Periodic Trends Elemental Properties and Patterns The Periodic








































- Slides: 50

Periodic Trends Elemental Properties and Patterns


The Periodic Law • Dimitri Mendeleev was the first scientist to publish an organized periodic table of the known elements.

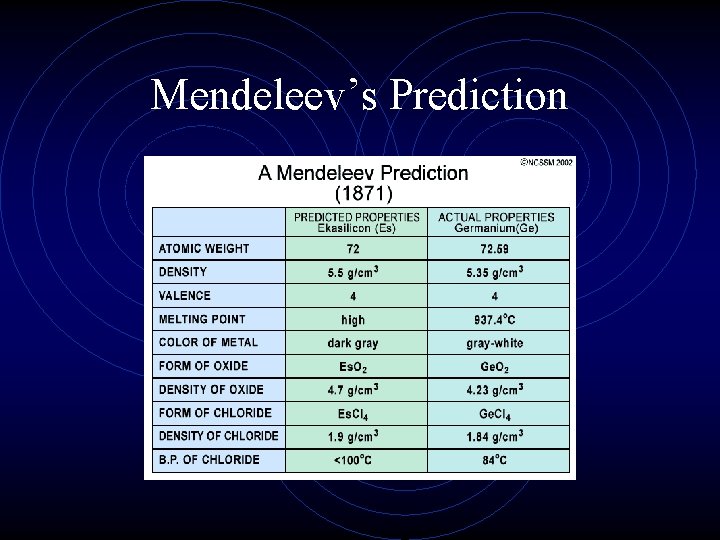
The Periodic Law • Mendeleev even went out on a limb and predicted the properties of 2 undiscovered elements. • He was very accurate in his predictions, which led the world to accept his ideas about periodicity and a logical periodic table.

Mendeleev’s Prediction

• Mendeleev arranged the elements by increasing atomic mass. • The modern periodic table is in order of increasing atomic number. • Mosley is given credit for arranging by atomic number.

The Periodic Law • When arranged by increasing atomic number, the chemical elements display a regular and repeating pattern of chemical and physical properties.

The Periodic Law • Atoms with similar properties appear in groups or families (vertical columns) on the periodic table. • They are similar because they all have the same number of valence (outer shell) electrons, which governs their chemical behavior.

Valence Electrons • Do you remember how to tell the number of valence electrons for elements in the s- and p-blocks? • How many valence electrons will the atoms in the d-block (transition metals) and the fblock (inner transition metals) have? • Most have 2 valence e-, some only have 1.

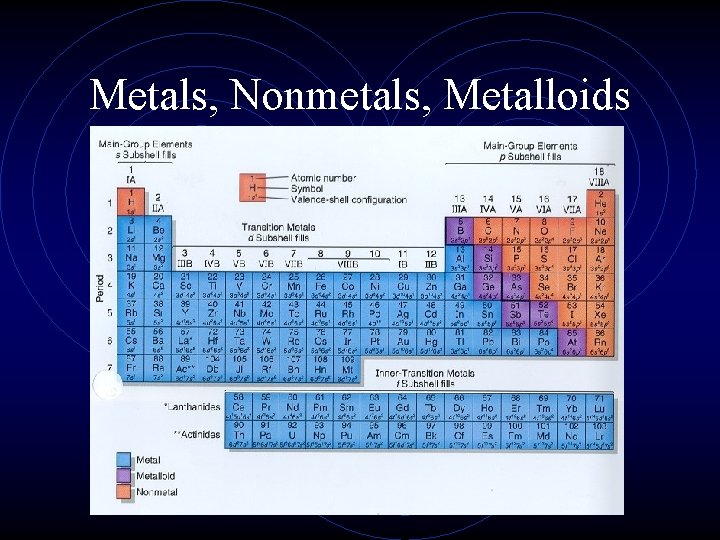
A Different Type of Grouping • Besides the 4 blocks of the table, there is another way of classifying element: • Metals • Nonmetals • Metalloids or Semi-metals.

