PERIODIC TRENDS THE PERIODIC LAW When arranged by
PERIODIC TRENDS

THE PERIODIC LAW When arranged by increasing atomic number, the elements display a regular and repeating pattern of chemical and physical properties

PERIODIC LAW (CONT) Atoms with similar properties appear in groups or families on the table. Properties are similar because they all have the same # of valence e-

Atomic Radius Atomic radius is the distance from the center of an atom’s nucleus to its outermost electron. SIZE OF ATOM Unit commonly used is pm picometer= 10 -12 m or angstrom
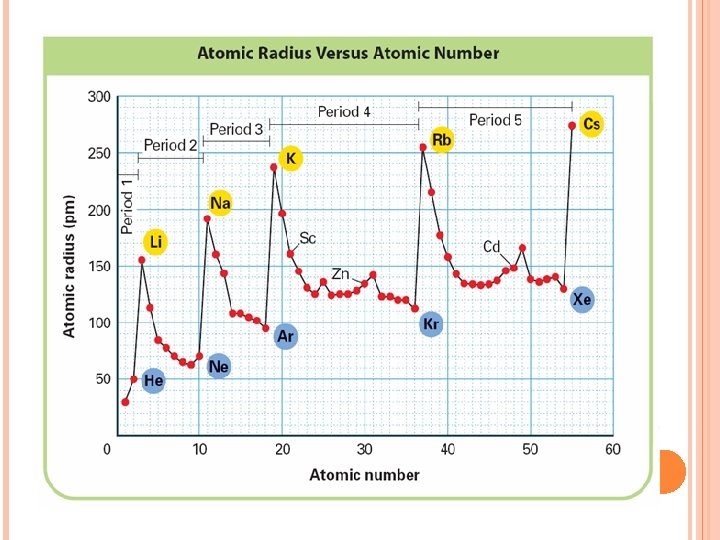
It increases down the group. Why? There are more energy levels as you go down

It decreases to the right. (across a period) Why? More protons in the nucleus higher electrical force pulls electrons closer to nucleus.
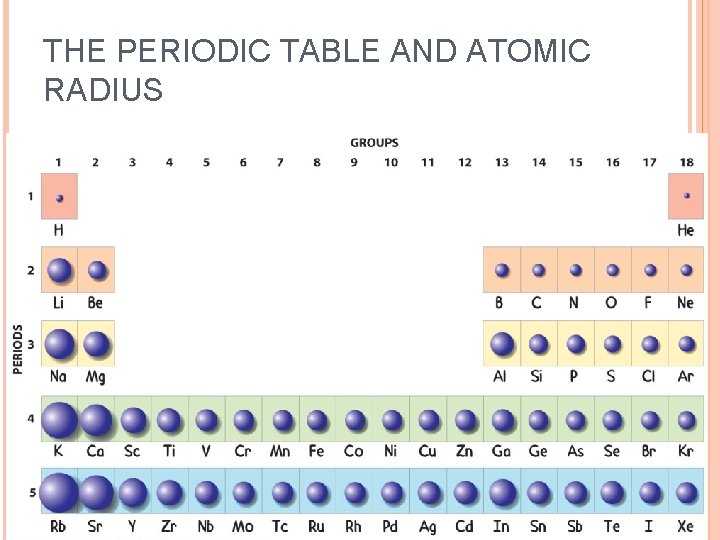
THE PERIODIC TABLE AND ATOMIC RADIUS

EXAMPLE: Which is larger: a lithium atom or a fluorine atom? A lithium atom

EXAMPLE: Which is larger: an arsenic atom or a sulfur atom? An arsenic atom
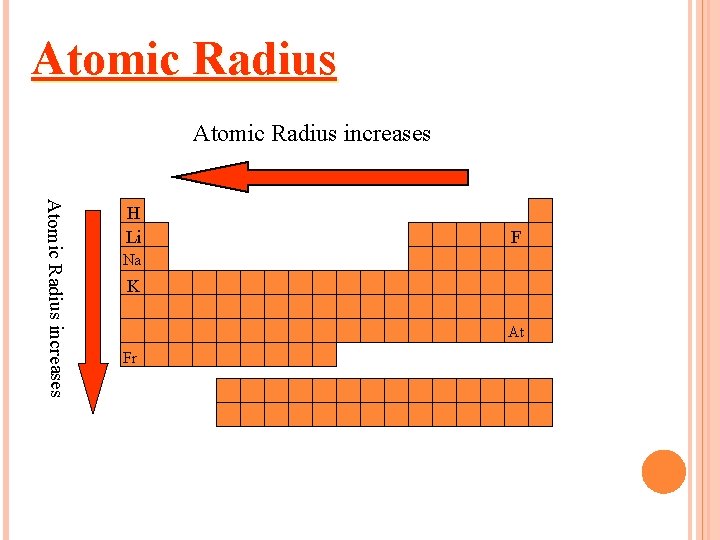
Atomic Radius increases H Li F Na K At Fr

