Periodic Trends Periodic Trends Because the elements are

Periodic Trends

Periodic Trends Because the elements are arranged as they are, they exhibit trends based on their chemical and physical properties.
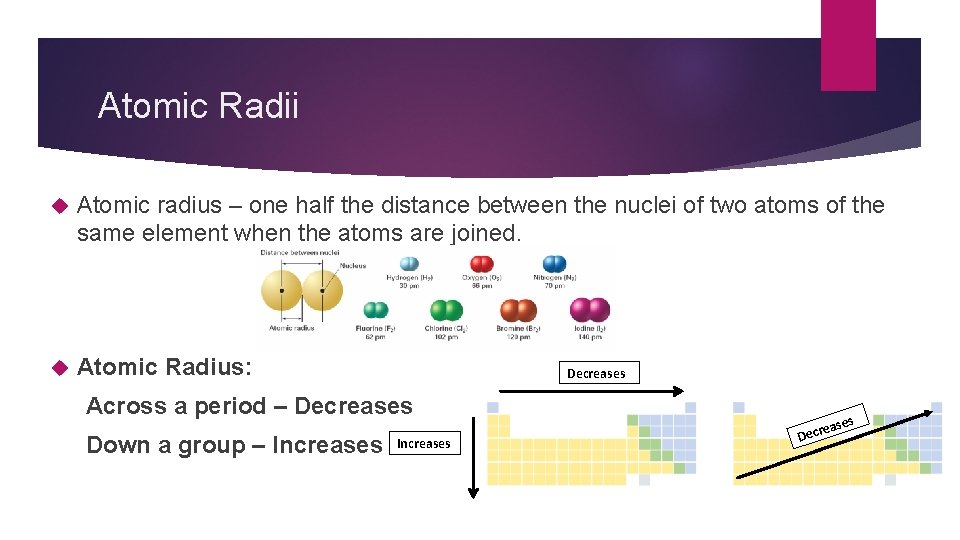
Atomic Radii Atomic radius – one half the distance between the nuclei of two atoms of the same element when the atoms are joined. Atomic Radius: Decreases Across a period – Decreases Down a group – Increases e Decr

Atomic Radii Down a group – increasing energy levels which makes the atom larger Across a period – remain in the same energy level, but add p+ which pull e- closer, so the atom gets smaller

Ionic Radius – radius of the atom after it creates an ion Across a period - cation size decreases and anion size decreases. Down a group - Increases. Cation size Decreases Increases Anion size Decreases

Ions & Ionic Radius During reactions, metal atoms tend to LOSE electrons and nonmetal atoms tend to GAIN electrons. When atoms lose or gain electrons, they create ions. This transfer of electrons changes the size of the ions that form. Cations Anions are smaller than their respective atoms. are larger than their respective atoms.

Ionization Energy When atoms lose or gain electrons, they create ions. Positive ions are called cations. They lose an electron to create a positive charge. Negative ions are called anions. They gain an electron to create a negative charge. Ionization Energy - the energy required to remove an electron from an atom. The energy required to remove the first electron from an atom is called the first ionization energy. The energy required to remove an electron from an ion with a 1+ charge is called the second ionization energy.
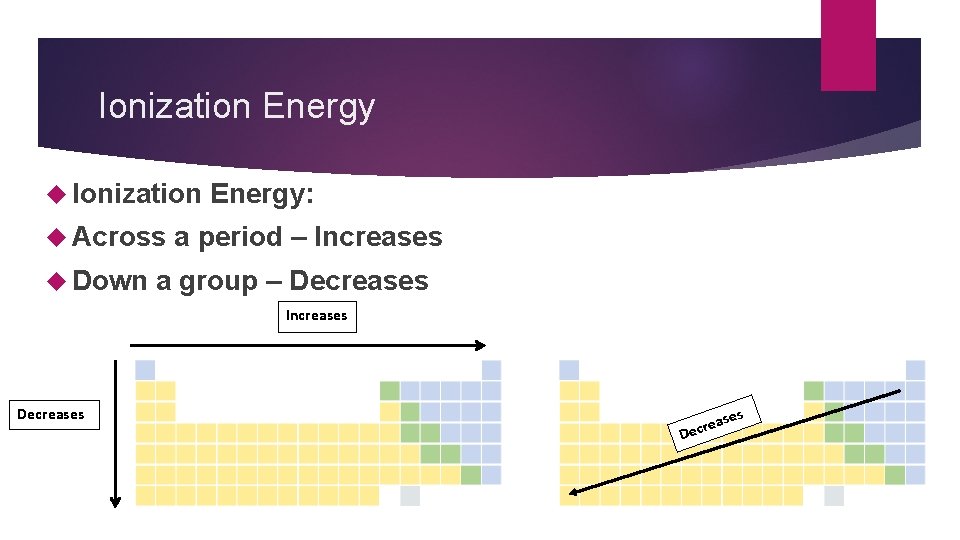
Ionization Energy Ionization Across Down Energy: a period – Increases a group – Decreases Increases Decreases D ase e r c e s

