Periodic Table Why Periodic The properties of the

Periodic Table ® Why Periodic? ? The properties of the elements repeat in in a periodic way. ® Invaluable tool for chemistry ® Used for organization ®

The Basics ® Elements are arranged by atomic number ® Typical box contains: ® Name of the element ® Symbol ® Atomic number ® Atomic mass
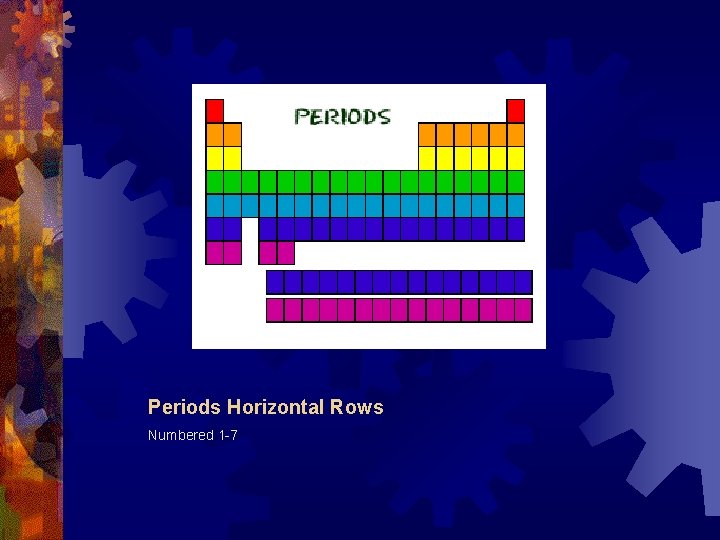
Periods Horizontal Rows Numbered 1 -7
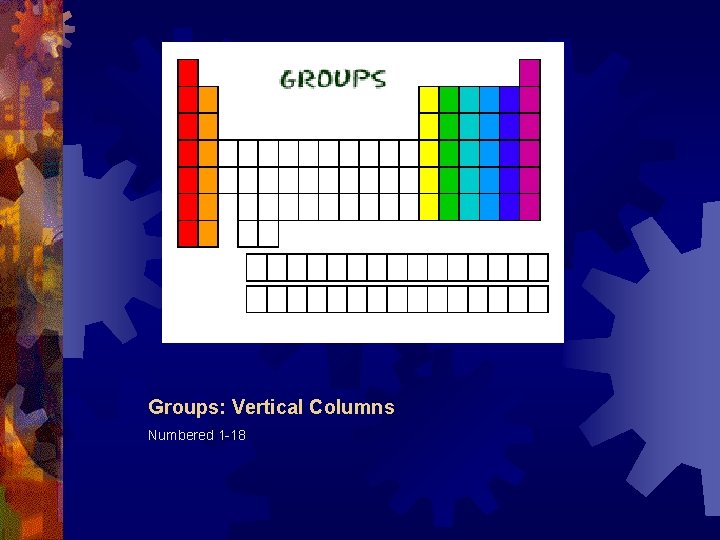
Groups: Vertical Columns Numbered 1 -18

Interactive Periodic Tables ® http: //periodic. lanl. gov/index. shtml ® www. webelements. com ® www. chemicool. com ® http: //education. jlab. org/itselemental/ele 016. html

The Families of Elements http: //www. privatehand. com/flash/elements. html
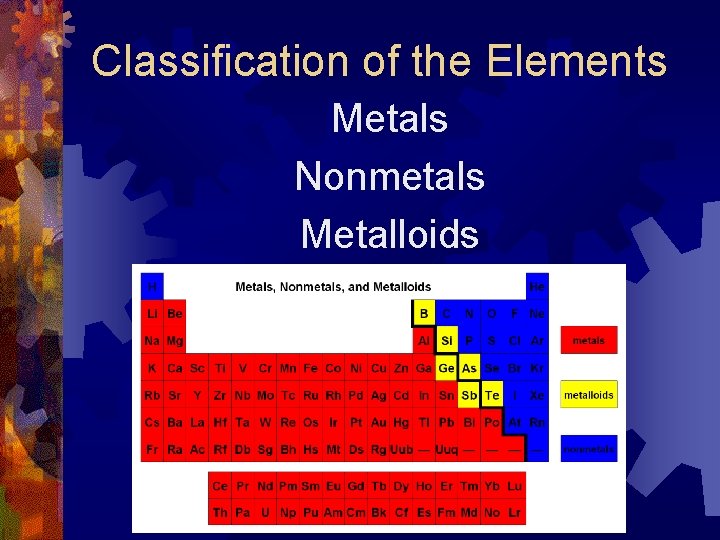
Classification of the Elements Metals Nonmetals Metalloids