Ionization Energy needed to remove one of atom’s electrons from its outermost shell Unit is e. V or k. J Reflection of how strongly an atom holds onto its outermost electron. Atoms with high ionization energies hold onto their electrons very tightly. Atoms with low ionization energies are more likely to lose one or more of their outermost electron.
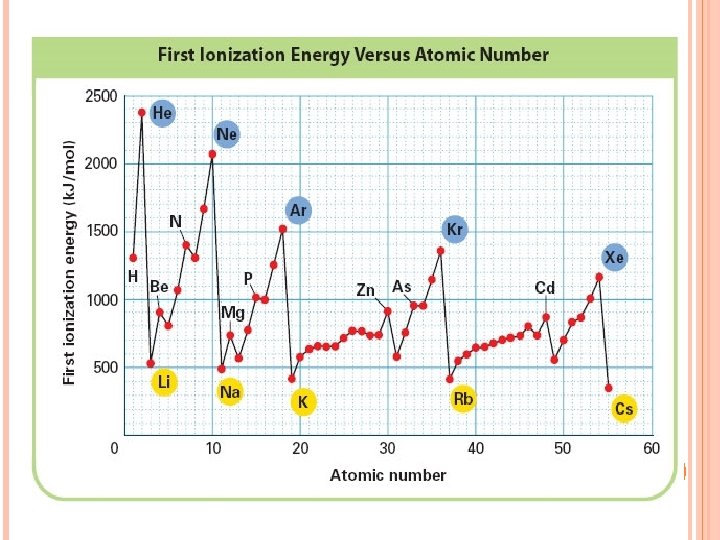

HOW DOES IONIZATION ENERGY CHANGE DOWN A GROUP? The first ionization energy decreases as you move down a group.

WHY? The size of the atom increases. (more energy levels added-the inner eshield the outer e- from the nucleus so less tightly held Electron is farther from the nucleus. (easy to snatch)
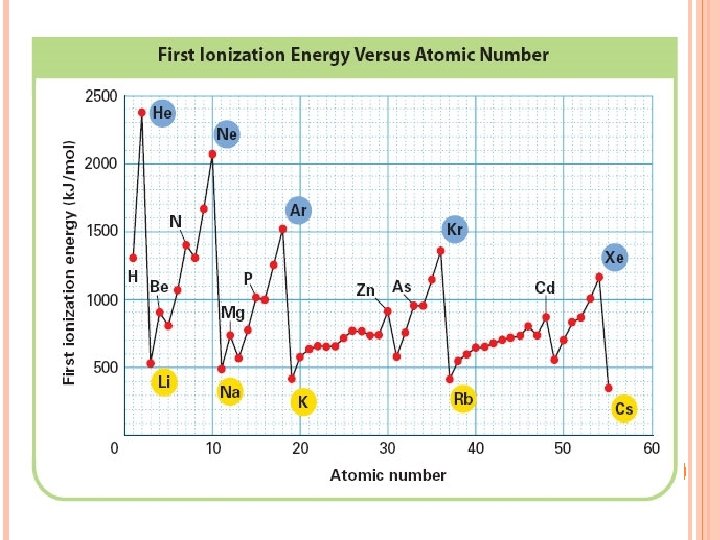

HOW DOES IONIZATION ENERGY CHANGE ACROSS A PERIOD? The first ionization energy increases as you move from left to right across a period.

WHY? Nuclear charge increases – more protons. Attraction of the electron to the nucleus increases. (harder to take)
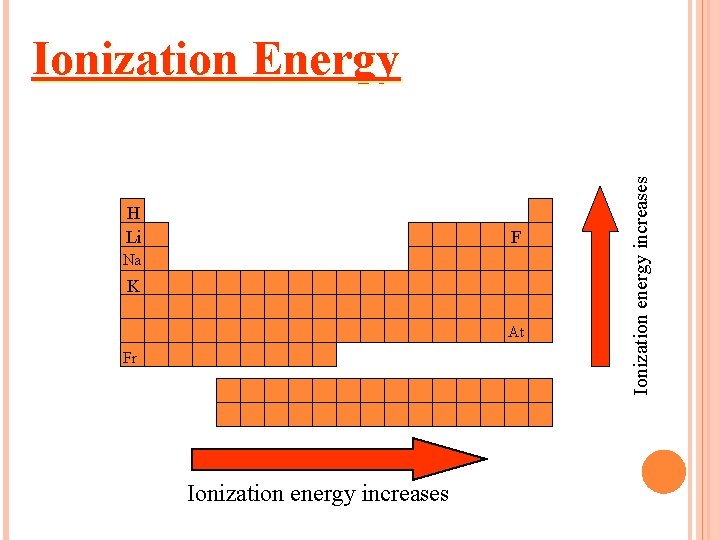
H Li F Na K At Fr Ionization energy increases Ionization Energy
DENSITY Ratio of mass to volume Units are g/cm^3 or g/m. L General increase down a group and across a period for metals –larger heavier atoms

