Periodontal regeneration When the periodontium is damaged by

ﻋﺬﺭﺍﺀ ﻋﻠﻲ ﺍﻟﻌﺎﻣﺮﻱ. ﺩ Periodontal regeneration When the periodontium is damaged by inflammation or as a result of surgical treatment, the defect heals either through periodontal regeneration or repair. In periodontal regeneration, healing occurs through the reconstitution of a new periodontium, which involves the formation of alveolar bone, functionally aligned PDL, and new cementum. Alternatively, repair due to healing by replacement with epithelial and/or connective tissue that matures into various nonfunctional types of scar tissue, is termed new attachment. Histologically, patterns of repair include long junctional epithelium, Connective tissue adhesion and root resorption, Root resorption and ankylosis, and/or new attachment. Traditional therapeutic modalities usually failed to predictably regenerate the periodontal tissue lost due to disease process. Regeneration: Is defined as a Reproduction or Reconstruction of a lost or injured part in such a way that the architecture and function are completely restored. New attachment: Is the embedding of new PDL fibers into new cementum and the attachment of the gingival epithelium to a tooth surface previously denuded by disease. Reattachment: Reunion of surrounding soft tissue and a root surface with preserved PDL tissues. Assessment of Periodontal Wound Healing The outcomes of periodontal regeneration are most frequently evaluated by: Histological methods It is only through histological analysis can one define the nature of the reparative tissue. Clinical changes in: • Attachment levels (CAL) • Bone levels (BL) • Probing pocket depths (PPD)
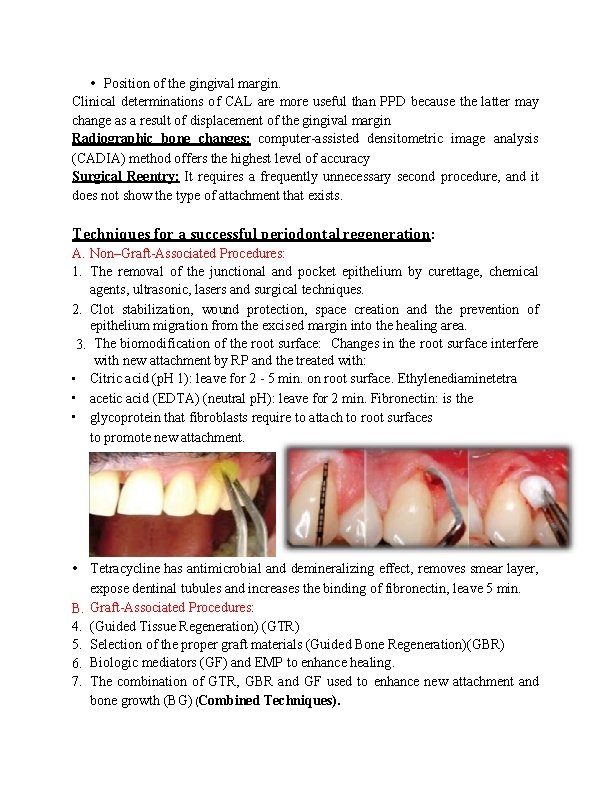
• Position of the gingival margin. Clinical determinations of CAL are more useful than PPD because the latter may change as a result of displacement of the gingival margin Radiographic bone changes: computer-assisted densitometric image analysis (CADIA) method offers the highest level of accuracy Surgical Reentry: It requires a frequently unnecessary second procedure, and it does not show the type of attachment that exists. Techniques for a successful periodontal regeneration: A. Non–Graft-Associated Procedures: 1. The removal of the junctional and pocket epithelium by curettage, chemical agents, ultrasonic, lasers and surgical techniques. 2. Clot stabilization, wound protection, space creation and the prevention of epithelium migration from the excised margin into the healing area. 3. The biomodification of the root surface: Changes in the root surface interfere with new attachment by RP and the treated with: • Citric acid (p. H 1): leave for 2 - 5 min. on root surface. Ethylenediaminetetra • acetic acid (EDTA) (neutral p. H): leave for 2 min. Fibronectin: is the • glycoprotein that fibroblasts require to attach to root surfaces to promote new attachment. • Tetracycline has antimicrobial and demineralizing effect, removes smear layer, expose dentinal tubules and increases the binding of fibronectin, leave 5 min. B. 4. 5. 6. 7. Graft-Associated Procedures: (Guided Tissue Regeneration) (GTR) Selection of the proper graft materials (Guided Bone Regeneration)(GBR) Biologic mediators (GF) and EMP to enhance healing. The combination of GTR, GBR and GF used to enhance new attachment and bone growth (BG) (Combined Techniques).

