Periodic Trends Elemental Properties and Patterns The Periodic



























- Slides: 32

Periodic Trends Elemental Properties and Patterns

The Periodic Law • Dmitri Mendeleev - first to publish an organized periodic table of known elements.

The Periodic Law • Mendeleev even predicted the properties of undiscovered elements. https: //www. youtube. com/watch? v=f. Pnw. BITSmg. U

The Periodic Law • Says that: “When arranged by increasing atomic number, the chemical elements display a regular and repeating pattern of chemical and physical properties. ”

So… • Atoms with similar properties appear in groups or families (vertical columns) on the periodic table. • They are similar because they all have the same number of valence (outer shell) electrons, which determines their chemical behavior.

Valence Electrons • For Groups 1, 2, 13, 14, 15, 16, 17, 18: The digit farthest to the right is the number of valence electrons. Example: 15 • Groups 3 -12? • Many have 1 -2, but figuring it out is more complicated.

A Different Type of Grouping Besides the 4 blocks of the table, there is another way of classifying element: • Metals • Nonmetals • Metalloids

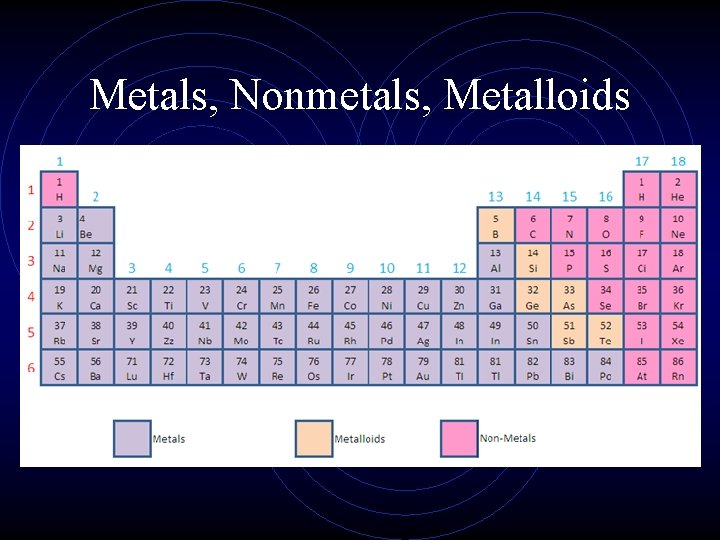
Metals, Nonmetals, Metalloids

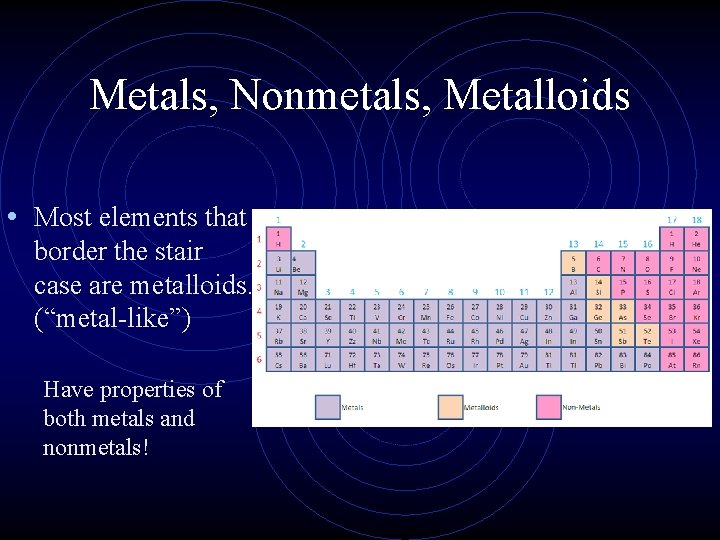
Metals, Nonmetals, Metalloids • Most elements that border the stair case are metalloids. (“metal-like”) Have properties of both metals and nonmetals!

