Periodic Trends Atomic Radii Ionization Energies Electronegativity I









- Slides: 10

Periodic Trends Atomic Radii Ionization Energies Electronegativity

I. Atomic Radius- used to estimate size of atom § half the distance between two nuclei of identical atoms that are bonded together. § Why?

Atomic Radii - Periodic Trends § Notice atomic radii increase as we go down a group. § Why? As we add more energy levels, size of atom increases § Notice they decrease as we go across a period from left to right even though we’re adding particles to the succeeding atoms. § Why? As we move across period, atomic # increases, so electrons in s and p sublevels are pulled closer to the more highly charged + nucleus -> smaller radius

Ionic radius § An ion is an atom or molecule that has gained or lost an electron. § A cation is a positive ion § How could an atom have a positive charge? § A negative ion is called an anion. § How could an atom have a negative charge? § Atoms – lose electrons , + ions, smaller radius § Atoms – gain electrons, -ions, larger radius § Trend? ? ?

Ionization Energy § energy required to remove an electron from an atom § Can you predict which elements would require the least energy to remove an electron and which would require the most?

Ionization energy § Period trend – increases as move across period, increased nuclear charge, higher +charge more strongly attracts electrons § Group trend – decreases as move down a group; electrons in higher energy levels are farther from the nucleus making them easier to remove

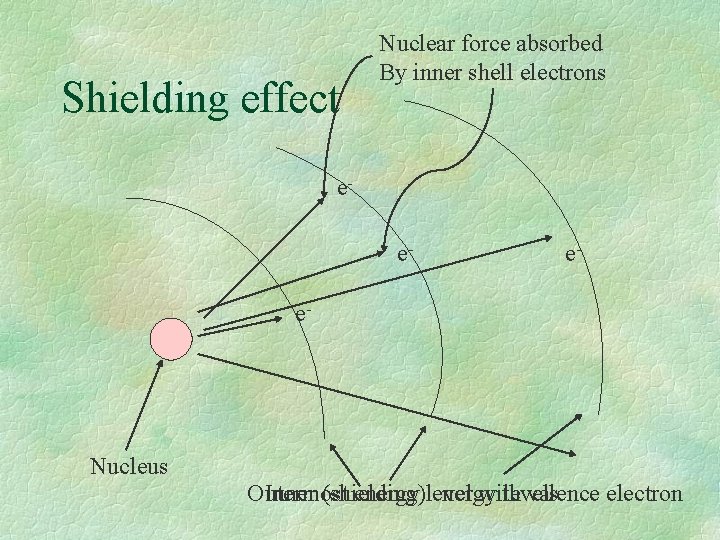
Ionization Energy § Factors affecting ionization energies include: § Nuclear charge - The larger the nuclear charge the greater the ionization energy. § Shielding effect - The greater the shielding effect, the less the ionization energy. § Radius - The greater the distance between the nucleus and the outer electrons of an atom, the less the ionization energy.

Shielding effect Nuclear force absorbed By inner shell electrons ee- e- e- Nucleus Outermost Inner (shielding) energy level energy with levels valence electron

Practice problems § Pg. 165 #’s 16 -18

Electronegativity § Electronegativity is the measure of an atom in a chemical compound to attract electrons. § Increases as move up a group and across a period from left to right