The theory of Guided Tissue Regeneration (GTR) it is a procedure through which the exclusion of epithelial and gingival connective tissue cells from the healing area by the use of physical barrier may allow or guide PDL cells to repopulate the detached root surface. Ideal properties: ✓ It should be biocompatible & allow tissue regeneration. ✓ It should be nontoxic and non-cariogenic. ✓ It should be chemically inert. ✓ It should be able of being sterilized. ✓ It should be easy to handle during surgery. ✓ It should be sufficiently rigid to maintain a space between it & the root surface. ✓ It should be supplied in different design to suit the specific clinic situation. ✓ It should be easily stored & should have a long shelf life. ✓ It should be easily retrievable in case of complication. ✓ It should be cost effective. The clinical indication for the use of GTR are: 1. Class II furcation defects 2. Two or three walled vertical Interproximal, and circumferential intrabony periodontal defects. 3. Class III furcation may be treated with GTR but with less predictability of success. 4. Ridge augmentation 5. Treatment of gingival recession.
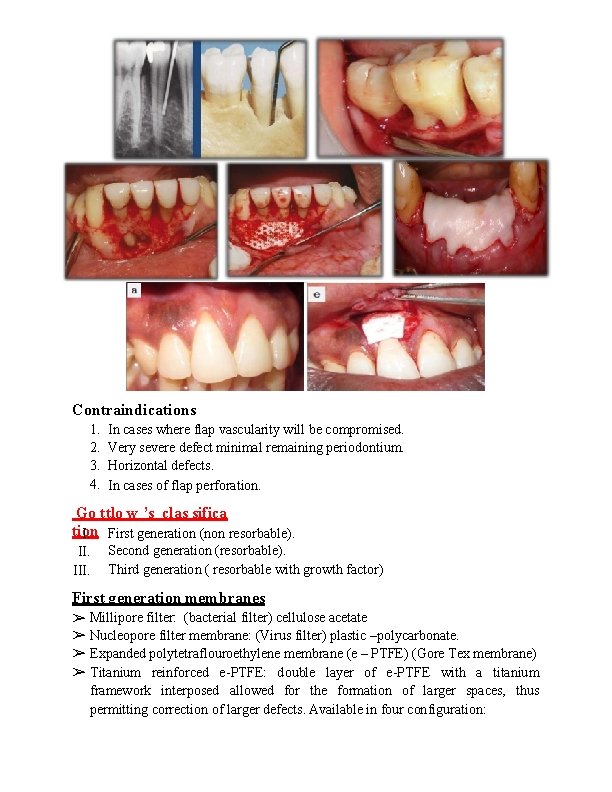
Contraindications 1. 2. 3. 4. In cases where flap vascularity will be compromised. Very severe defect minimal remaining periodontium. Horizontal defects. In cases of flap perforation. Go ttlo w ’s clas sifica tion I. First generation (non resorbable). III. Second generation (resorbable). Third generation ( resorbable with growth factor) First generation membranes ➢ Millipore filter: (bacterial filter) cellulose acetate ➢ Nucleopore filter membrane: (Virus filter) plastic –polycarbonate. ➢ Expanded polytetraflouroethylene membrane (e – PTFE) (Gore Tex membrane) ➢ Titanium reinforced e-PTFE: double layer of e-PTFE with a titanium framework interposed allowed for the formation of larger spaces, thus permitting correction of larger defects. Available in four configuration:

1. Interproximal 2. Wrap around 3. Single tooth narrow 4. Single tooth wide ➢ Rubber dam: synthetic fiber (nylon) Second generation membrane : i. e. Cargile membrane. ➢ Collagen ➢ Polyglycoside synthetic polymers: i. e. (Polylactic acid membrane) (Atrisorb gel) (guidor) and Vicryl mesh (Polyglactin 910). ➢ Calcium sulfate. Collagen membrane Collagen has been used in medicine and dentistry because of its Biocompatibility and Improvement of healing. Collagen has many auspicious biologic activities: ✓ low immunogenicity ✓ Hemostatic ✓ Attracts and activate PDL cells & gingival fibroblast ✓ Reduced water absorption, ✓ Decreased solubility and increased tensile strength. When a collagen membrane is implanted in the human body it is resorbed by the enzymatic activity of macrophages & polymorph nuclear leucocytes. Several complications were reported following the use of collagen: • Early degradation. • Epithelial down growth along the material. Disadvantages of non-resorbable membranes: 1. Second Surgery Required. 2. Exposure to oral environment. 3. Bacterial contamination. Disadvantages of resorbable membranes: 1. Risk of exposure. 2. Collapse into the defect area- (bone filler is needed). 3. Technique is sensitive & technically demanding. 4. Harmful degradation products of synthetic membranes.