Metals • Occupy the left side of the periodic table • Have luster, shiny • Solids at room temp except Hg • Ductile: ability to be drawn into wires • Malleable: ability to be hammered into sheets • Excellent conductors of heat and electricity • Tend to form positive ions

Non. Metals ® Occupy the right side of the Periodic Table ® Generally gases or brittle solids ® Dull-looking ® Brittle ® Poor conductors of heat and electricity ® Bromine is the only liquid at room temp ® Tend to form negative ions

Metalloids Characteristics of metals and nonmetals

Classification of the Elements ® Families of elements share the same ending electron configuration ® therefore they share similar chemical characteristics ® Valence Electrons: electrons in the highest principal energy level Determine Chemical reactivity ® Elements in a group share the same number of valence electrons ®
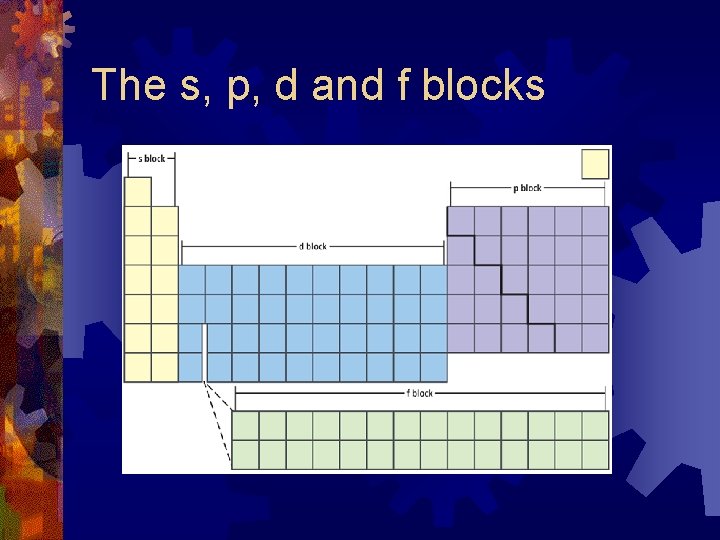
The s, p, d and f blocks

Number of Valence Electrons Elements on the right • Nonmetals • 4 or more valence electron • tend to gain electrons • become negative ions Elements on the left • Metals • 3 or less valence electrons • tend to lose valence electrons • form positive ions
Families of elements ® Elements of the same family (group) share structural and chemical (behavioral) characteristics ® Alkali Metals ® Alkaline Earth Metals ® Transition Elements ® Halogens ® Nobel Gases
Group 1: Alkali Metals ® Soft, highly reactive metals ® Usually stored under oil or kerosene to prevent their interaction with air and water

Properties of Alkali Metals ® React vigorously with water ® Oxidize readily in air ® Good conductors of electricity
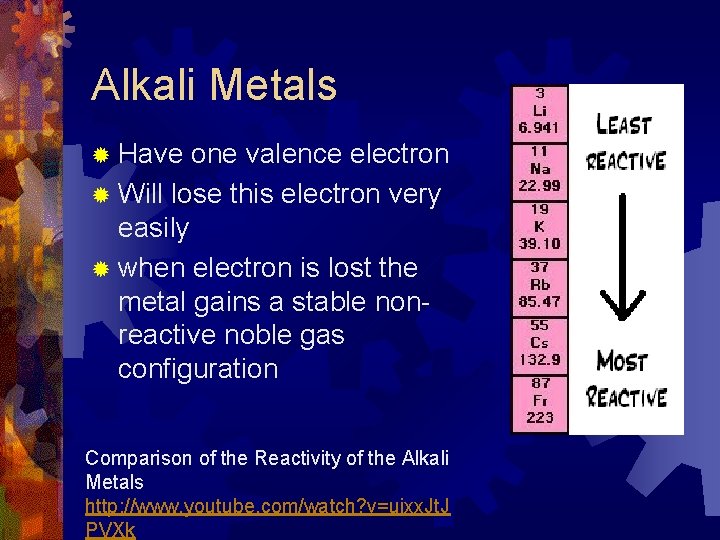
Alkali Metals ® Have one valence electron ® Will lose this electron very easily ® when electron is lost the metal gains a stable nonreactive noble gas configuration Comparison of the Reactivity of the Alkali Metals http: //www. youtube. com/watch? v=uixx. Jt. J PVXk