Metals • Lustrous (shiny), malleable, ductile, and are good conductors of heat and electricity. • Mostly hard – not brittle • They are mostly solids at room temp.

Nonmetals • The opposite! • They are dull, brittle, nonconductors (insulators). • Some are solid, but many are gases, and a few are liquid.

Metalloids • Have properties both metals & nonmetals! • Shiny but brittle. • Semiconductors.

The Octet Rule • The “goal” of most atoms (except H, Li and Be) is to have an octet (group of 8 electrons) in their valence energy level. • Metals generally give electrons, nonmetals take them from other atoms. Remember… • Atoms that have gained or lost electrons are called ions.

Ions • When an atom gains an electron, it becomes negatively charged and is called an (anion. ) • When an atom loses an electron, it becomes positively charged (cation). Think of the “t” like a “+” sign.

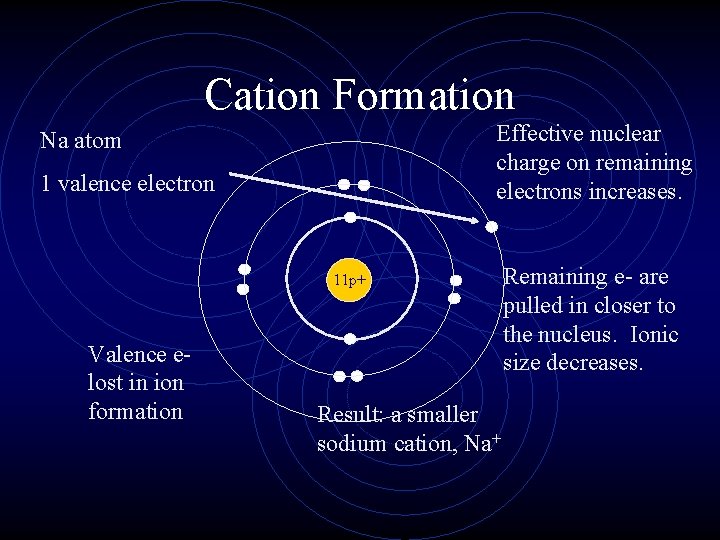
Cation Formation Effective nuclear charge on remaining electrons increases. Na atom 1 valence electron 11 p+ Valence elost in ion formation Result: a smaller sodium cation, Na+ Remaining e- are pulled in closer to the nucleus. Ionic size decreases.

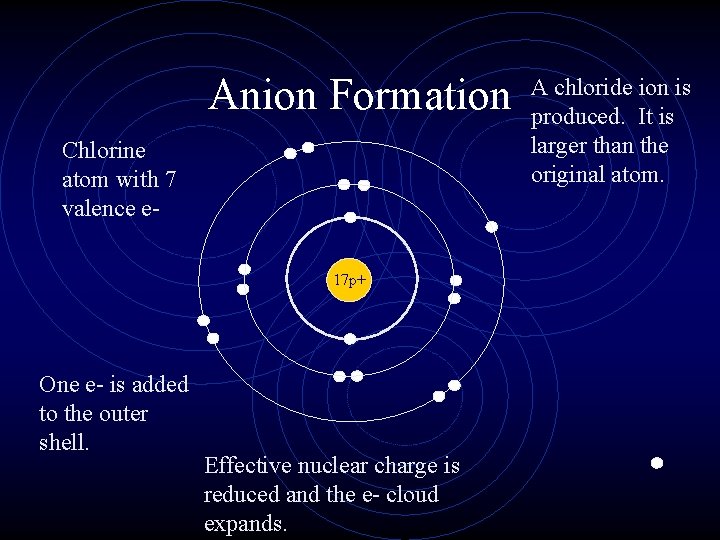
Anion Formation Chlorine atom with 7 valence e 17 p+ One e- is added to the outer shell. Effective nuclear charge is reduced and the e- cloud expands. A chloride ion is produced. It is larger than the original atom.