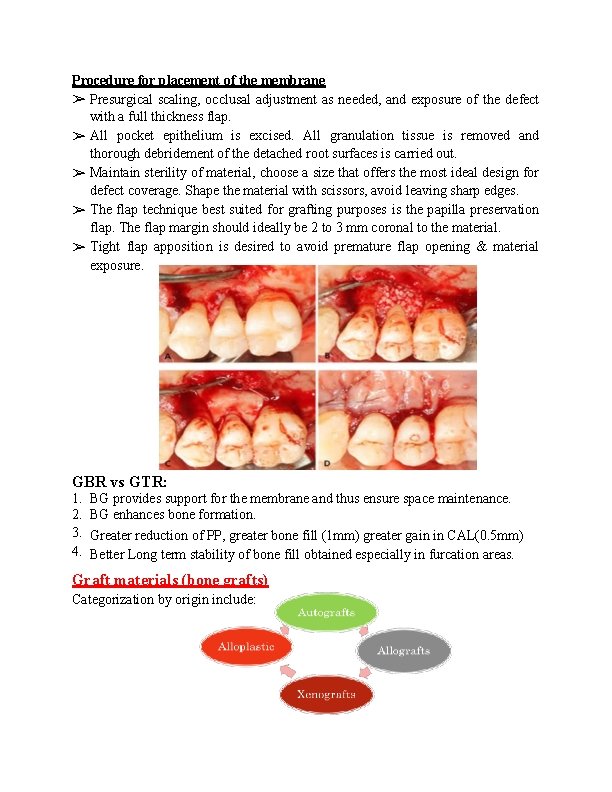
Procedure for placement of the membrane ➢ Presurgical scaling, occlusal adjustment as needed, and exposure of the defect with a full thickness flap. ➢ All pocket epithelium is excised. All granulation tissue is removed and thorough debridement of the detached root surfaces is carried out. ➢ Maintain sterility of material, choose a size that offers the most ideal design for defect coverage. Shape the material with scissors, avoid leaving sharp edges. ➢ The flap technique best suited for grafting purposes is the papilla preservation flap. The flap margin should ideally be 2 to 3 mm coronal to the material. ➢ Tight flap apposition is desired to avoid premature flap opening & material exposure. GBR vs GTR: 1. 2. 3. 4. BG provides support for the membrane and thus ensure space maintenance. BG enhances bone formation. Greater reduction of PP, greater bone fill (1 mm) greater gain in CAL(0. 5 mm) Better Long term stability of bone fill obtained especially in furcation areas. Graft materials (bone grafts) Categorization by origin include:

They are also evaluated based on biological mechanism into: 1. Osteogenesis: is the formation of new bone by cells in the graft. 2. Osteoinduction: is a chemical process by which molecules contained in the graft (e. g. , BMPs) convert the neighboring cells into osteoblasts, which form bone. 3. Osteoconduction: is a physical effect by which the matrix of the graft forms a scaffold that favors outside cells to penetrate the graft and form new bone. Characteristics of graft materials biologic acceptability clinical feasibility minimal operative hazards minimal postoperative complications patient acceptance Autogenous bone grafts: Bone obtained from the same individual A. From intraoral sites : 1. extraction socket 2. edentulous ridges 3. bone from the jaw without damaging the roots 4. Max. tuberosity and the ramus 5. Bone removed during osteoplasty and ostectomy. B. From extraoral sites: e. g. , (iliac). Types of intraoral Autogenous bone grafts are: cortical bone chips (large particles), osseous coagulum(trituration of bone mixed with blood, small particles), bone blend (in capsule), intraoral cancellous bone marrow, bone swaging. Allografts: From a different individual of the same species. From cortical bone of the donor, defatted, cut in pieces, washed in alcohol, and deep-frozen. Then it may be demineralized, and subsequently ground and sieved to a particle size of 250 to 750 μm and freeze-dried. Finally, it is vacuum-sealed in glass vials. 1. Freeze-dried bone allografts (FDBA): is a mineralized BG, which through the manufacturing process loses cell viability (osteoconduction). 2. Demineralized freeze-dried bone allograft (DFDBA): Demineralization in cold, diluted hydrochloric acid exposes the components of bone matrix, which are closely associated with collagen fibrils (BMPs). This enhances its osteogenic.

