Periodic Trends Atomic Radius Defined as half of












- Slides: 21

Periodic Trends

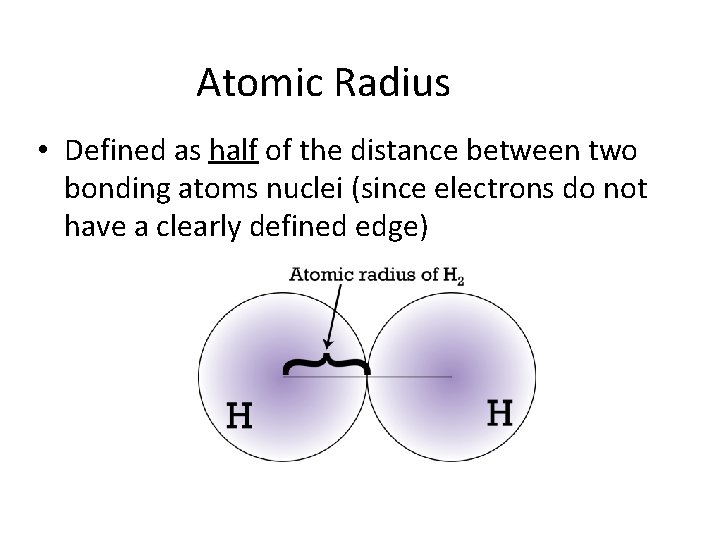
Atomic Radius • Defined as half of the distance between two bonding atoms nuclei (since electrons do not have a clearly defined edge)

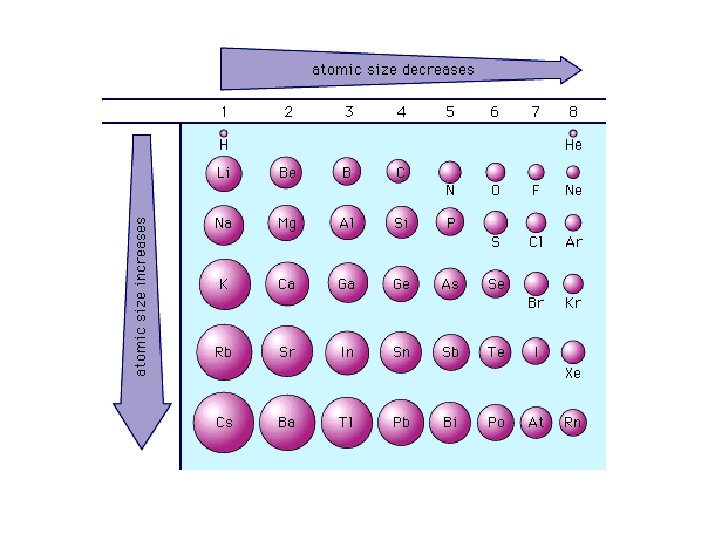
Atomic Radius Across a Period • Atomic radius generally decreases in size as you move left to right across the period – Increasing positive charge in the nucleus pulls the electrons of the same energy level in.

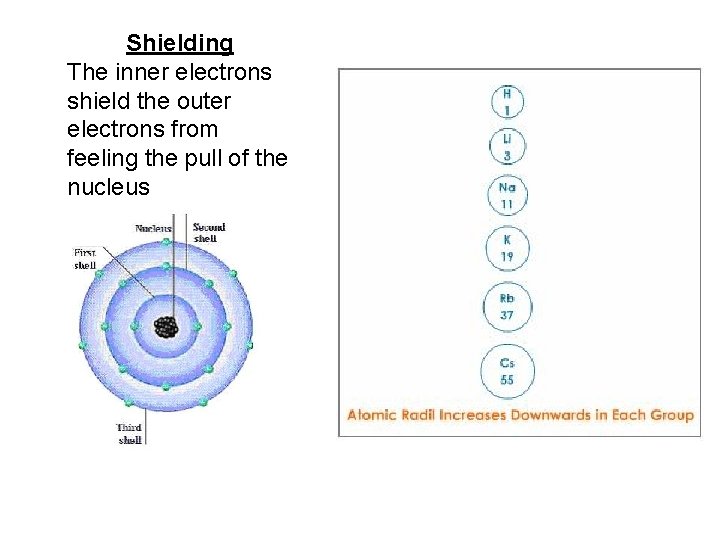
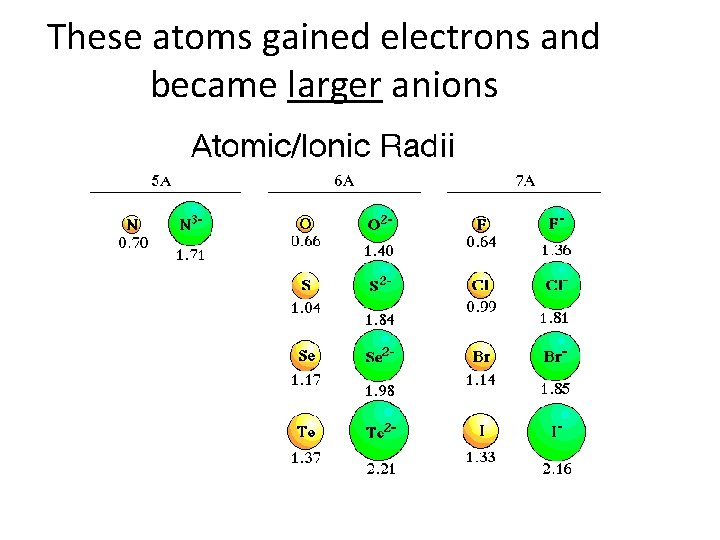
Atomic Radius Down a Group • Atomic radius increases as you move down a group – Orbital size increases as you move down a group with increasing energy level – Larger orbitals means that outer electrons are farther from the nucleus. This increased distance offsets the greater pull of the increased nuclear charge. – As additional orbitals between the nucleus and the outer electrons are occupied, the inner electrons shield the outer electrons from the pull of the nucleus this is called shielding.