Metals, Nonmetals, Metalloids

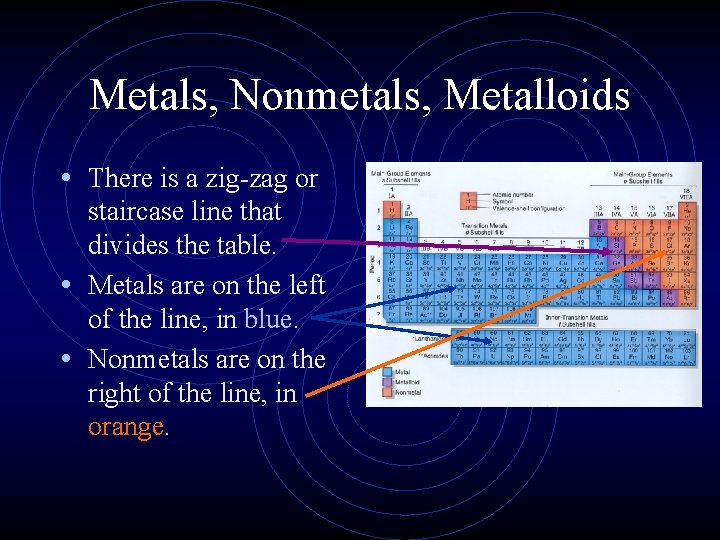
Metals, Nonmetals, Metalloids • There is a zig-zag or staircase line that divides the table. • Metals are on the left of the line, in blue. • Nonmetals are on the right of the line, in orange.

Metals, Nonmetals, Metalloids • Elements that border the stair case, shown in purple are the metalloids or semimetals. • There is one important exception. • Aluminum is more metallic than not.

Metals • Metals are lustrous (shiny), malleable, ductile, and are good conductors of heat and electricity. • They are mostly solids at room temp. • What is one exception?

Nonmetals • Nonmetals are the opposite. • They are dull, brittle, nonconductors (insulators). • Some are solid, but many are gases, and Bromine is a liquid.

Metalloids • They have characteristics of both metals and nonmetals. • They are shiny but brittle. • And they are semiconductors. • What is our most important semiconductor?


Periodic Trends - Important Concepts • Valence electrons- electrons in the outermost energy level • Core electrons- electrons in the inner energy levels • When an atom loses an electron, which electron is removed? valence

Effective Nuclear Charge (Zeff) • What keeps electrons from simply flying off into space? • Effective nuclear charge is the pull that an electron “feels” from the nucleus. • The closer an electron is to the nucleus, the more pull it feels. (Higher Zeff) • As effective nuclear charge increases, the electron cloud is pulled in tighter.

Shielding Effect • The core electrons in an atom “shield” the valence electrons from the pull of the nucleus • As the shielding increases (more ebetween nucleus and valence e-), the effective nuclear charge _____. decreases

Bohr Models

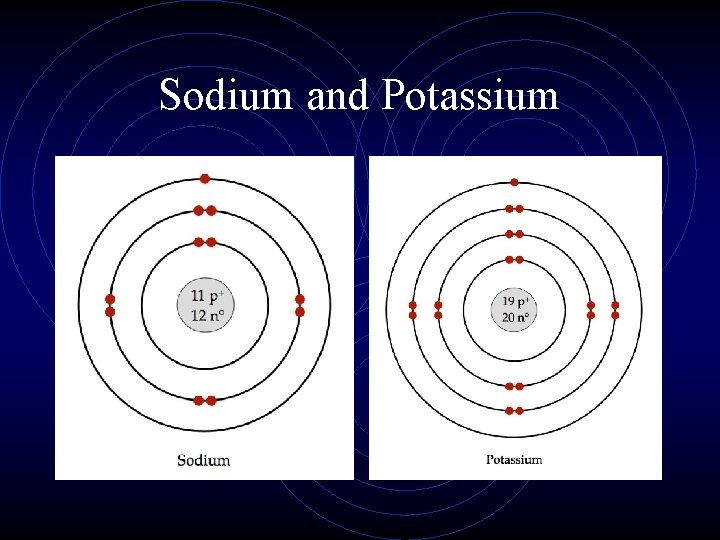
Sodium and Potassium

Applications • • Which atom (Na or K) is a larger atom? Why? Which atom has more shielding? Which atom has a higher effective nuclear charge? • Which atom would require less energy to remove the valence electron? (easiest to remove)