REACTIVITY The ability of an element to combine with other elements

REACTIVITY IN METALS Increases as you go down a group (lower ionization energy, easier to remove an e-) Decreases as you move across a period (higher ionization energy, harder to remove an e-) Most reactive metal--Fr
REACTIVITY IN NONMETALS Decrease down a group Increase across a period Most reactive nonmetal--F

ELECTRONEGATIVITY Ability of an atom to attract e- in a chemical bond
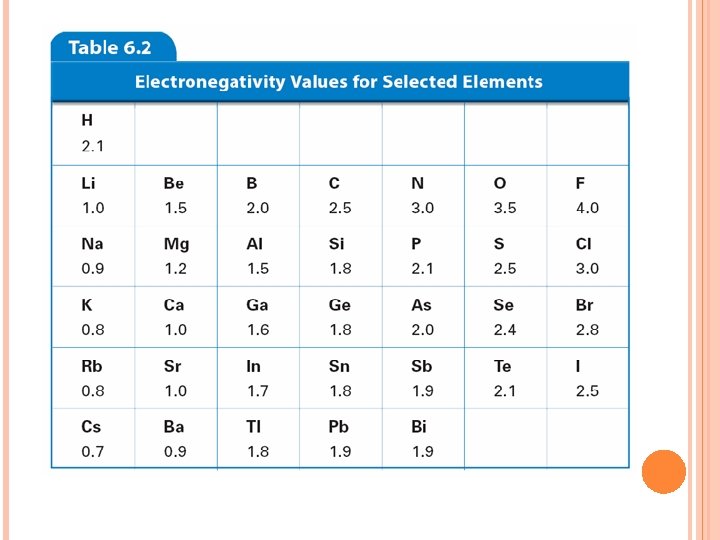

TRENDS Decreases down a group Why? Atom is larger, harder for it to attract e Increases across a period Why? Atom wants the e- more Fluorine is the most electronegative element
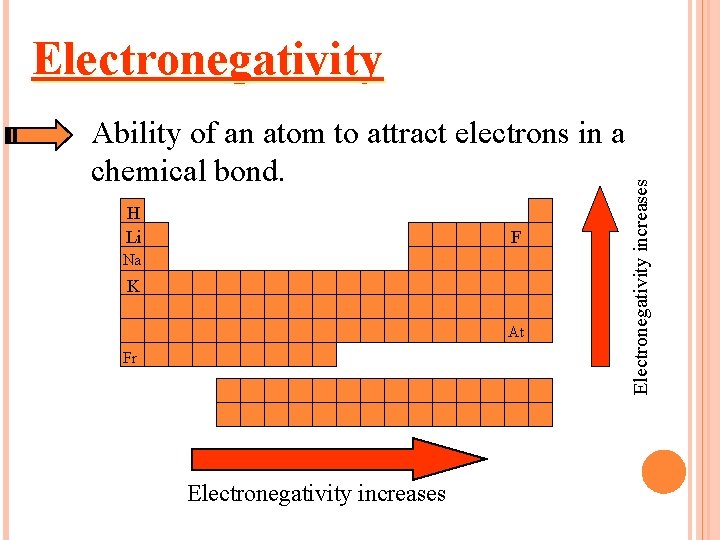
Ability of an atom to attract electrons in a chemical bond. H Li F Na K At Fr Electronegativity increases Electronegativity

ION—CHARGED ATOM Positive ion ---removal of electron (cation) Negative ion--- addition of electron (anion)

IONIC RADIUS The size of the ion— (charged atom)

IONIC SIZE Metallic elements easily lose electrons. Non-metals more readily gain electrons. How does losing or gaining an electron effect the size of the atom (ion) ?