Shielding The inner electrons shield the outer electrons from feeling the pull of the nucleus


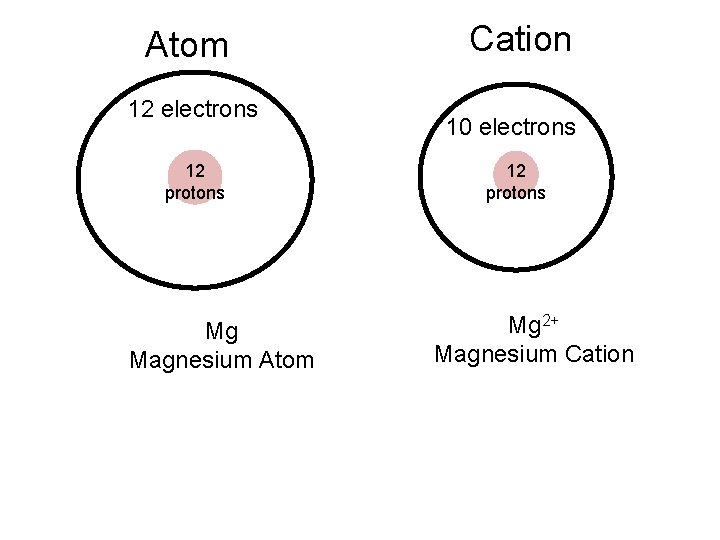
Cation and Anion • An ion is a positively or negatively charged atom that gains or loses an electron. • A cation loses electrons and produces a positive charge • An anion gains electrons and produces a negative charge

Ionic Radius - Cations • Groups 1 A, 2 A, 3 A, and other metals lose electrons and form cations. • When atoms lose electrons they become smaller – The electron lost will be a valence electron leaving a completely empty outer orbital – Protons in nucleus can pull fewer electrons tighter

Atom 12 electrons 12 protons Mg Magnesium Atom Cation 10 electrons 12 protons Mg 2+ Magnesium Cation

These atoms lost electrons and became smaller cations.

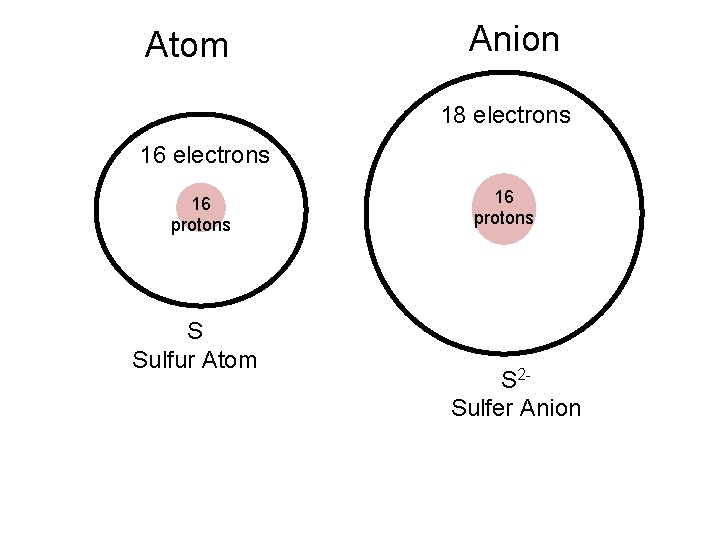
Ionic Radius - Anions • Group 5 A, 6 A, and 7 A tend to gain electrons and form anions • When atoms gain electrons and form negatively charged ions, they become larger. – Protons in nucleus have more electrons to pull and cannot pull in as tight