Group 2: Alkaline Earth Metals ® Harder, denser, stronger, and have higher melting points than alkali metals ® All are reactive not as reactive as group 1

Alkaline Earth Metals ® Must lose two electrons to gain a stable configuration

Groups 3 -12: Transition Metals ® Not as reactive as Groups 1 and 2 ® Huge variety but all shiny ® Multi valent…form multiple ions ® d-block elements ® Also include: Inner Transition Elements (Rare Earth Elements) Elements 58 -71 Lanthanides ® Elements 90 -103 Actinides ®

Group 17: Halogens ® Most reactive non-metals ® Combine easily with metals; especially the alkali metals

Halogens ® 7 valence electrons, one short of a stable octet. ® Will gain one electron to become stable ® -1 ions Reaction of chlorine (a halogen) with sodium (an alkali metal) https: //www. youtube. com/watch? v=1 x. T 4 OFS 03 j. E

Element Dating

Hydrogen ® Most common element in the universe ® Chemical family by itself because it behaves so differently ® Reacts with most other elements ® Rarely found in a free state in nature ® 1 valence electron

The Hindenberg ® Filled with H ® Very reactive with oxygen gas ® He used in blimps today ® much less reactive than H

Group 18: Noble Gases ® Very low reactivity ® Filled valence shells: s and p levels in the highest principal energy levels are full ® Very stable electron configuration ® Many uses: signs, weather balloons and the airships (Blimps)

The Octet Rule ® Atoms tend to gain, lose or share electrons in order to acquire a full set of eight valence electrons. ® Elements on the left (metals) tend to lose valence electrons and form positive ions ® Elements on the right (nonmetals) tend to gain electrons to become negative ions


Periodic Trends ® Properties of Elements tend to occur in a predictable way ® Known as a trend, as you move across a period or down a group ® Knowing element trends allows us to make predictions about an element’s behavior

Periodic Properties ® Properties Atomic Radius ® Ionic Radius ® Electronegativity ® Ionization Energy ® ® Questions we will answer: Definition ® How does the property vary across the table? ® Why? ® How does it vary down a group? ® Why? ®
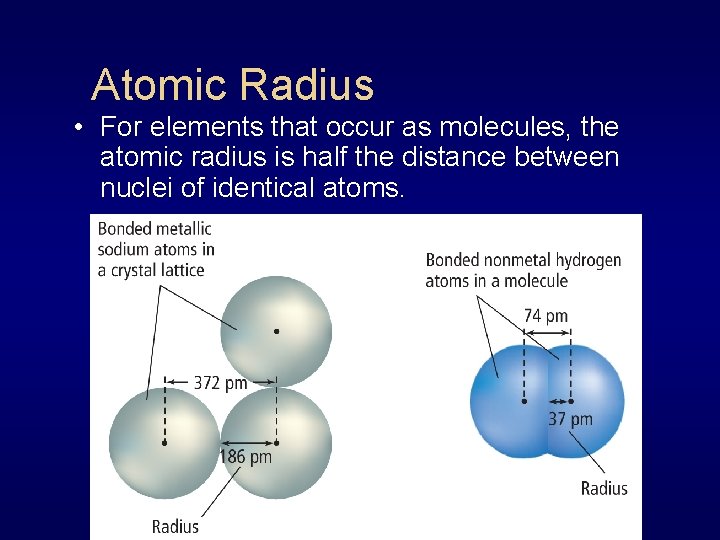
Atomic Radius • For elements that occur as molecules, the atomic radius is half the distance between nuclei of identical atoms.

Atomic Radius ® The atomic radius is a measure of the size of an atom. ® The larger the radius, the larger is the atom.