IONIC RADIUS Anion, (negative ion) Gains e its size increases, since the nuclear charge(protons)remains the same but the additional electron(s) enlarges the electron cloud.
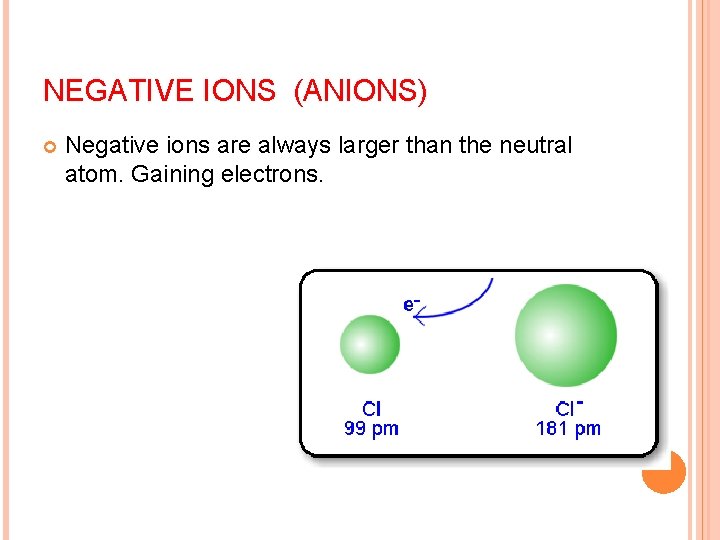
NEGATIVE IONS (ANIONS) Negative ions are always larger than the neutral atom. Gaining electrons.

IONIC RADIUS Cation (positive ion) Loses e smaller than neutral atom, since removing one or more e- makes the cloud shrink, the nuclear charge remains the same

POSITIVE IONS (CATIONS) Positive ions are always smaller that the neutral atom. Loss of outer shell electrons.

ION SIZE TRENDS IN PERIODS. Going from left to right there is a decrease in size of positive ions. Starting with group 5, there is sharp increase followed by a decrease in the size of the anion as you move from left to right.

ION SIZE TRENDS IN COLUMNS. Ion size increases as you move down a column for both positive and negative ions
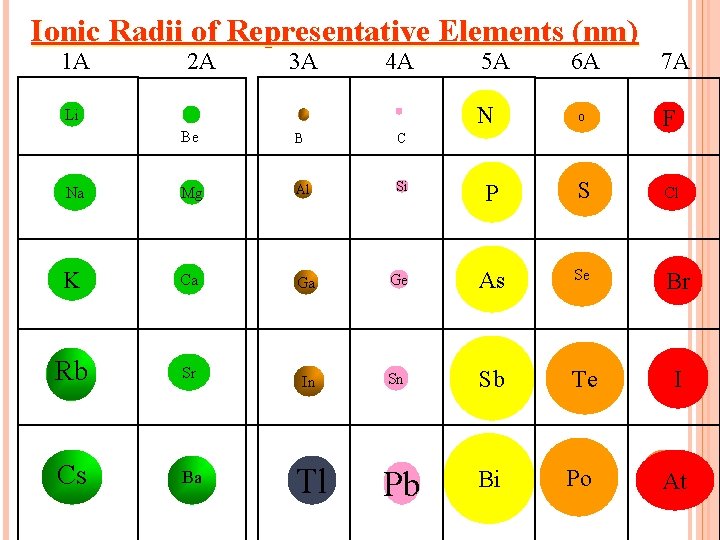
Ionic Radii of Representative Elements (nm) 1 A 2 A 3 A 4 A 5 A N Li 6 A 7 A O F Be B C Na Mg Al Si P S Cl K Ca Ga Ge As Se Br Rb Sr In Sn Sb Te I Cs Ba Pb Bi Po At Tl
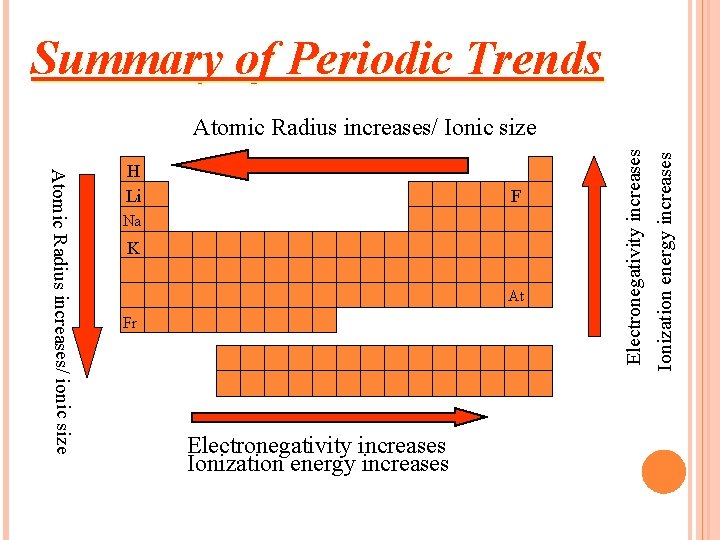
Summary of Periodic Trends F Na K At Fr Electronegativity increases Ionization energy increases Atomic Radius increases/ ionic size H Li Electronegativity increases Atomic Radius increases/ Ionic size
- Slides: 38