Atom Anion 18 electrons 16 protons S Sulfur Atom 16 protons S 2 Sulfer Anion

These atoms gained electrons and became larger anions

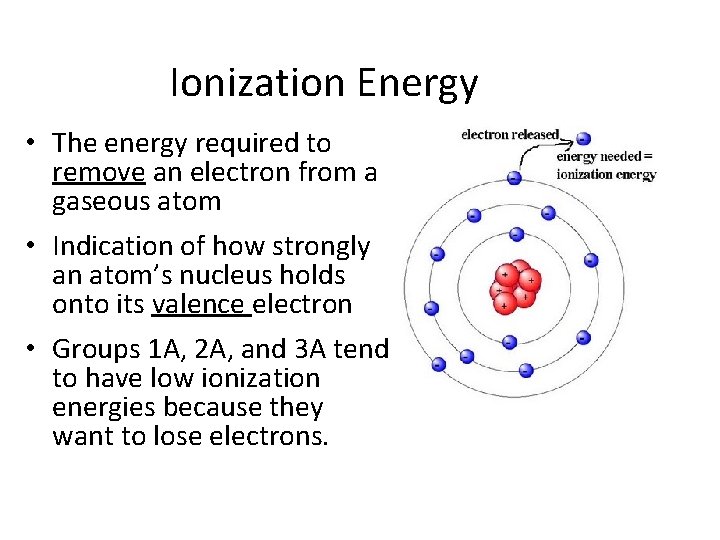
Ionization Energy • The energy required to remove an electron from a gaseous atom • Indication of how strongly an atom’s nucleus holds onto its valence electron • Groups 1 A, 2 A, and 3 A tend to have low ionization energies because they want to lose electrons.

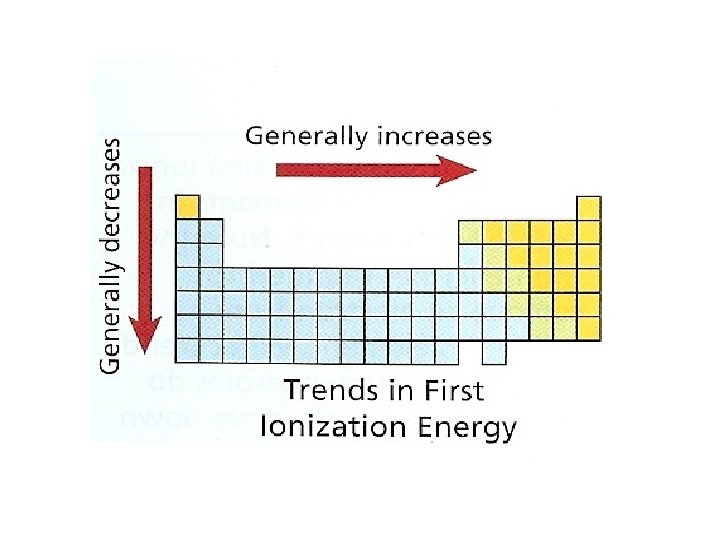
Ionization Energy Trends – Across a Period • Ionization energy generally increases as you move left to right – Across a period electrons are added to the same energy level (same distance away from the nucleus), yet the nuclear charge is increasing across a period increasing the attraction to the electrons.

Ionization Energy Trends – Down a Group • Ionization energy decreases as you move down a group – Down a group electrons are added to a higher energy level (farther distance away from the nucleus), making it easier to remove an electron


Octet Rule Sodium atom 1 s 22 p 63 s 1 Sodium ion 1 s 22 p 6 (Sodium atom lost 1 electron) Neon 1 s 22 p 6 Sodium ion has the same electron configuration as neon • Octet rule states that atoms gain, lose, or share electrons to acquire a full set of eight valence electrons (to be like a noble gas) • •

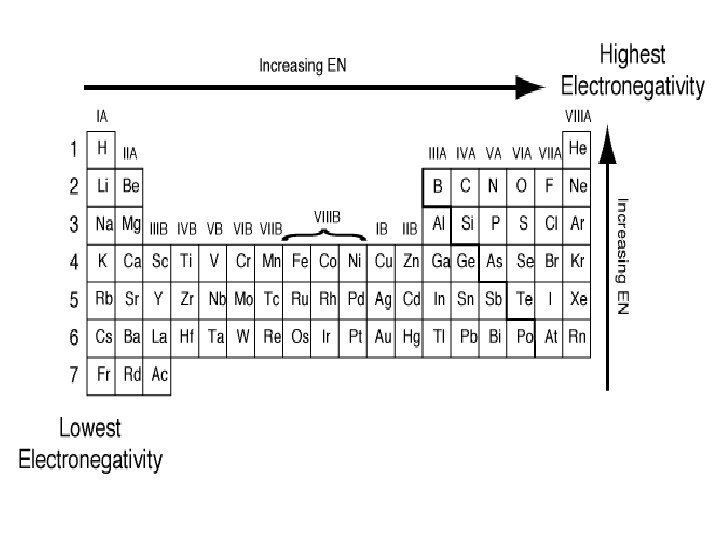
Electronegativity • Indicates an element’s ability to attract electrons in a shared chemical bond • fluorine (F) is the most electronegative element • cesium (Cs) and francium (Fr)are the least electronegative • Noble gases do not tend to have an electronegativity number since they tend not to form compounds

Trends with Electronegativity • Electronegativity increases as you move left-to -right across a period • Electronegativity decreases as you move down a group
