Periodicity Periodic Table Trends Periodic Trends patterns in

Periodicity Periodic Table Trends

Periodic Trends patterns in e- config = patterns in properties= Periodic Trends.

3 factors that effect Periodic Trends

1. Energy level (front of first flap) (on back of flap) Rings/orbits/ shells: “how far out the e- cloud spans” More E levels= valence e- further away from nucleus

1. Energy level (draw under flap)

(on back of sheet) Who has more Energy Levels? 1. Ca or Ge 2. Al or Ba 3. Fr or Cl

(2 nd flap front) 2. Nuclear Charge (# protons) (back) How strong is the nucleus. The more p+ the greater the charge, the stronger the pull on the e- , which pulls them in closer & makes the atom smaller!! Ie. More p+ = greater charge= e-

(under) 2. Nuclear Charge (# protons)

(behind) Nuclear Charge- Who has more? 1. Na or Rb 2. Ca or Se 3. S or Mg 4. Li or O
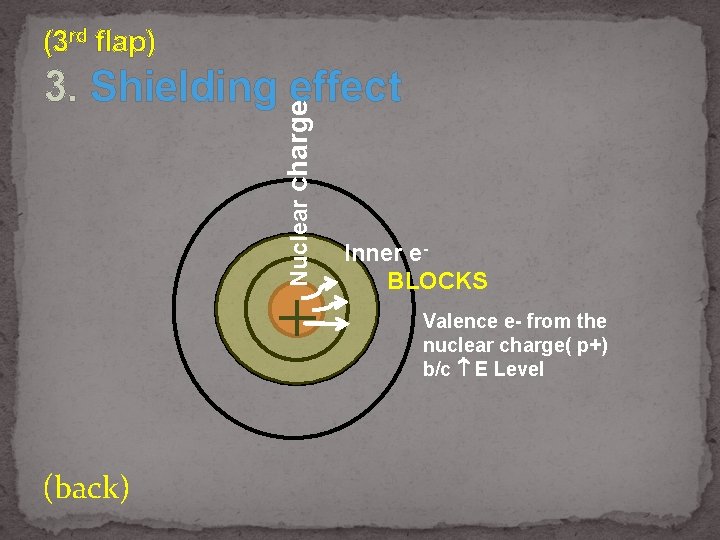
(3 rd flap) Nuclear charge 3. Shielding effect + (back) Inner e. BLOCKS Valence e- from the nuclear charge( p+) b/c E Level

(under) 3. Shielding effect

(back) Who has more shielding effect? 1. Na or Rb 2. Sn or C 3. Br or F
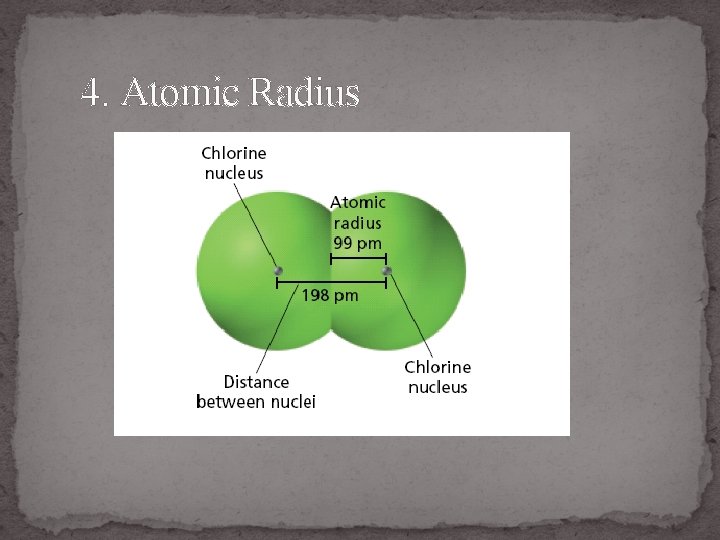
4. Atomic Radius
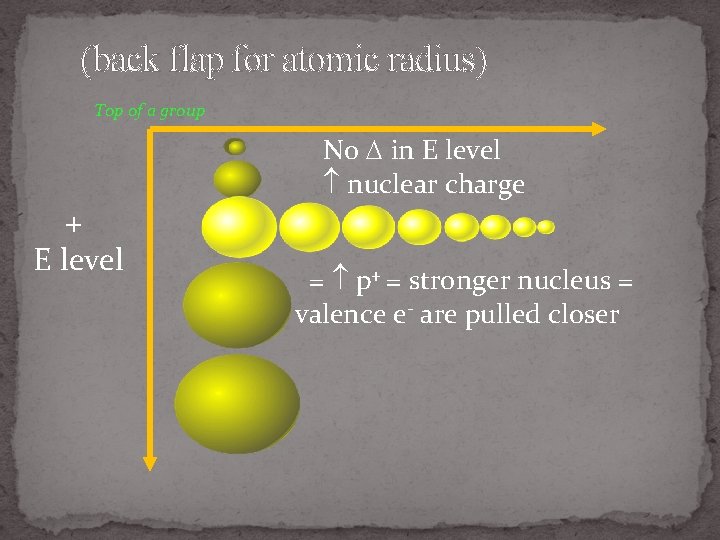
(back flap for atomic radius) Top of a group No in E level nuclear charge + E level = p+ = stronger nucleus = valence e- are pulled closer