Trends in Atomic Radius ® There is a general decrease in atomic radius from left to right, caused by increasing positive charge in the nucleus. ® Valence electrons are not shielded from the increasing nuclear charge because no additional electrons come between the nucleus and the valence electrons.

Trends in Atomic Radius ® The atomic radius decreases as you move across a period ® Why? ® Increased nuclear charge pulls the electrons in tighter ® Added electrons are in the same principal energy levels

Group Trends in Atomic Radius ® Atomic Radius increases as you move down a group ® Why? The increasing number of electrons are in higher energy levels and instead of pulling the electrons closer to the nucleus we see the …
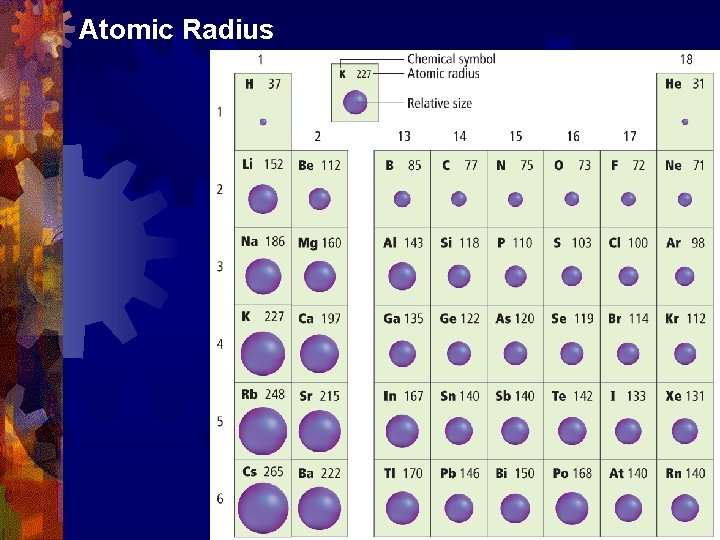
Atomic Radius
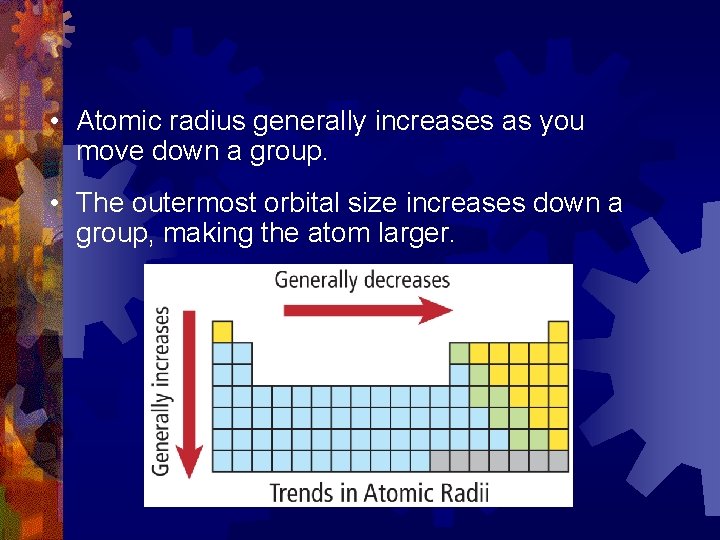
• Atomic radius generally increases as you move down a group. • The outermost orbital size increases down a group, making the atom larger.

Shielding Effect ® More inner electrons shield the outer electron from the nucleus and reduce their attraction to the nucleus therefore the overall atomic radius is larger

® For any element (A) the process of removing an electron can be represented as follows: ® A + energy -----> A+ + e® What is the periodic trend in ionization energy? Why?

