PERIODIC TABLE TRENDS MENDELEEV MOSELEY Dmitri Mendeleev a

PERIODIC TABLE TRENDS

MENDELEEV & MOSELEY • Dmitri Mendeleev , a Russian chemist, noticed that when the elements were arranged by atomic mass, there was a periodic pattern in properties – He left blank spaces open for new elements to be discovered – He was one of the first to arrange the elements in an organized table • His arrangement had its faults though…
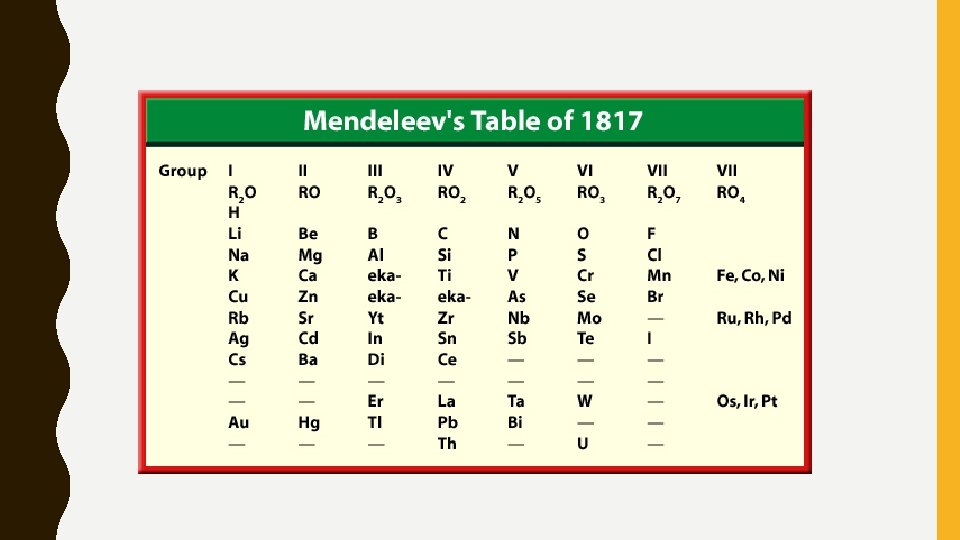

• When new elements were discovered, it became apparent that they didn’t fit into Mendeleev’s table properly – When arranged by mass, the properties did not correspond • Moseley (1913) – Arranged periodic table by atomic number – Elements close to one another have similar properties
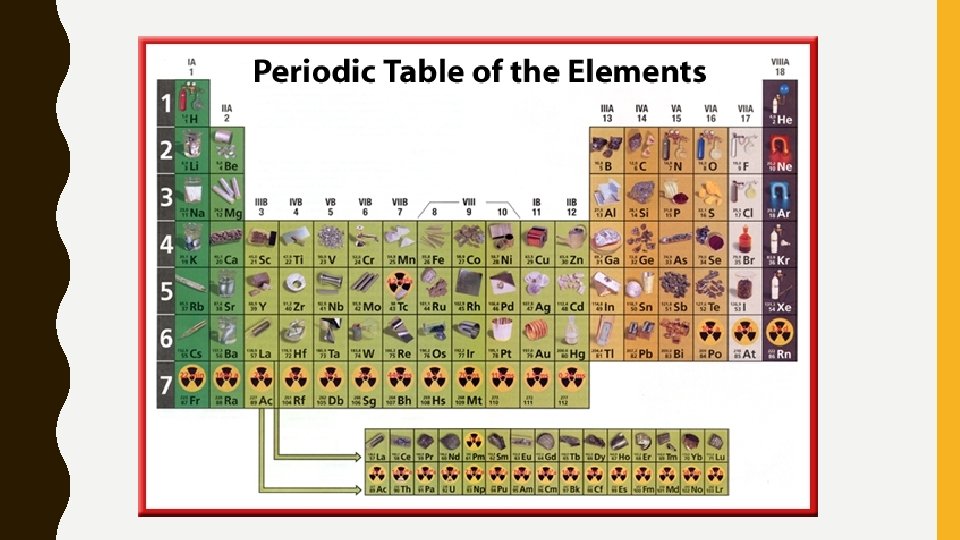

