Periodic Trends Atomic Radius Definition Half of the






- Slides: 15

Periodic Trends

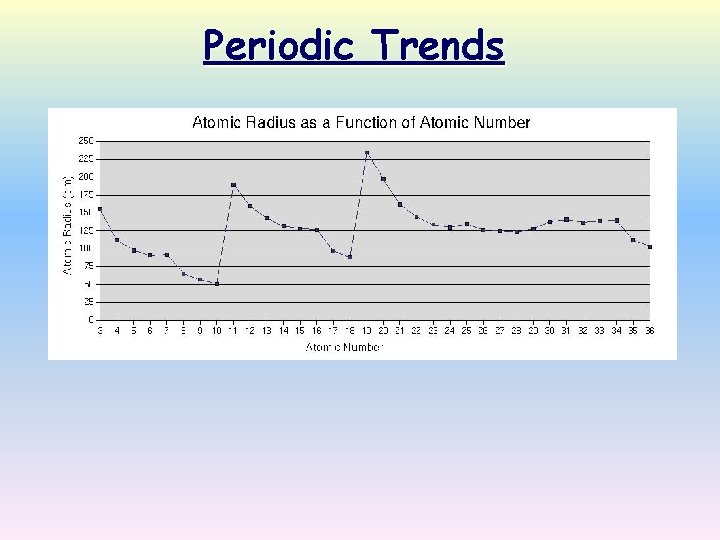
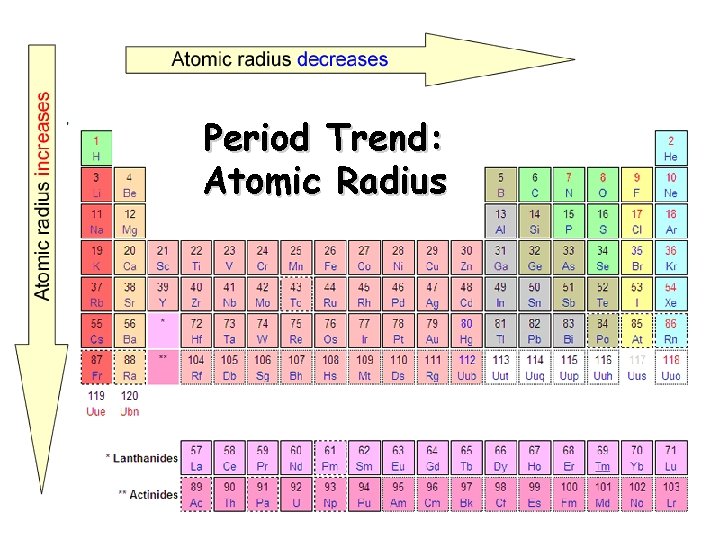
Atomic Radius Definition: Half of the distance between nuclei in covalently bonded diatomic molecule v. Radius decreases across a period v Increased effective nuclear charge due to decreased shielding v. Radius increases down a group v Each row on the periodic table adds a “shell” or energy level to the atom

Table of Atomic Radii

Period Trend: Atomic Radius

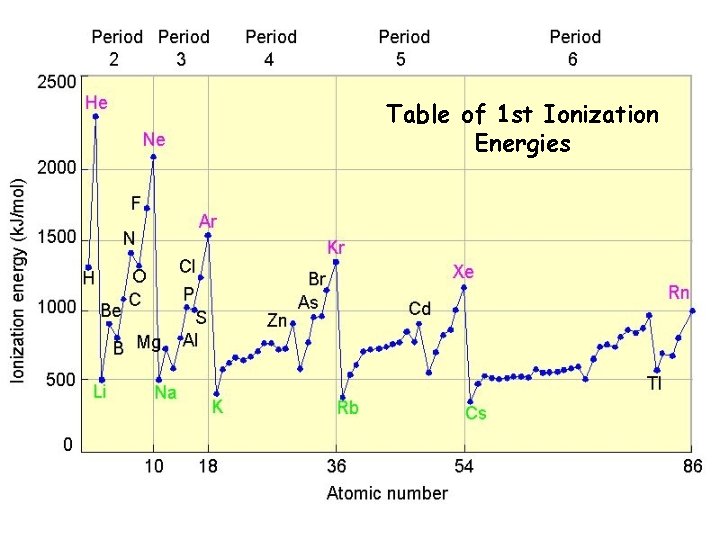
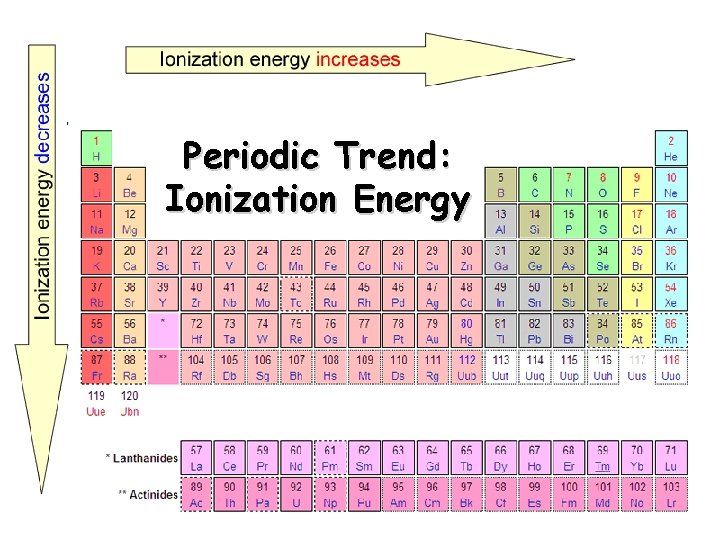
Ionization Energy Definition: the energy required to remove an electron from an atom q Increases for successive electrons taken from the same atom q. Tends to increase across a period q Electrons in the same quantum level do not shield as effectively as electrons in inner levels q Irregularities at half filled and filled sublevels due to extra repulsion of electrons paired in orbitals, making them easier to remove q Tends to decrease down a group q Outer electrons are farther from the nucleus, more shielded, so easier to remove