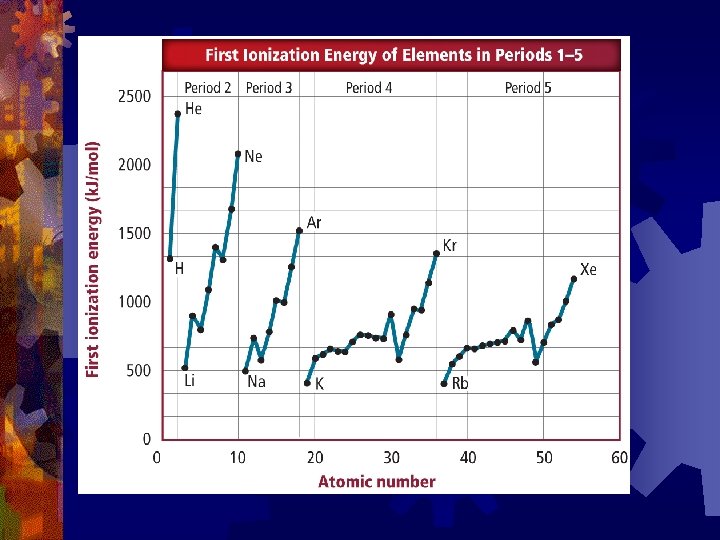
Trends for Ionization Energy ® Generally increases as you move across a period ® because increased nuclear charge causes an increased hold on the electrons ® Ionization Energy decreases as you move down a group ® due to increasing atomic size

Successive Ionization Energies ® There is an ionization energy for each electron that is removed from an atom ® After the valence electrons are removed Ionization Energies Jump Dramatically First Ionization Energy: removes 1 electron ® Second Ionization Energy: removes a second electron ® Third Ionization Energy: removes a third electron ®
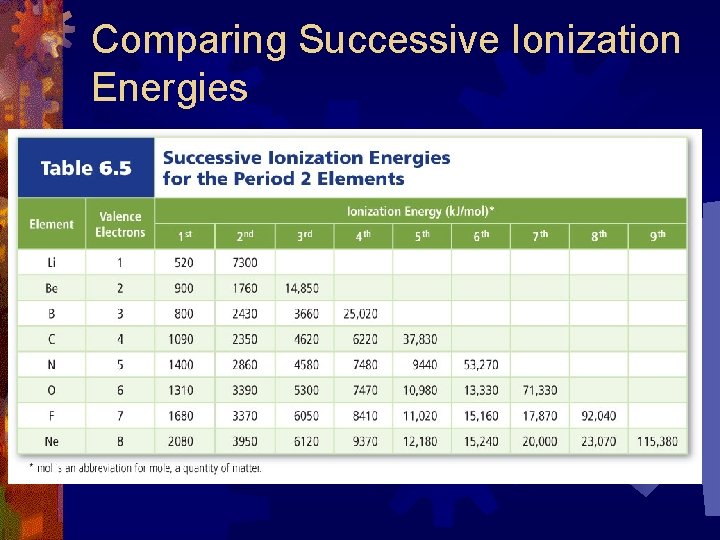
Comparing Successive Ionization Energies
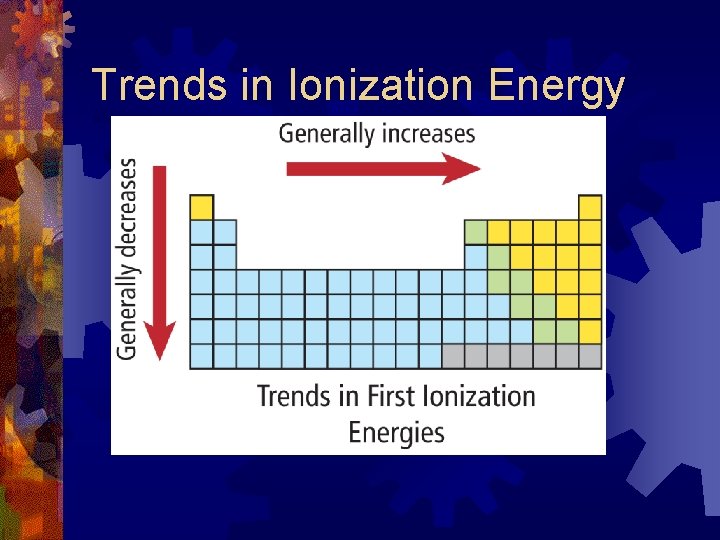
Trends in Ionization Energy

Electronegativity ® The ability of an an atom to attract electrons to itself when it is combined with another atom ® Expressed in terms of a relative scale: fluorine is assigned a value of 4 and all other elements are calculated relative to this. ® The units of electronegativity are arbitrary units called Paulings. ® Noble gases have no values because of few chemical compounds

Electronegativity ® Greater ® the electronegativity the higher an atom’s ability to pull an electron to itself when it is bonded to another atom ® What are the periodic trends in electronegativity? ® Why?

