diagnostic requirements. ”

AORN Recommended Practices for the Care and Handling of Specimens in the Perioperative Environment Recommended Practice IX: “Forensic specimens should be handled in a manner that preserves the condition of the evidence and verifies that the evidence has been in secure possession at all times. ” Evidence such as bullets should be placed in a envelope or bag. Contents should be given to the Responsible law enforcement officer. Microsoft Clipart

AORN Recommended Practices for the Care and Handling of Specimens in the Perioperative Environment Recommended Practice X: “Each healthcare organization should develop written procedures for handling radioactive pathology specimens. These procedures should include, but are not limited to, specimen handling, labeling, transportation, storage, and disposal. The procedures should keep radiation exposure to hospital workers as low as reasonably achievable. ”
- Slides: 119