Table of 1 st Ionization Energies

Periodic Trend: Ionization Energy

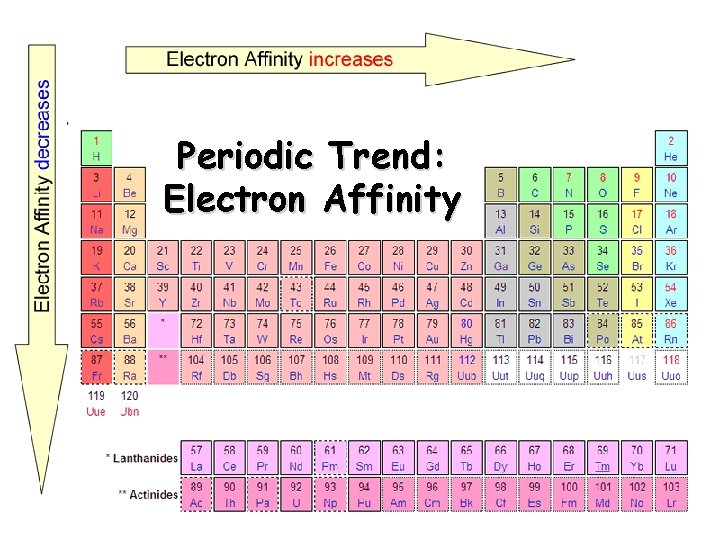
Electron Affinity Definition - the energy change associated with the addition of an electron q Affinity tends to increase across a period q Affinity tends to decrease as you go down in a group Electrons farther from the nucleus experience less nuclear attraction Some irregularities due to repulsive forces in the relatively small p orbitals