Trends in Electronegativity ® Electronegativity Increases as you move ® Electronegativity decreases you move down a across a period group Where are the elements with highest electronegativity? Where are the elements with lowest electronegativity?

Electronegativity
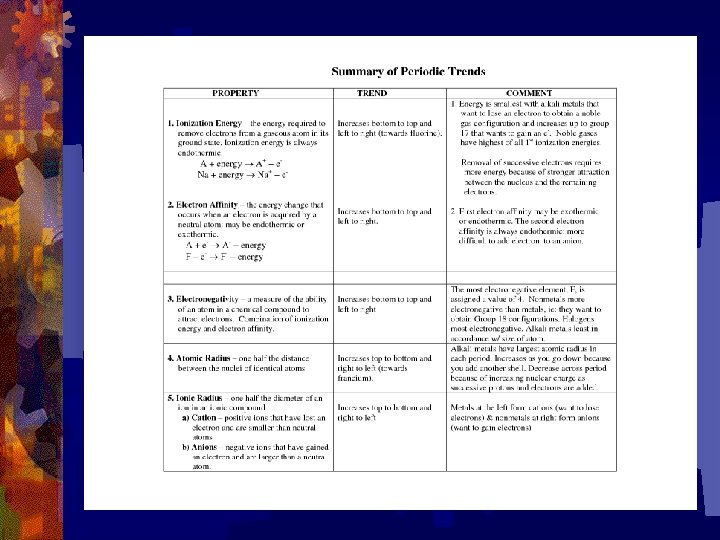
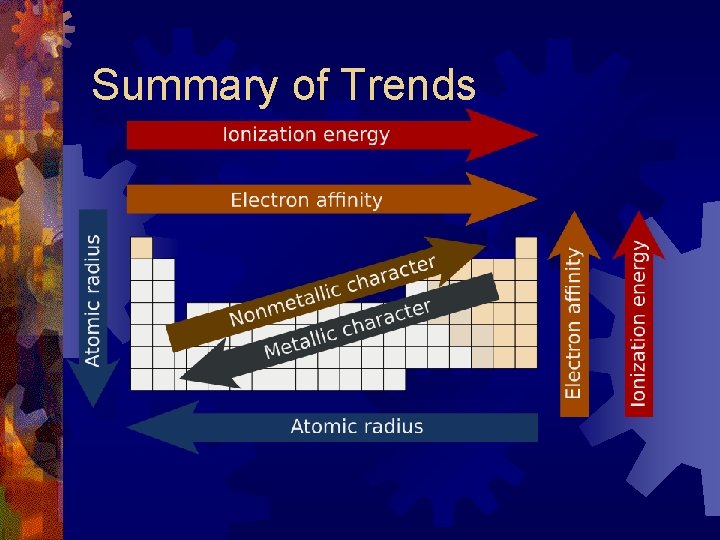
Summary of Trends
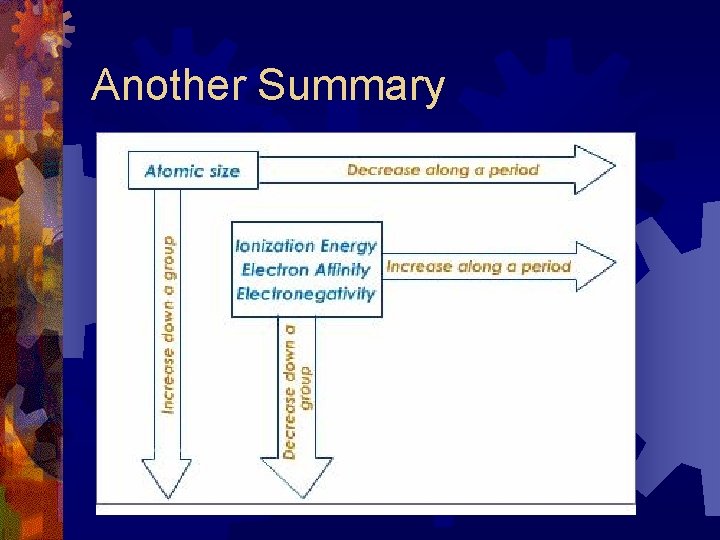
Another Summary
- Slides: 51