Periodic Trends All Arrows point to increases Electronegativity


































- Slides: 45

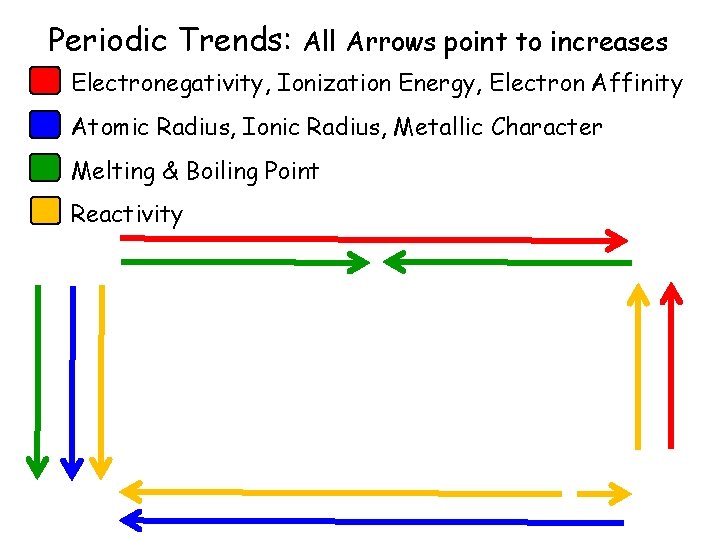
Periodic Trends: All Arrows point to increases Electronegativity, Ionization Energy, Electron Affinity Atomic Radius, Ionic Radius, Metallic Character Melting & Boiling Point Reactivity

Periodic Table Follow along in your text Chapter 4 Sections 1 & 2 Pages 116 - 131 I II III

Newlands z. John Newlands (1865, English) y. Organized elements by increasing atomic mass. y. Elements with similar properties were grouped together. y. Created Law of Octaves. x. Every 8 elements, there is a similarity.

Mendeleev z. Dmitri Mendeleev (1869, Russian) y. Used Newlands’ Law of Octaves to create the first Periodic Table. y. There were some discrepancies.

Mendeleev z. Predicted properties of undiscovered elements.

Moseley z. Henry Mosely (1913, British) y. Organized elements by increasing atomic number. y. Resolved discrepancies in Mendeleev’s arrangement.

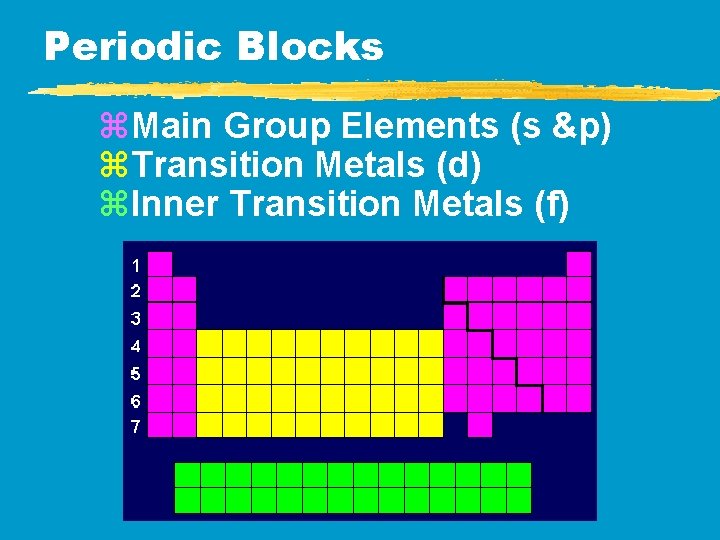
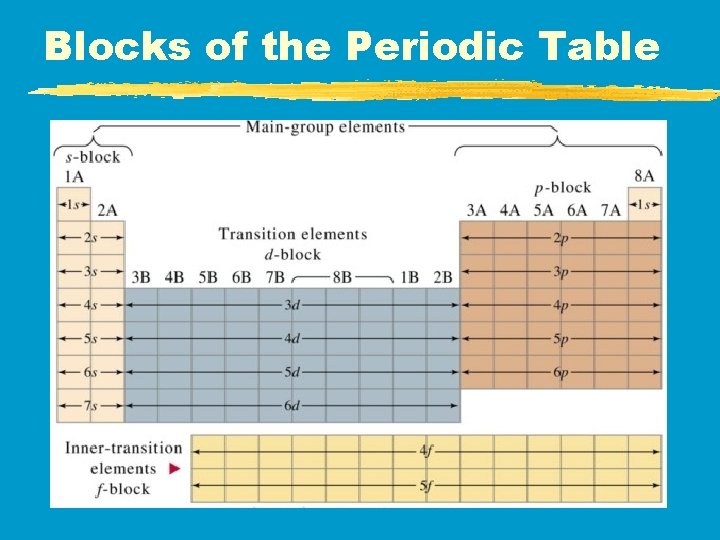
Periodic Blocks z. Main Group Elements (s &p) z. Transition Metals (d) z. Inner Transition Metals (f)