PERIODIC LAW • Periodic Law: there is a periodic repetition of chemical and physical properties of the elements when they are arranged by increasing atomic number
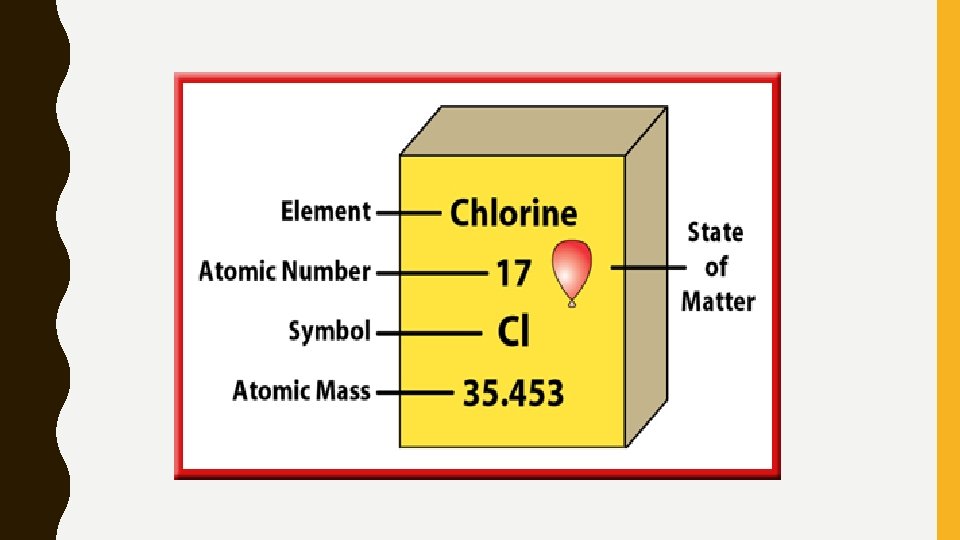

HOW TO READ THOSE NUMBERS… • Atomic Mass: Approx. the mass of the protons in the element – 1 atomic mass unit (amu)= mass of 1 Carbon 12 atom • Atomic Number: # of protons in the nucleus – The atomic number increases by 1 as you go across a row – Also the number of electrons in the element if it is neutral

WHAT IS IN AN ELEMENT/ATOM? • Protons: Positive Charge (+) • Neutrons: No Charge • Electrons: Negative Charge (-) • Protons +Neutrons make up a nucleus • Electrons circle around the nucleus in orbitals • In general, #protons=#electrons (neutral) • Also #neutrons= atomic mass-atomic number – If they differ in neutrons, it is called an isotope • C 12, C 13, C 14


SO… • If elements are ordered in the periodic table by atomic number, then they are also ordered according to the number of electrons they have. • The lineup starts with hydrogen, which has one electron. • Helium comes next in the first horizontal row because helium has two electrons. Lithium has three.

VALENCE ELECTRONS • In an atom there are multiple orbitals (think planets) • Valence electrons: electrons in the outer most orbital…these determine the chemical properties of the element – These are also the “bonding” electrons (remember, core electrons don’t bond)

BACK TO THE PERIODIC TABLE • Period: Horizontal Row (think sentence) – Numbered from top to bottom • Group/Family: Column/Vertical – Numbered from left to right
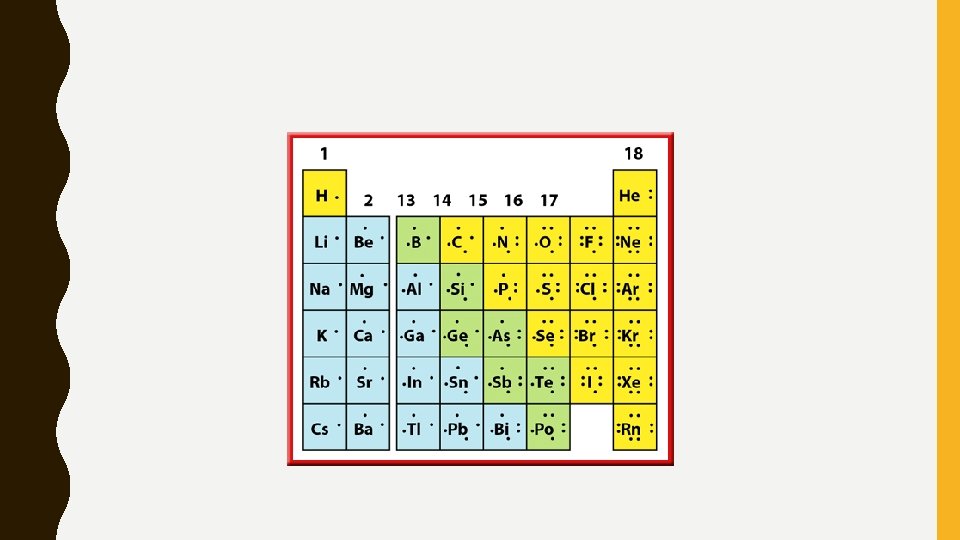