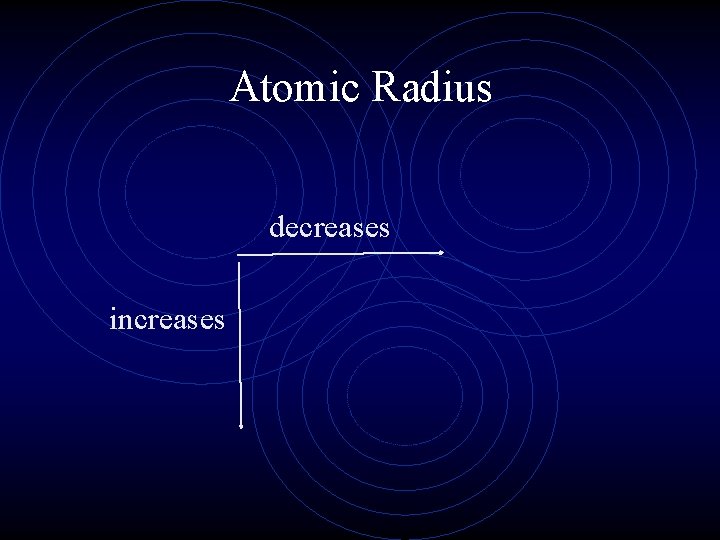
Atomic Radius (Size) • The trend for atomic radius in a vertical column is to go from smaller at the top to larger at the bottom of the family. Increases down the group. • Why? • With each step down the family, we add an entirely new energy level to the electron cloud, making the atoms larger.

Atomic Radius • The trend across a horizontal period is less obvious. • What happens to atomic structure as we move from left to right across a period? • Each step adds a proton and an electron (and 1 or 2 neutrons). • Electrons are added to existing energy levels or sublevels, we don’t add a new energy level.

Atomic Radius • The effect is that the more positive nucleus has a greater pull on the electron cloud. • The nucleus is more positive and the electron cloud is more negative. • The increased attraction pulls the cloud in, making atoms smaller as we move from left to right across a period. • The Zeff increases but the shielding does not change.

Atomic Radius decreases increases

Practice 1. Arrange the following in order of increasing size: A) O, Se, S O, S, Se B) P, Mg, Cl Cl, P, Mg C) S, F, Rb F, S, Rb D) P, Ga, O O, P, Ga

Ionization Energy • If an electron is given enough energy (in the form of a photon) to overcome the effective nuclear charge holding the electron in the cloud, it can leave the atom completely. • The atom has been “ionized” or charged. • The number of protons and electrons is no longer equal.

Ionization Energy • The energy required to remove an electron from an atom is ionization energy. (measured in kilojoules, k. J) • The larger the atom is, the easier its electrons are to remove. • Ionization energy and atomic radius are inversely proportional. • Ionization energy is always endothermic, which means energy is added to the atom to remove the electron.