Blocks of the Periodic Table

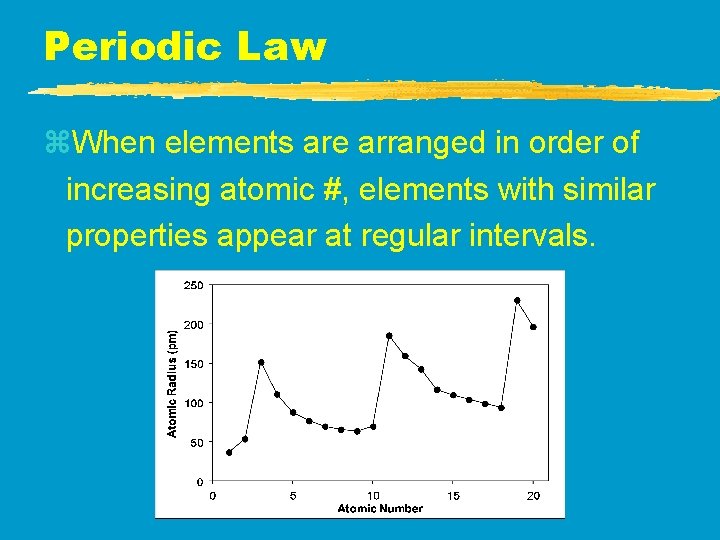
Periodic Law z. When elements are arranged in order of increasing atomic #, elements with similar properties appear at regular intervals.

Let’s Get Organized! z. Vertical columns are called “groups” or families z. Same group = same number of valence electrons

Let’s Get Organized!(cont. ) z. Horizontal rows are called “periods” z. Elements in each row have the same amount of principal energy levels occupied

Main-Group Elements z. Are in s & p blocks z. Silicon & oxygen account for 4 of every 5 atoms found on or near Earth’s surface z 4 groups have special names

Group 1: Alkali Metals z. React with water to make alkaline solutions (reactive) z. Never in nature as lone elements but are in compounds z 1 valence electron z. Li, Na, K, Ru, Cs, Fr

Group 2: Alkaline-Earth Metals z. Hardly in nature as lone elements but are usually in compounds (highly reactive but not as much as Group 1) z 2 valence electrons z. Be, Mg, Ca, Sr, Ba, Ra

Groups 3 - 12: Transition Metals z. Called d-block elements because in the center of the table z. Do not always have the same outer configurations but their outer s & d orbitals will have the total amount of electrons as their group number z. Can 1, 2, or 3 valence e- depending on the element with which it reacts

Group 17: Halogens z. Most reactive nonmetals z 7 valence electrons z. F, Cl, Br, I, At

Group 18: Noble Gases z. Once call inert gases because once thought to be nonreactive z. Full set of electrons in outermost energy level z. Except He, all have config of ns 2 np 6 z. He, Ne, Ar, Kr, Xe, Rn

Lanthanides & Actinides (La # 57 - 70 & Ac #89 -102) z. Called the f-block elements z. Lanthanides are similar in reactivity to alkaline-earth metals z. Actinides are all radioactive due to their unique nuclear structure

Lonely Hydrogen z. Most common element in universe (3 of 4 atoms) z. Only 1 electron so reacts with just about anything

Metals z. Most elements are metal z. Excellent conductors of electricity & heat z. Some are brittle; others are ductile (can be made into wire) & malleable (can be hammered into sheets) z. Can be mixed with other elements to form alloys (mixtures that improve disadvantages of metals)