Atomic Radius ( under flap) Fatty Francium!!!

Atomic Radius

Which atom is bigger & Why? 1. Li or Cs 2. Ga or B 3. O or C 4. Be or Ba 5. Si or S

(front) 5. Ionization Energy (IE)

(back) Ionization Energy- energy needed to remove higher IE Top of a group 1 e. Bigger = valence e- farther away= less E (easier) to remove e- Closer to being noble gas = more E (harder) to remove e- lower IE
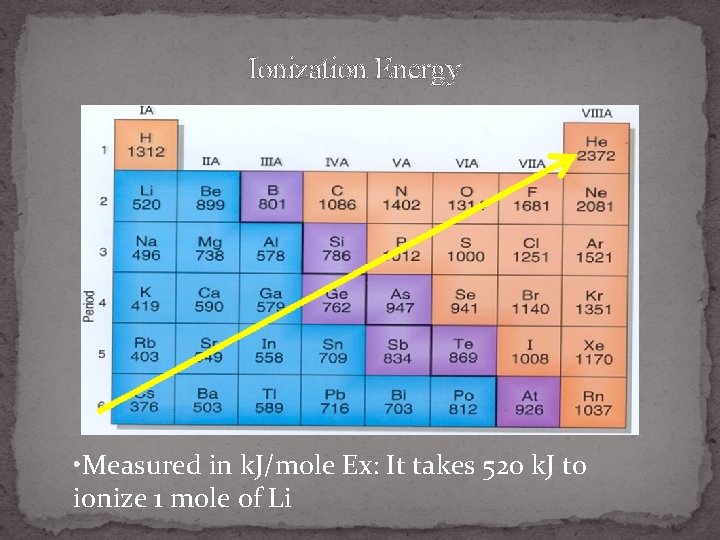
Ionization Energy • Measured in k. J/mole Ex: It takes 520 k. J to ionize 1 mole of Li
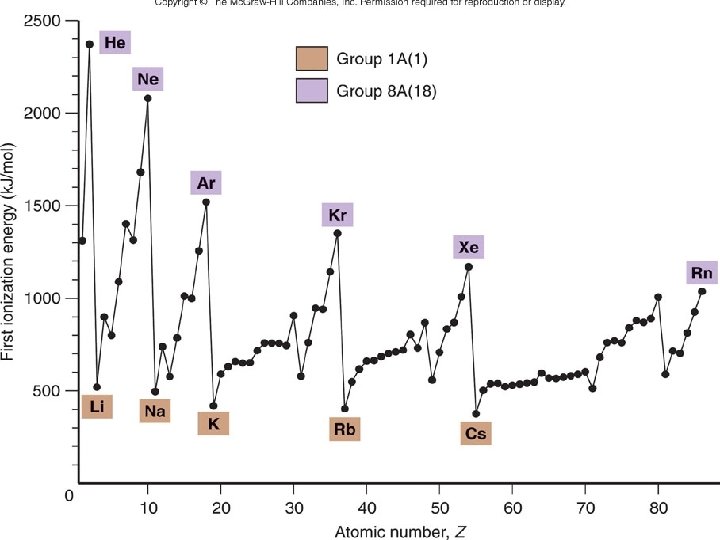