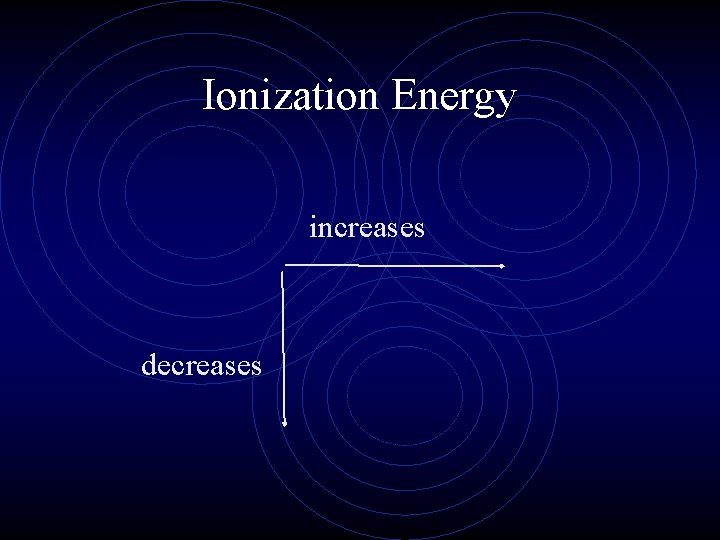
Ionization Energy • As you move down a group, the ionization energy decreases. It becomes easier to remove the valence electron. • Why? • As you move across a period, the ionization energy increases. • Why?

Ionization Energy increases decreases

Exceptions (Honors) *does not vary uniformly across period* • 2 nd period in order of increasing I. E. • Li, B, Be, C, Why? O, N, F, Ne

Practice 1. Arrange in order of increasing ionization energy: A) Br, Cl, I I, Br, Cl B) Sb, Sr, I Sr, Sb, I C) (Honors) Ge, As, Se Ge, Se, As

Electron Affinity (Honors) • What does the word ‘affinity’ mean? • Electron affinity is the energy change that occurs when an atom gains an electron (also measured in k. J). • Where ionization energy is always endothermic, electron affinity is usually exothermic, but not always.

Electron Affinity (Honors) • Electron affinity is exothermic if there is an empty or partially empty orbital for an electron to occupy. • If there are no empty spaces, a new orbital or energy level must be created, making the process endothermic. • This is true for the alkaline earth metals and the noble gases.

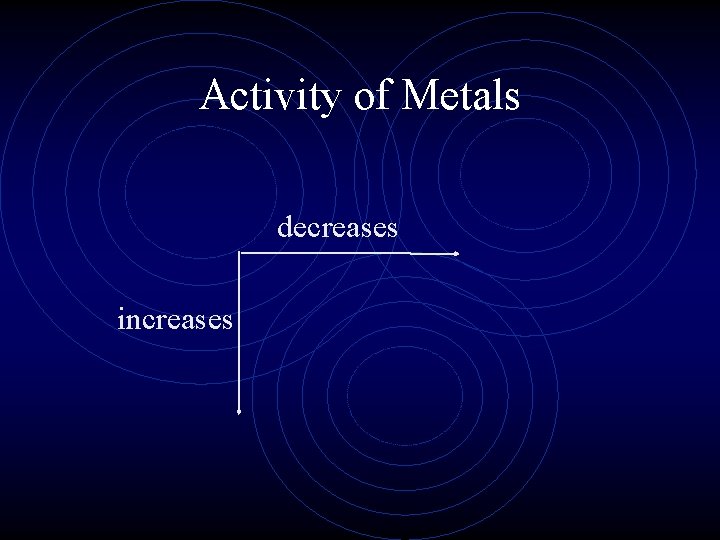
Metallic Character (Activity of Metals) • Which family of metals would be the most reactive? • Why? • As you move down a group, the activity of metals increases. • As you move across a period, the activity of metals decreases.

Activity of Metals • The activity of metals is based on how easily the metal loses an electron. • Very active metals lose their valence electrons easily.

Activity of Metals decreases increases

Examples 1. What is the most active metal A) Ca, Be, Sr Sr B) Sc, K, Ca K C) Pd, Cd, Sr Sr

Nonmetallic Character (Activity of Nonmetals) • What family is the most active of the • • nonmetals? Why? As you move down a group, the activity of nonmetals decreases. As you move from left to right across a period, the activity of nonmetals increases. What is the most active nonmetal? F

Activity of Nonmetals • The activity of nonmetals is based on how easily an atom will gain an electron. • Very active nonmetals have a strong attraction for electrons.