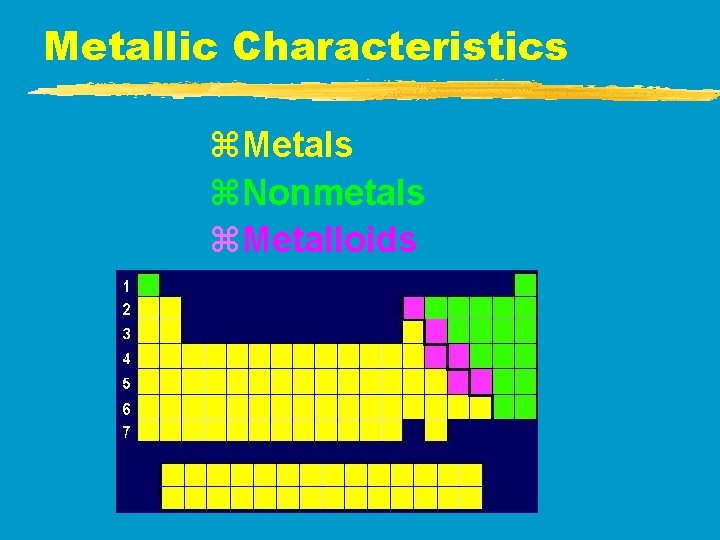
Metallic Characteristics z. Metals z. Nonmetals z. Metalloids

Chemical Reactivity z. Families y. Similar valence e- within a group result in similar chemical properties

Chemical Reactivity z. Alkali Metals z. Alkaline Earth Metals z. Transition Metals z. Halogens z. Noble Gases

Periodic Properties z. Atomic Radius ysize of atom y. Measured by half of bond distance © 1998 LOGAL z. First Ionization Energy y. Energy required to remove one efrom a neutral atom. z. Melting/Boiling Point © 1998 LOGAL

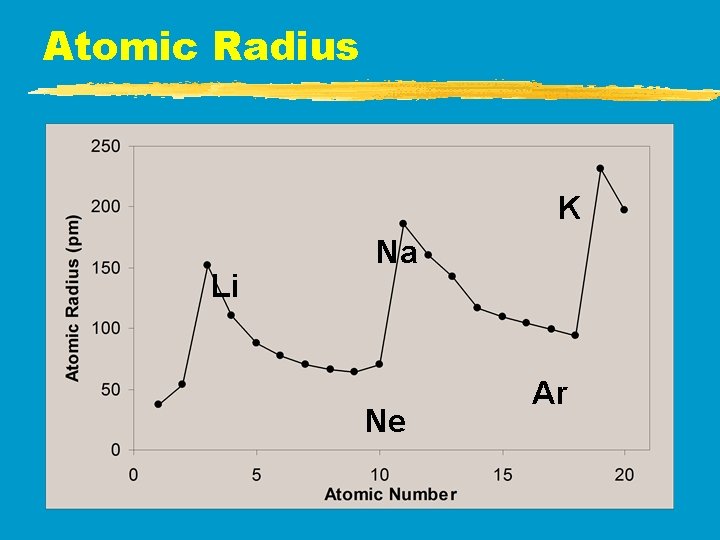
Atomic Radius z. Atomic Radius y. Increases to the LEFT and DOWN

Atomic Radius K Li Na Ne Ar

Atomic Radius z. Why radius larger going down? y. Higher energy levels have larger orbitals y. Shielding - core e- block the attraction between the nucleus and the valence ez. Why smaller to the right? y. Increased nuclear charge without additional shielding pulls e- in tighter