Ionization Energy Across a period – electrons are closer to nucleus so more energy is needed to remove Down a group – electrons are further from nucleus, so less energy is needed to remove

Electron Affinity - the amount of energy released or spent when an electron is added to a neutral atom or molecule in the gaseous state to form a negative ion. It is a negative value because it refers to energy and a negative sign means energy is released Across a period – Increases (more energy released) Down a group – Decreases (less energy released) Increases ase e r c De Decreases s
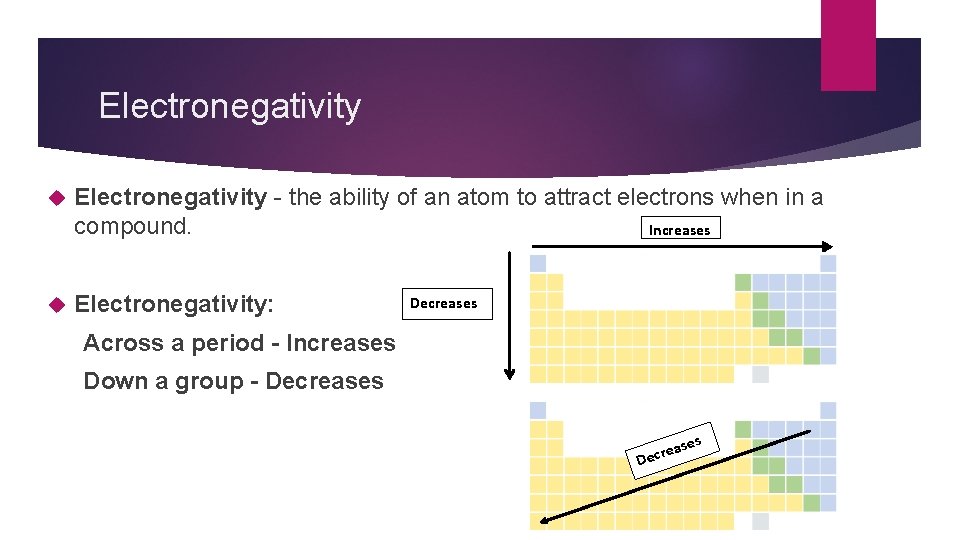
Electronegativity - the ability of an atom to attract electrons when in a compound. Increases Electronegativity: Decreases Across a period - Increases Down a group - Decreases s D ase ecre
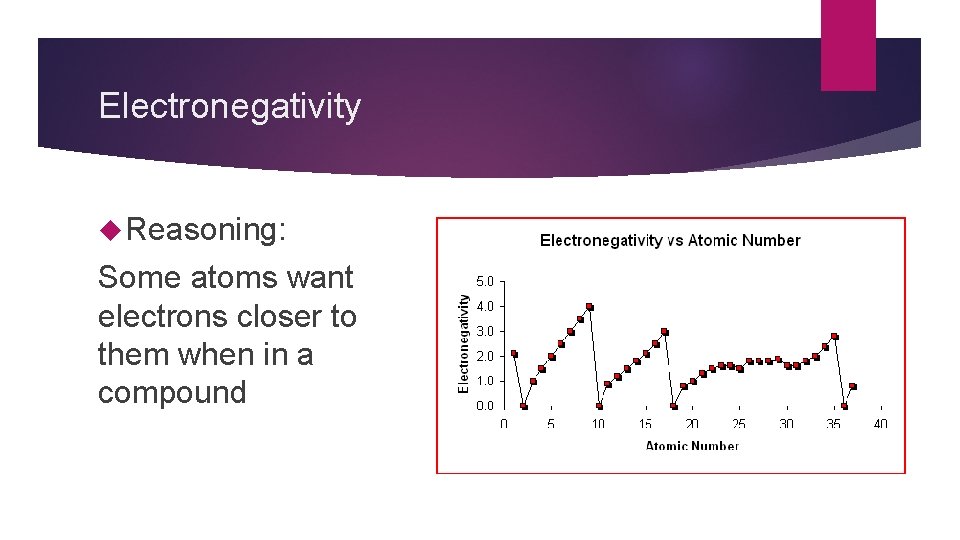
Electronegativity Reasoning: Some atoms want electrons closer to them when in a compound
- Slides: 12