Which atom has larger (1 st ) Ionization Energy ? 1. Na or S 2. Mg or Ba 3. Ga or Br 4. P or Bi

2 nd , 3 rd …. Ionization Energies The 1 st electron is the easiest to remove It take more energy to remove a 2 nd electron than it did the 1 st and so forth

(front) 6. Electronegativity (back) Ability to attract e- into a chemical bond Like tug of war
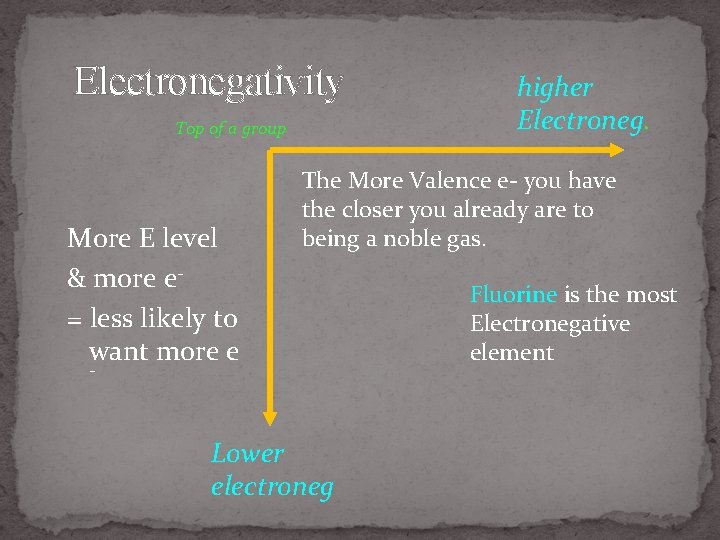
Electronegativity Top of a group More E level & more e= less likely to want more e higher Electroneg. The More Valence e- you have the closer you already are to being a noble gas. - Lower electroneg Fluorine is the most Electronegative element
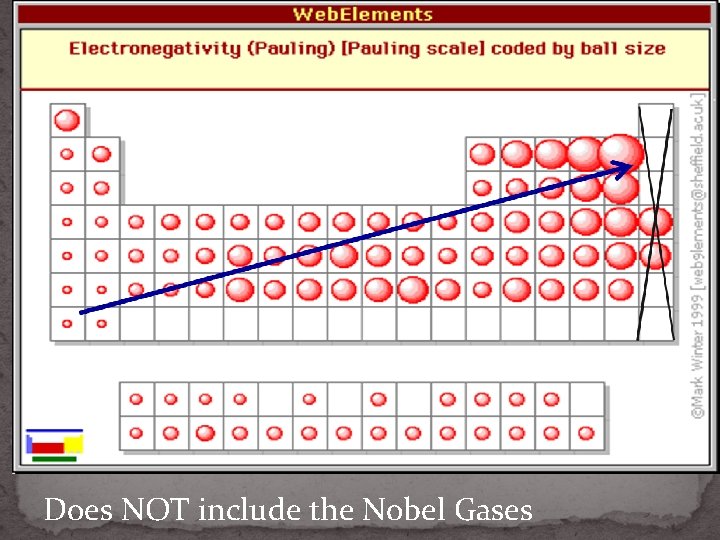
Does NOT include the Nobel Gases

Which atom has higher Electronegativity? 1. 2. 3. 4. 5. 6. Na or Mg K or Br F or Br Ca or Ga Li or S Br or As

7. Octet Rule = 8 (valence e ) - (back) Gain, lose, or share e- to be STABLE like Noble gases s 2 p 6 **Exception energy level 1 Hydrogen only wants 1 more e- to be like He (already stable)

Ions: atoms trying to obey the octet rule will… Gain or Lose e- based on what's EASIER Cation (+) loses e- = positive ion Anion (-) gains e- = negative ion Group must Charge Group must charge 1 loses 1 e- 1+ 15 Add 3 e- 3 - 2 Loses 2 e- 2+ 16 Add 2 e- 2 - 13 Loses 3 e- 3+ 17 Add 1 e- 1 -