Xenografts: are bone from a different species. Osteoconductive • Bovine bone: crystal size of approximately 10 mm in the form of cortical granules. e. g. Bio-Oss (Osteohealth) Bio-Oss Collagen • Natural coral: consisting of exoskeleton of corals, it turns into non-resorbable porous hydroxyapatite or to a resorbable Coralline calcium carbonate. • Porcine bone. Alloplastic materials: They are synthetic, inorganic, biocompatible and/or bioactive BG (osteoconduction). 1. Hydroxyapatite (HA): new bone was only observed around particles in close proximity to host bone. Non-resorbable: polycrystalline ceramic and coralline porous HA. Resorbable: non-ceramic, Nanocrystalline hydroxyapatite. 2. β-tricalcium phosphate (β-TCP): can be bound to collagen carriers or mixed with fibrin. The concept is that collagen and fibrin form a network on which minerals can crystallize 3. Polymers: non-resorbable: calcium hydroxide coated co-polymer polymethyl-methacrylate (PMMA) and poly-hydroxyl-ethyl-methacrylate (PHEMA) referred to as HTR (hard tissue replacement). Resorbable: polylactic acid (PLA) polymer. 4. Bioactive glasses (bioglasses): Si. O 2, Na 2 O, P 2 O 5 (osteoconductive) Resorbable: (e. g. Perio. Glass, Bio. Gran®) Non-Resorbable: depending on the relative proportion of these components. • When they are exposed to tissue fluids (blood), a double layer of silica gel and calcium phosphate is formed on their surface. Through this layer the material promotes absorption and concentration of proteins used by osteoblasts to form extracellular bone matrix which promote bone formation. Third generation membranes Tissue Engineering with Biologic Mediators Growth or biological factors and cells mediators: are polypeptide hormones regulate cellular activities such as (Chemotaxis, migration, proliferation,

differentiation and matrix synthesis). They bind to high affinity to cell membrane receptors and activate cellular mechanism. Early clinical examples involving tissue engineering principles include the use of bone allografts and autologous plateletrich plasma (PRP) (Combined Techniques). Investigations indicated that the success rates with these materials were inconsistent. With the development of recombinant growth factors and morphogens, and the use of synthetic scaffolds, the level of success has improved. Once considered experimental in nature, tissue engineering is now clinically applicable with two commercially available tissue engineering systems for periodontal regeneration which involve the use of: A. Enamel Matrix Derivative (EMD) B. platelet-derived growth factor (PDGF) • Fibroblast growth factor (FGF) • Insulin like growth factor (IGF) • Transforming growth factor-β (TGF-β): including (morphogenetic bone protein). A. EMD- Enamel Matrix Derivative Major (>95%) component of EMD is amelogenins. Extracellular Matrix Proteins purified acid extract of proteins from pig enamel matrix Mechanism of action of EMD is to initiate periodontal regeneration through recruitment of cementoblasts to the root-surface and hence to stimulate these to form root-cementum. This will secondarily lead to regeneration of periodontal fibers and alveolar bone through stimulation of local growth factor secretion. • Reduces surgical time • Creates a custom-fitted barrier to promote bone and soft-tissue regeneration • Reduces bacterial contamination.