Activity of Nonmetals increases decreases Do not include the noble gases!

Way To Remember The Trends What trends increase down a group and across a period? decrease increase size, activity of metals


What is the Name of the Snowman? Size Activity of Metals

Ionic Radius • Cations are always smaller than the original • • • atom. The valence electrons are removed during ionization. Ca 2+ is ______than Ca. Conversely, anions are always larger than the original atom. Electrons are added to the outer energy level. S 2 - is _______than S.

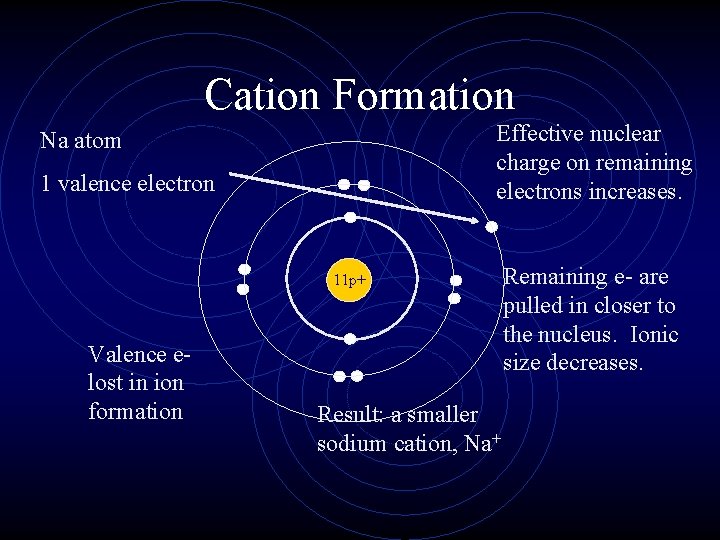
Cation Formation Effective nuclear charge on remaining electrons increases. Na atom 1 valence electron 11 p+ Valence elost in ion formation Result: a smaller sodium cation, Na+ Remaining e- are pulled in closer to the nucleus. Ionic size decreases.

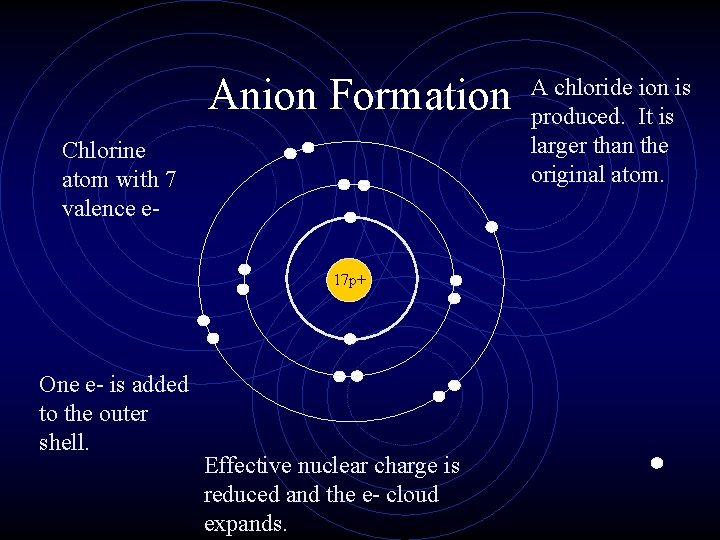
Anion Formation Chlorine atom with 7 valence e 17 p+ One e- is added to the outer shell. Effective nuclear charge is reduced and the e- cloud expands. A chloride ion is produced. It is larger than the original atom.

Examples 1. Arrange from smallest to largest: A) H, H+, H, HB) K+, Cl-, S-2, Ca+2, K+, Cl-, S-2 2. Given the following elements, which would be the largest when forming an ion? Br, Se, Rb, Sr Se 2 -