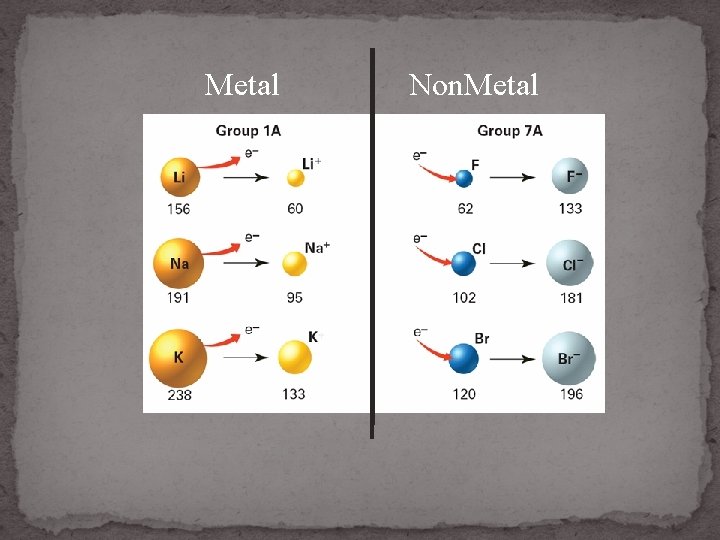
Metal ION Size 1. Lose valence e- , lose E level 2. Stronger nuclear charge = becomes smaller
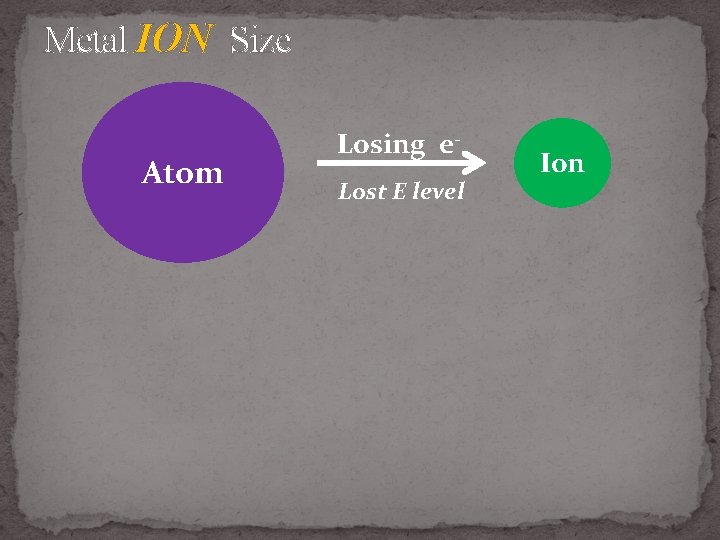
Metal ION Size Atom Losing e. Lost E level Ion