PROXIMITY MATTERS • Elements in the same group have characteristics similar to each other, yet different from the other elements in the periodic table. – Similarities occur because elements in the same group have the same number of valence electrons

NAMES & ORGANIZATION • Periodic table is divided into different groups – Think about the different colors we colored…

• Groups 1 A – Alkali metals – – Very reactive Explosive reactions with water One valence electron All metals • Groups 2 A – Alkaline Earth Metals – Quite reactive – Two valence electrons – All metals

• Groups 3 A – Boron Family – Varies from semi-metallic to non-metallic in properties – Three valence electrons • Group 4 A – Carbon Family – Varies from non metallic to metallic in properties – Four valence electrons

• Group 5 A – Nitrogen Family – Varies from non-metallic to metallic properties – Five valence electrons • Groups 6 A – Oxygen Family – Mostly non-metallic – Six valence electrons • Group 7 A – Halogens – All are non-metallic and make colorful gases – Seven valence electrons

• Group 8 A – Noble gases – All non metals and highly unreactive – Eight valence electrons (OCTET) • Transition metals – All have metallic properties – Valence number varies • Lanthanoids & Actinoids – Radioactive, many are non-natural or man-made – Valence number varies

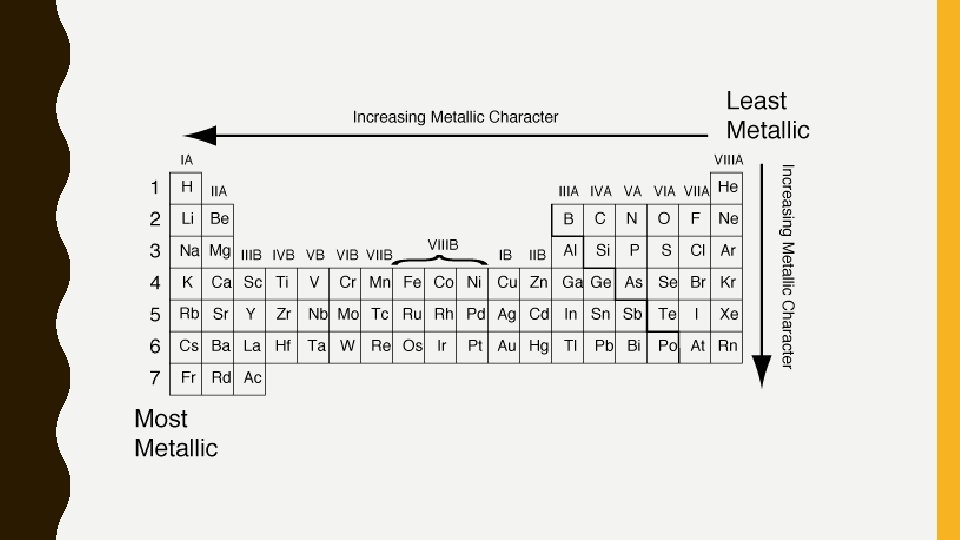
METALLIC CHARACTER Metallic Character – Element on the left side of the periodic table are metals, while those on the right side are non metals. – Elements that touch the dividing line between metals and nonmetals are called metalloids – Metallic character increases as you move down a column and across a row to the left
REACTIVITY Reactivity – Reactivity for metallic element increases as you move down a column and across a row to the left • The most reactive metal is Francium (bottom, left corner) – Reactivity for nonmetal increases as you move up a column and across a row to the right • The most reactive nonmetal is Fluorine (top, right corner) • WE DO NOT EVEN LOOK AT NOB GASES!!!
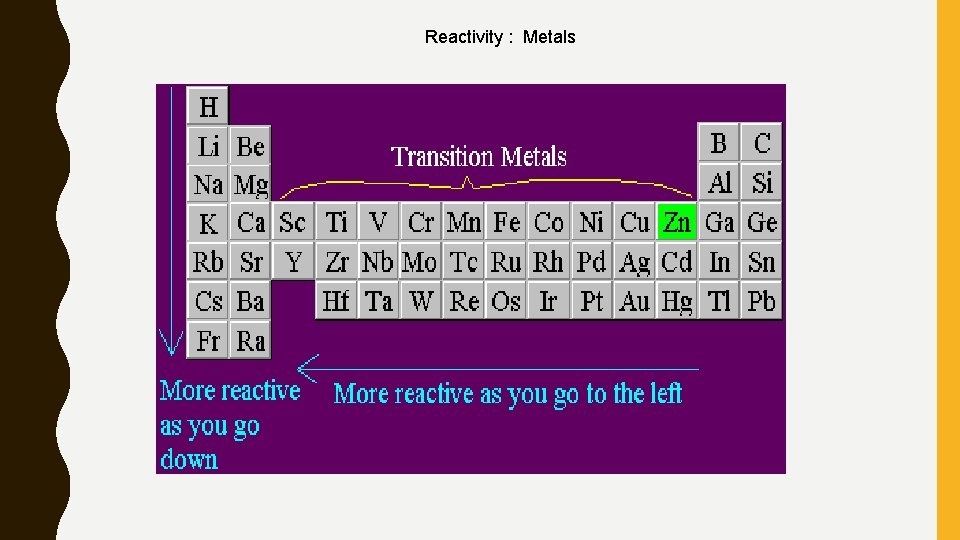
Reactivity : Metals
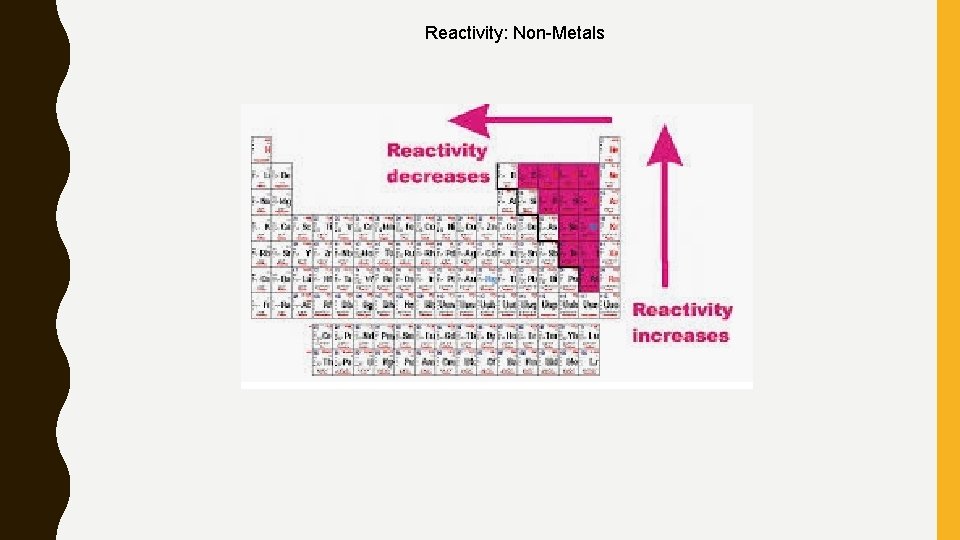
Reactivity: Non-Metals