B. Platelet Derived Growth Factor PDGFs: are secreted primarily from the platelet a-granules during clotting, but also from macrophages, fibroblasts, myocytes, endothelial and bone marrow hematopoietic cells. The effects of PDGF: 1. It stimulates cellular activities of osteoblasts and PDL cells. 2. It enhances collagen type I and osteopontin production in both PDL cells and osteoblasts. 3. Gingival wound healing. • Insulin like Growth factor IGF is found in platelets. It is released with other GF present in platelets during clotting. IGF-I is produced by osteoblasts. The effects of IGF: 1. Proliferation and differentiation of osteoblasts with increase osteogenesis. 2. Increases type I collagen formation, bone matrix apposition rate, and inhibits bone collagen degradation due to the blocking of collagenase activity by osteoblasts. 3. Protecting cells from programmed cell death. • Fibroblast Growth factor Topical application of FGF-2 into intraosseous defects in alveolar bones induces significant periodontal regeneration. Histological observation revealed new cementum with Sharpey’s fibers, new functionally oriented PLF, and new AB. This suggests that FGF-2 may be efficacious in regeneration of periodontal tissue that has been destroyed by periodontitis. • Transforming Growth Factor This is stored in the α-granules of the platelets. TGF-β has chemotactic for fibroblasts and has effect on matrix synthesis, increased collagen production and fibronectin and decreased matrix degrading enzymes production. Bone Morphogenetic Proteins Group of regulatory glycoproteins that are members of the TGF-B Superfamily. That function as differentiation factors. These proteins induce cellular differentiation of stem cells into chondroblastic and osteogenic cells. BMP-2 (OP-2) BMP-3 (Osteogenin) BMP-7 (OP-1).
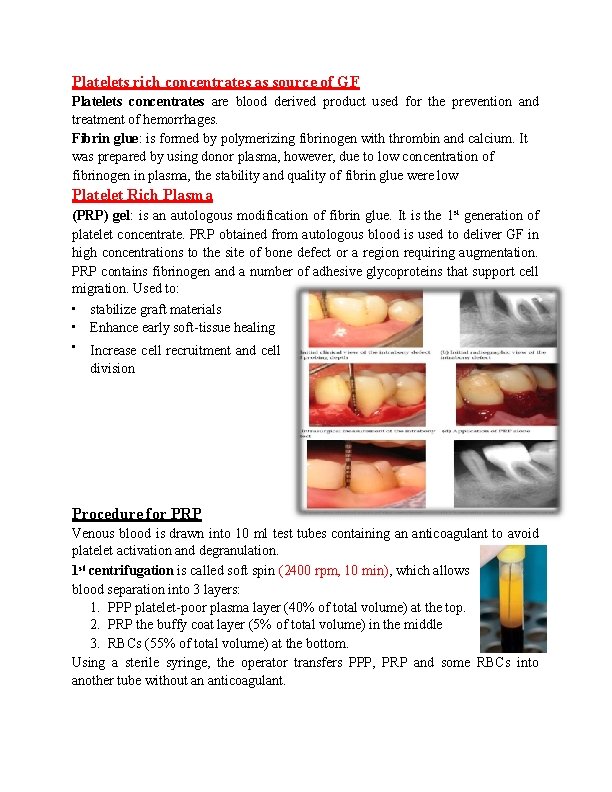
Platelets rich concentrates as source of GF Platelets concentrates are blood derived product used for the prevention and treatment of hemorrhages. Fibrin glue: is formed by polymerizing fibrinogen with thrombin and calcium. It was prepared by using donor plasma, however, due to low concentration of fibrinogen in plasma, the stability and quality of fibrin glue were low Platelet Rich Plasma (PRP) gel: is an autologous modification of fibrin glue. It is the 1 st generation of platelet concentrate. PRP obtained from autologous blood is used to deliver GF in high concentrations to the site of bone defect or a region requiring augmentation. PRP contains fibrinogen and a number of adhesive glycoproteins that support cell migration. Used to: • stabilize graft materials • Enhance early soft-tissue healing • Increase cell recruitment and cell division Procedure for PRP Venous blood is drawn into 10 ml test tubes containing an anticoagulant to avoid platelet activation and degranulation. 1 st centrifugation is called soft spin (2400 rpm, 10 min), which allows blood separation into 3 layers: 1. PPP platelet-poor plasma layer (40% of total volume) at the top. 2. PRP the buffy coat layer (5% of total volume) in the middle 3. RBCs (55% of total volume) at the bottom. Using a sterile syringe, the operator transfers PPP, PRP and some RBCs into another tube without an anticoagulant.