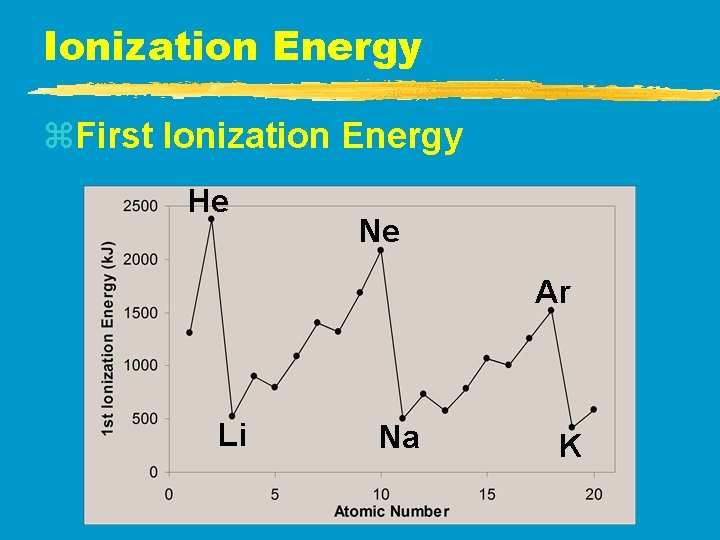
Ionization Energy z. First Ionization Energy y. Increases UP and to the RIGHT

Ionization Energy z. First Ionization Energy He Ne Ar Li Na K

Ionization Energy z. Why opposite of atomic radius? y. In small atoms, e- are close to the nucleus where the attraction is stronger z. Why small jumps within each group? y. Stable e- configurations don’t want to lose e-

Melting/Boiling Point z. Melting/Boiling Point y. Highest in the middle of a period.

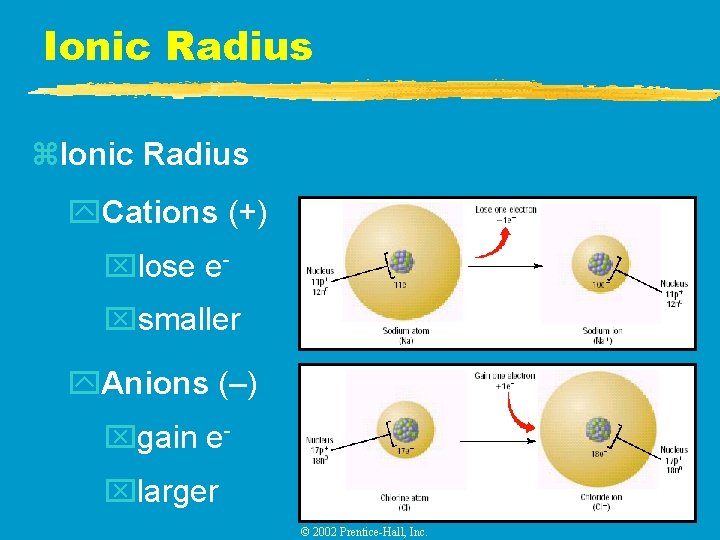
Ionic Radius z. Ionic Radius y. Cations (+) xlose exsmaller y. Anions (–) xgain exlarger © 2002 Prentice-Hall, Inc.

Ionic Radius z. Increases DOWN and to the LEFT for each type of ion

Examples z. Which atom has the larger radius? y. Be or Ba Ba y. Ca or Br Ca

Examples z. Which atom has the higher 1 st Ionization Energy? y. N or Bi N y. Ba or Ne Ne

Examples z. Which atom has the higher melting/boiling point? y. Li or C C y. Cr or Kr Cr

Examples z. Which particle has the larger radius? y. S or S 2 - y. Al or Al 3+ Al

Electronegativity z. Numerical value of how much an atom in a molecule attracts electrons z. Scale created by Linus Pauling z. Flourine is 4. 0 & other elements are calculated relative to this value

Electronegativity z. Increases UP and to the RIGHT y. Same as Ionization Energy

Electron Affinity z. The energy change that occurs when a neutral atom gains an electron z. This is also due to increasing nuclear charge z. Literally means “love of electrons”

Electron Affinity z. Increases UP and to the RIGHT y. Same as Ionization Energy & Electronegativity

Examples z. Which atom is most electronegative? y. N or Bi N y. Ba or Rb Ba

Examples z. Which atom has the highest electron affinity? y. Be or Ba Be y. Ca or Br Br

Examples z. Which atom is more metallic? y. S or Fr Fr y. Al or Ge Ge

Summary of Trends z. Ionization Energy, Electronegativity, Electron Affinity all Increase UP & RIGHT z. Atomic Radius & Ionic Radius all Increase DOWN & LEFT z. Boiling/Melting point Increases then Decreases across blocks z. Metals are to the left of the “stairs” & non-metals are to the right