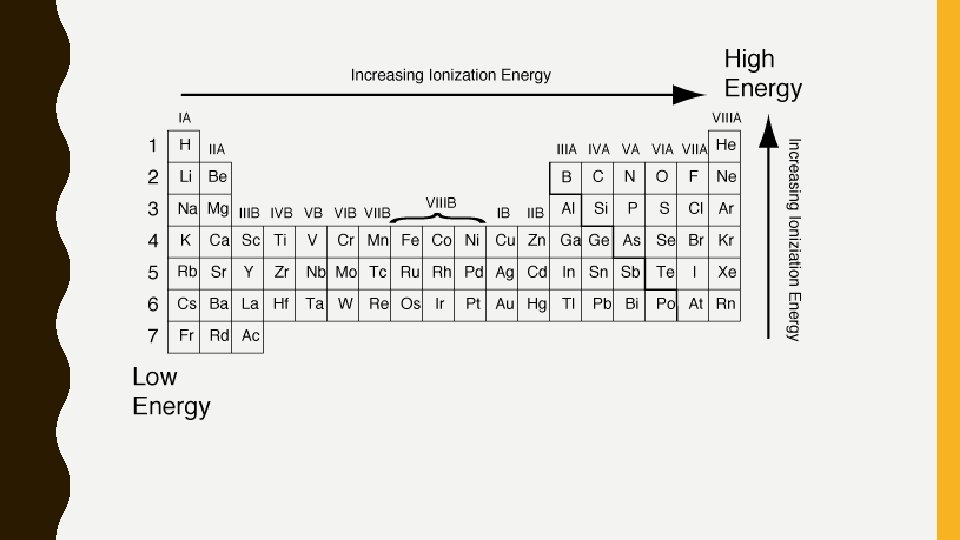
IONIZATION ENERGY • Ionization Energy 1. The energy needed to remove the outermost electron from an element (increases dramatically with successive removals) 2. Ionization energy depends on the number of valence electrons and the size of the atom • Elements with nearly full valence shells will have higher ionization energy than those with nearly empty valence shells • Larger elements will have a lower ionization energy than smaller elements

• Ionization energy increases as you move up a column and across a row to the right.

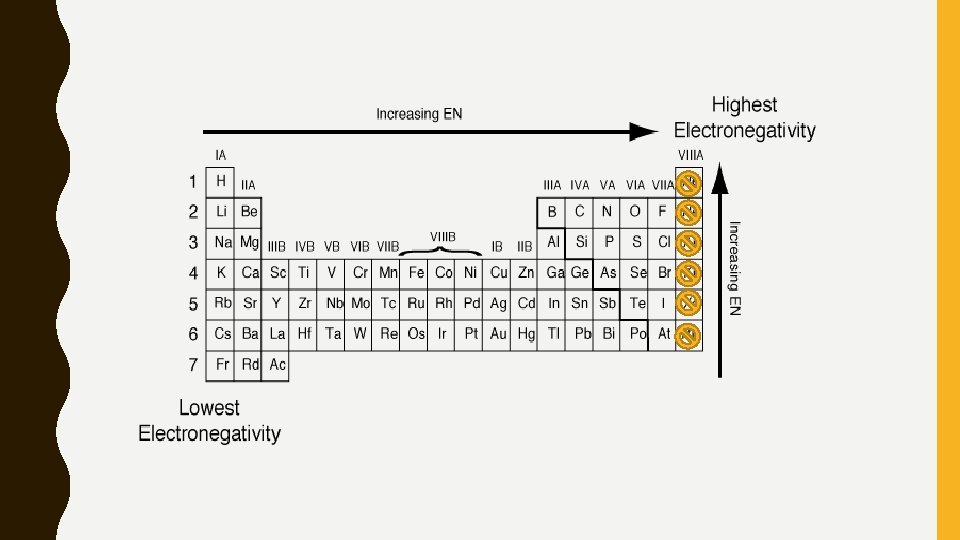
ELECTRONEGATIVITY/ELECTRON AFFINITY • Electronegativity (similar trend to ionization) – Ability of an element to attract shared electrons to itself – Depends on the number of valence electrons and the size of the element – Increases as you move up a column and across a row to the right

ATOMIC RADIUS Atomic Radius – Close approximation of the relative size of an atom – Determined by finding the distance from the center of an atom nucleus to it’s outermost electron – Increases as you move down a column • Each step down in a column increases the number of orbitals (electron shells) – Increases as you move across a row to the left • Decreasing the number of protons in the nucleus causes the electrons to be held more loosely


HOW SIZE CHANGES… Ionic Size – Size of an atom when electrons are added or removed – Electrons removed atom become smaller (Cations + charges) – Electrons added atoms become larger (Anions – charges)

WHY THESE TRENDS? • Effective Core Charge (increases by 1 as you go right across periodic table) • Electron shielding (the more electrons, the more repulsion from each of those electrons)
- Slides: 35