2 nd centrifugation, which is longer and faster than the 1 st, ‘hard spin’ (3600 rpm, 15 min). This allows (PRP) to settle at the bottom of the tube with a very few RBCs, which explains the red tinge of the final PRP preparation. The acellular plasma, PPP (80% of the volume), is found at the top. Most of the PPP is removed with a syringe, and the remaining PRP is shaken well. PRP mixed with bovine thrombin (activating the fibrinogen, which is converted to fibrin) and calcium chloride (nullifies the effect of citrate anticoagulant) at the time of application. Platelet rich fibrin This is the 2 nd generation of platelet concentrate. PRF has many advantages over PRP: 1. It eliminates the addition of anticoagulant 2. It eliminates the addition of bovine thrombin in PRP. 3. Blood is centrifuged by a tabletop centrifuge (REMY® Laboratories): i. e. (2700 rpm/ 12 min) for L-PRF. 4. Doesn't dissolve quickly after application. (strong fibrin matrix) 5. Affinity of osteoblasts to the PRF membrane to be superior compared. Procedure for A-PRF The blood is drawn into 10 ml test tubes without anticoagulant and centrifuged immediately (1300 rpm/8 min). The resultant product consists of 3 layers: 1. Top most layer consisting of acellular PPP 2. PRF clot in the middle 3. RBCs at the bottom • By the PRF box can be convert PRF clot to PRF membrane that can be used as GTR membrane. Procedure for i-PRF The blood is drawn into 10 ml tubes without an anticoagulant and centrifuged immediately (700 rpm / 3 min). The resultant product consists of: 1. Top most layer consisting of i-PRF 2. RBCs at the bottom i-PRF remains liquid for about 10 -12 minutes, then it will clot.
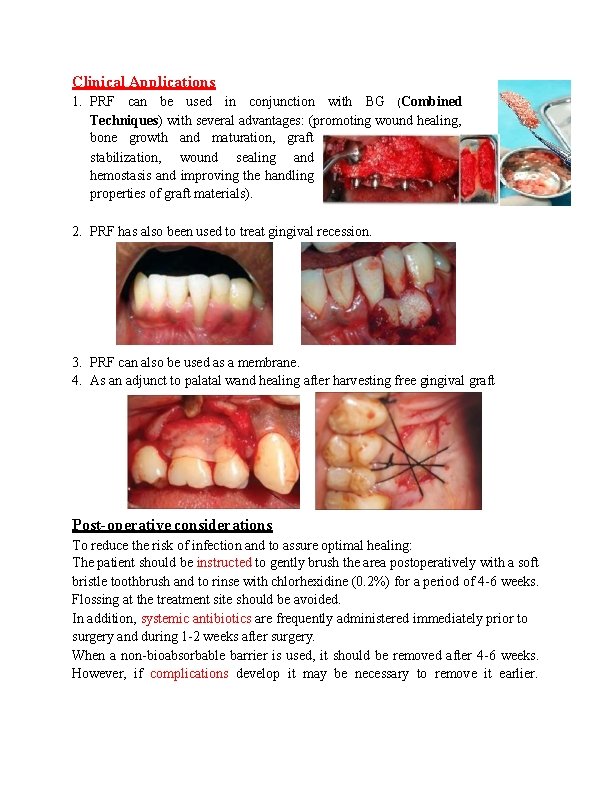
Clinical Applications 1. PRF can be used in conjunction with BG (Combined Techniques) with several advantages: (promoting wound healing, bone growth and maturation, graft stabilization, wound sealing and hemostasis and improving the handling properties of graft materials). 2. PRF has also been used to treat gingival recession. 3. PRF can also be used as a membrane. 4. As an adjunct to palatal wand healing after harvesting free gingival graft Post-operative considerations To reduce the risk of infection and to assure optimal healing: The patient should be instructed to gently brush the area postoperatively with a soft bristle toothbrush and to rinse with chlorhexidine (0. 2%) for a period of 4 -6 weeks. Flossing at the treatment site should be avoided. In addition, systemic antibiotics are frequently administered immediately prior to surgery and during 1 -2 weeks after surgery. When a non-bioabsorbable barrier is used, it should be removed after 4 -6 weeks. However, if complications develop it may be necessary to remove it earlier.
- Slides: 13