Periodic Trends • There are several important atomic characteristics that show predictable trends that you should know. • The first and most important is atomic radius.

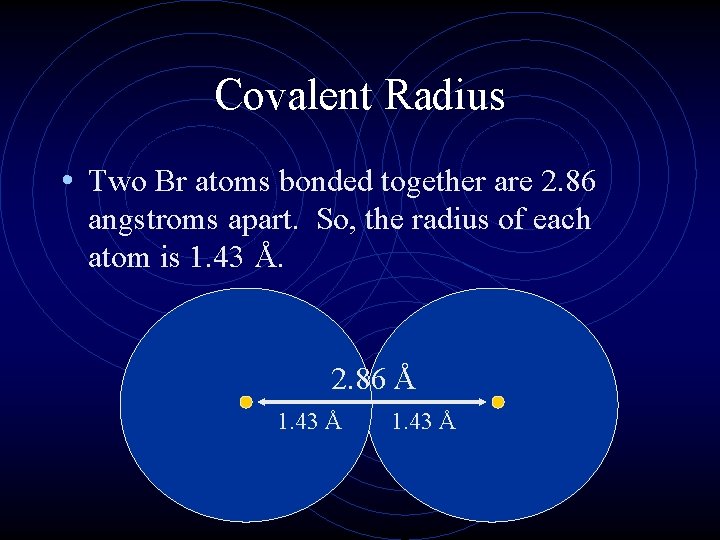
Atomic Radius • Is the distance from the center of the nucleus to the “edge” of the electron cloud. • Since that is difficult to define, scientists use covalent radius, half the distance between the nuclei of 2 bonded atoms. • Atomic radii are usually measured in picometers (pm) or angstroms (Å). An angstrom is 1 x 10 -10 m.

Covalent Radius • Two Br atoms bonded together are 2. 86 angstroms apart. So, the radius of each atom is 1. 43 Å. 2. 86 Å 1. 43 Å

Atomic Radius The trend for atomic radius in a GROUP is to go from smaller at the top to larger at the bottom. • Why? • With each step down the family, we add energy levels to the electron cloud, making the atoms larger.

Atomic Radius • As you move ACROSS a PERIOD, atoms are smaller because of an increased attraction between nucleus and electron cloud. • The increased attraction pulls the cloud in, making atoms smaller as we move from left to right.

Effective Nuclear Charge • What keeps electrons from simply flying off into space? • Effective nuclear charge is the pull that an electron “feels” from the nucleus. • The closer an electron is to the nucleus, the more pull it feels. • As effective nuclear charge increases, the electron cloud is pulled in tighter.

Ionization • If an electron is given enough energy to overcome its attraction to the nucleus, it can leave the atom completely. • The atom has been “ionized” or charged. • Number of protons ≠ number of electrons.

Ionization Energy • Energy required to remove an electron from an atom is ionization energy. (measured in kilojoules, k. J) • The larger the atom is, the easier its electrons are to remove. • Ionization energy and atomic radius are inversely proportional. • As you move down, it decreases! • As you move across, it increases!

Electronegativity • Electronegativity is a measure of an atom’s attraction for another atom’s electrons. • Metals are usually electron givers and have low electronegativity. • Nonmetals are electron takers and have high electronegativity. • What about the noble gases? Make a prediction to your table partners.

Ionic Radius • Cations are always smaller than the original atom. • Conversely, anions are always larger than the original atom.

Review Video for Atomic Trends • https: //www. youtube. com/watch? v=0 t. P 6 b. V 89 log

Link to bonding animations • http: //bcs. whfreeman. com/thelifewire/conte nt/chp 02/02020. html • http: //www. youtube. com/watch? v=c. Zy 8 t. GF V 8 QE&list=TL 6 TJ_jx 1 X 7 SKn. BP 20 k. At 90 e Sd. Tpg. QOR 8 H