Periodic Trends Ionization Energy Electron Affinity Coulombs Law

Periodic Trends— Ionization Energy, Electron Affinity

Coulomb’s Law Addresses the force of attraction between the positive nucleus and negative electron F = q e q p r 2 qe = charge of electron qp = charge of nucleus INVERSE relationship between attractive force and distance Attractive force is DIRECTLY related to the charge on the electron and the charge in the nucleus

First Ionization Energy (IE) If an electron is given enough energy (in the form of a photon) to overcome the effective nuclear charge holding the electron in the cloud, it can leave the atom completely. Energy required to remove the outermost electron from an atom Amount of energy needed to remove ONE electron from a neutral atom Energy needed to remove electrons from ground state to gaseous state in an atom Formation of positive ions (cations)

Ionization Energy (cont. ) Formation of positive ions (cations) The larger the atom is, the easier its electrons are to remove. Ionization energy and atomic radius are inversely proportional.
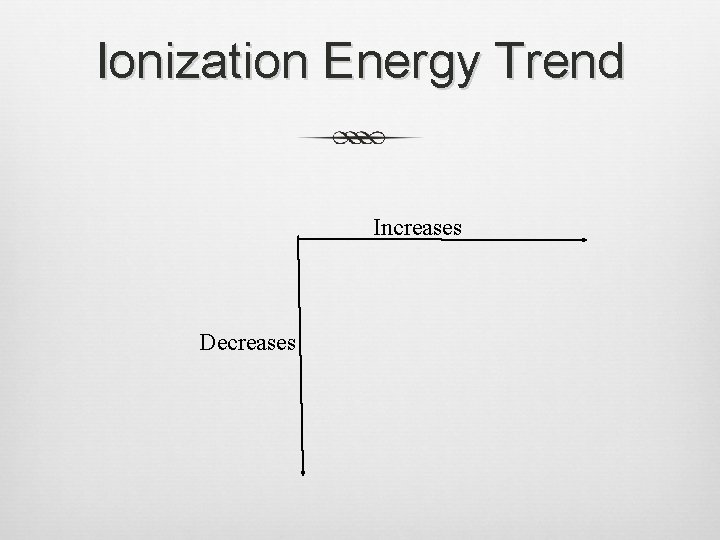
Ionization Energy Trend Increases Decreases
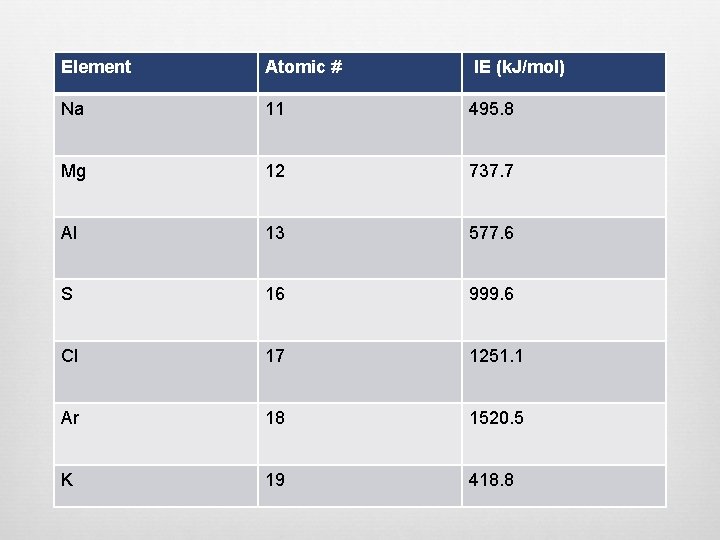
Element Atomic # IE (k. J/mol) Na 11 495. 8 Mg 12 737. 7 Al 13 577. 6 S 16 999. 6 Cl 17 1251. 1 Ar 18 1520. 5 K 19 418. 8

Ionization Energy (cont. ) 1 st ionization energy Energy required to remove 1 st electron from atom Taken from highest energy level Easiest to remove Energy increases as more electrons are removed

Can we have 2 nd, 3 rd, 4 th, etc. Ionization Energies? Energy needed to remove additional electrons from an atom Energy increases as more electrons are removed. Removing electrons from lower energy levels, close to nucleus
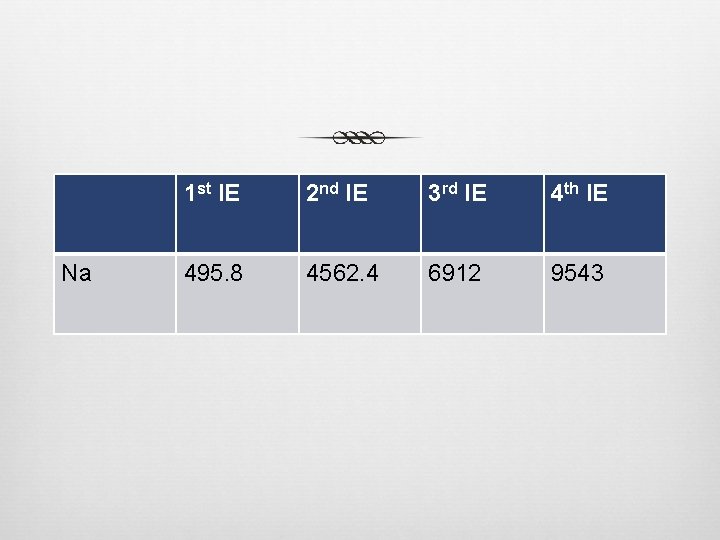
Na 1 st IE 2 nd IE 3 rd IE 4 th IE 495. 8 4562. 4 6912 9543
Ionization Energy

Example 1: Place these elements in order of INCREASING 1 st ionization energy. 1) Mg, S, Si 2) As, N, P (p. 321 in text)
What does affinity mean?

Electron Affinity Creating negative ions (anions) Energy change with the addition of an electron to an atom energy change that occurs when electrons added to gaseous atom Energy is released when electrons are added, exothermic process (negative value)

Electron Affinity Electron tends to enter partially filled subshell or go to the next energy level Does not follow predictable trend like atomic radii and ionization energy An atom’s “desire/affinity” for more electrons, wants to get more electrons ! ! ! Metals—decrease electron affinity. Nonmetals—increase electron affinity, more reactive Stable atoms—full octet

Electron Affinity Trend Increases Decreases

Homework Atomic Theory III Worksheet #1 -21 due Thursday !!!
- Slides: 16