Periodic Trend: Electron Affinity

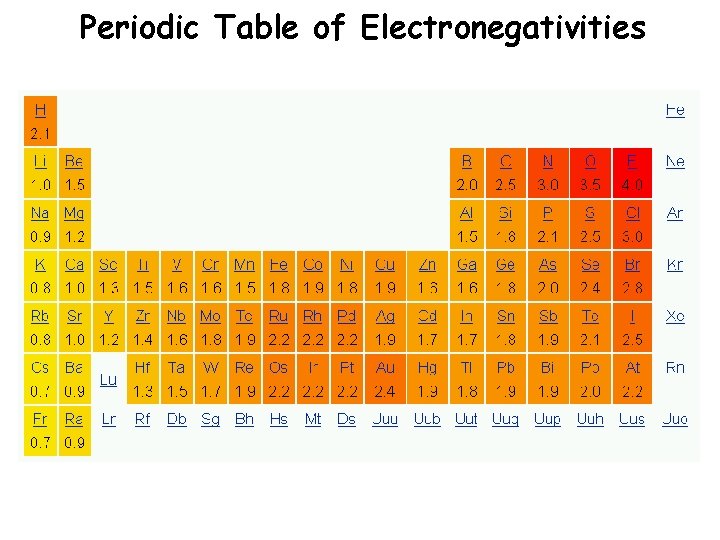
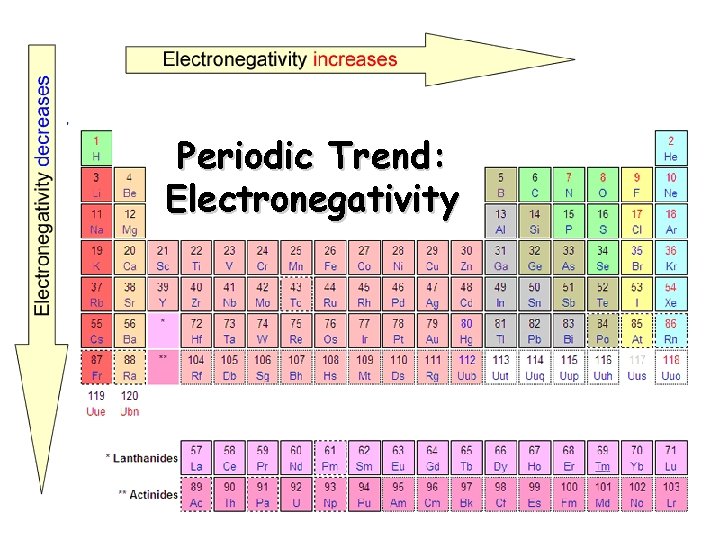
Electronegativity Definition: A measure of the ability of an atom in a chemical compound to attract electrons o Electronegativity tends to increase across a period o As radius decreases, electrons get closer to the bonding atom’s nucleus o Electronegativity tends to decrease down a group or remain the same o As radius increases, electrons are farther from the bonding atom’s nucleus

Periodic Table of Electronegativities

Periodic Trend: Electronegativity

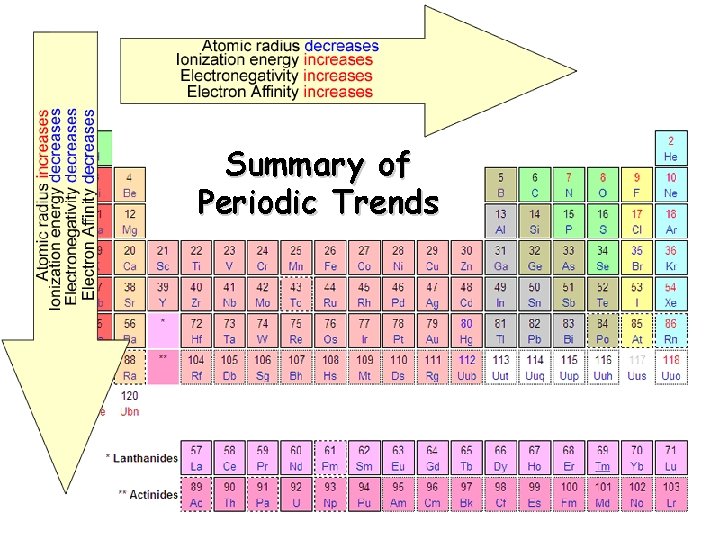
Summary of Periodic Trends

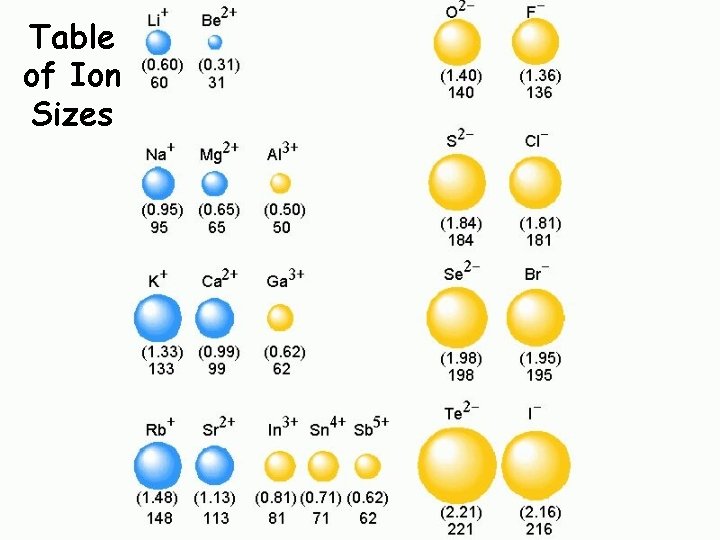
Ionic Radii Cations q Positively charged ions formed when an atom of a metal loses one or more electrons q Smaller than the corresponding atom q Negatively charged ions formed when nonmetallic atoms gain one Anions or more electrons q Larger than the corresponding atom

Table of Ion Sizes