Non-Metal ION Size 1. Gain valence e 2. Weaker nuclear charge = become BIGGER
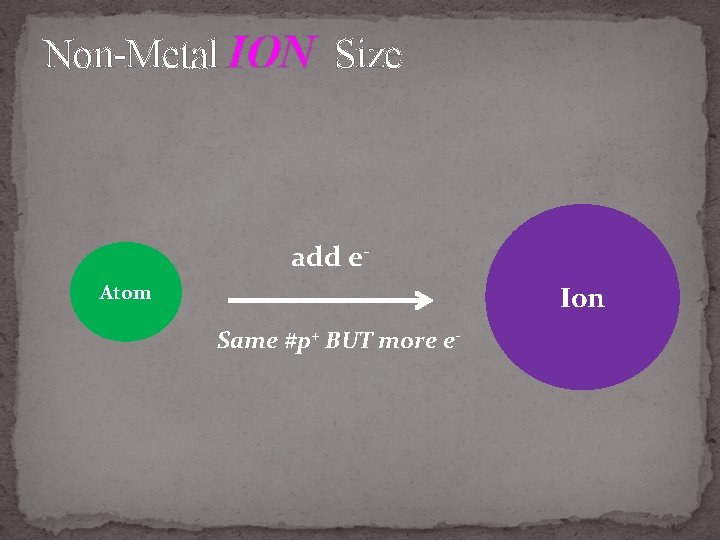
Non-Metal ION Size add e. Atom Ion Same #p+ BUT more e-
Metal Non. Metal
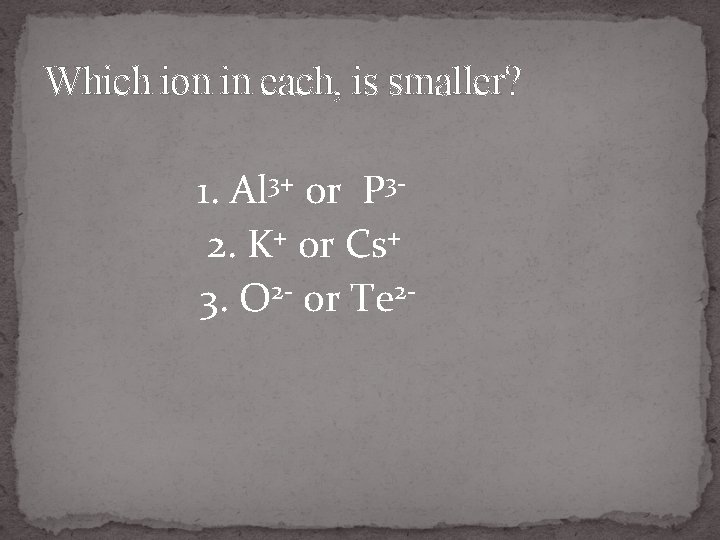
Which ion in each, is smaller? 1. Al 3+ or P 32. K+ or Cs+ 3. O 2 - or Te 2 -
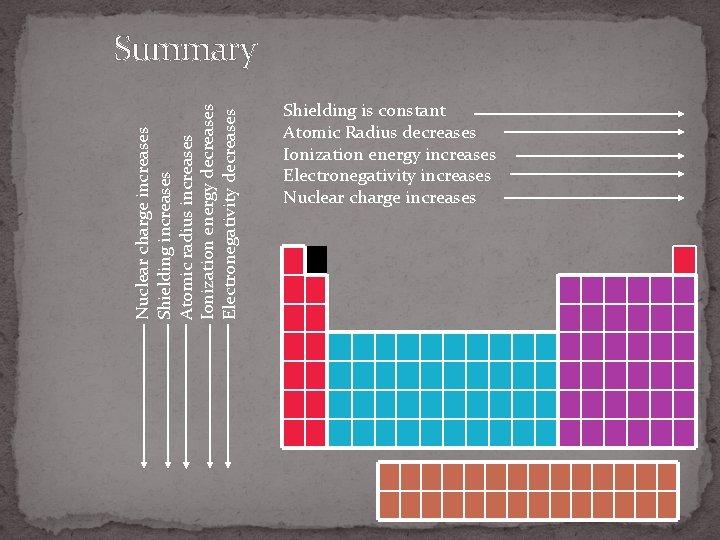
Nuclear charge increases Shielding increases Atomic radius increases Ionization energy decreases Electronegativity decreases Summary Shielding is constant Atomic Radius decreases Ionization energy increases Electronegativity increases Nuclear charge increases

Warm Up 3. 1 Ranking Elements by Atomic Size PROBLEM: Using only the periodic table (not Figure 8. 15), rank each set of main group elements in order of decreasing atomic size: (a) Ca, Mg, Sr (b) K, Ga, Ca (c) Br, Rb, Kr (d) Sr, Ca, Rb PLAN: Elements in the same group increase in size and you go down; elements decrease in size as you go across a period. SOLUTION: (a) Sr > Ca > Mg (b) K > Ca > Ga These elements are in Group 2 A(2). These elements are in Period 4. (c) Rb > Br > Kr Rb has a higher energy level and is far to the left. Br is to the left of Kr. (d) Rb > Sr > Ca Ca is one energy level smaller than Rb and Sr. Rb is to the left of Sr.

Warm Up 3. 2 PROBLEM: Ranking Elements by First Ionization Energy Using the periodic table only, rank the elements in each of the following sets in order of decreasing IE 1: (a) Kr, He, Ar PLAN: (b) Sb, Te, Sn (c) K, Ca, Rb (d) I, Xe, Cs IE decreases as you proceed down in a group; IE increases as you go across a period. SOLUTION: (a) He > Ar > Kr Group 8 A(18) - IE decreases down a group. (b) Te > Sb > Sn Period 5 elements - IE increases across a period. (c) Ca > K > Rb Ca is to the right of K; Rb is below K. (d) Xe > I > Cs I is to the left of Xe; Cs is further to the left and down one period.
- Slides: 38