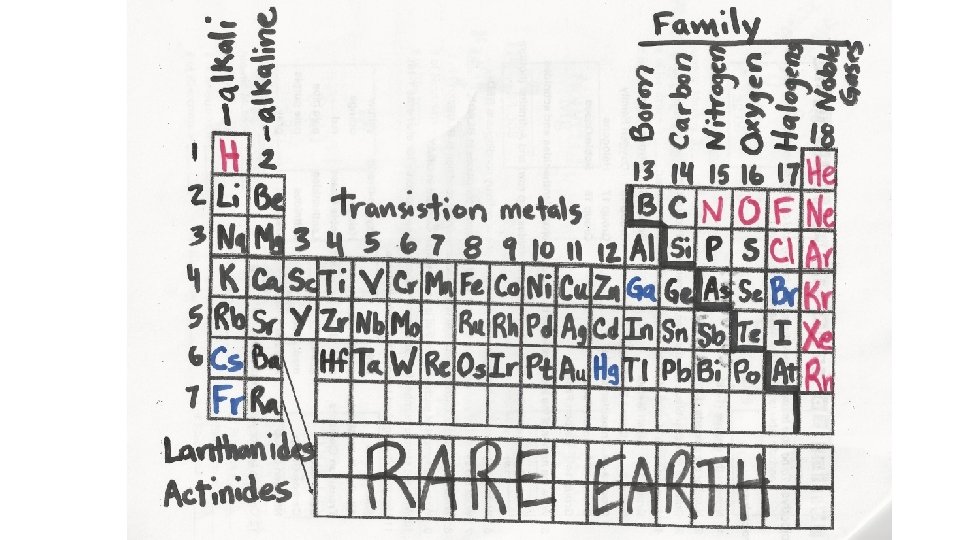
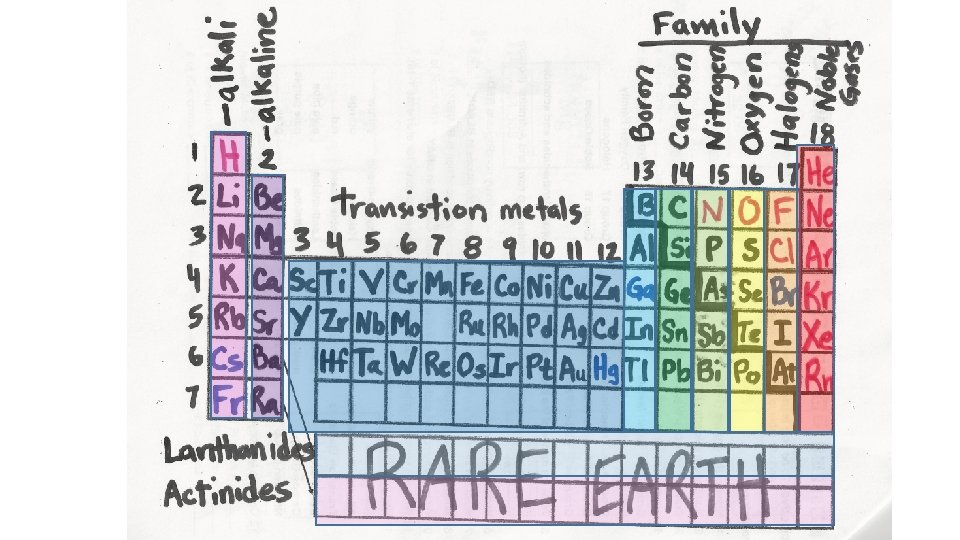
Periodic Table Trends in groups and rows Balanced

Periodic Table Trends in groups and rows; Balanced and unbalanced equations
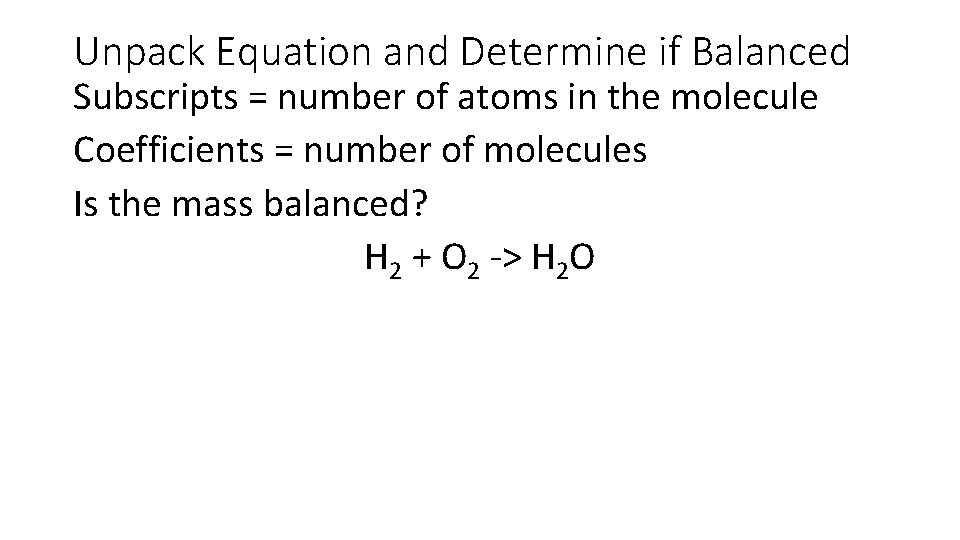
Unpack Equation and Determine if Balanced Subscripts = number of atoms in the molecule Coefficients = number of molecules Is the mass balanced? H 2 + O 2 -> H 2 O
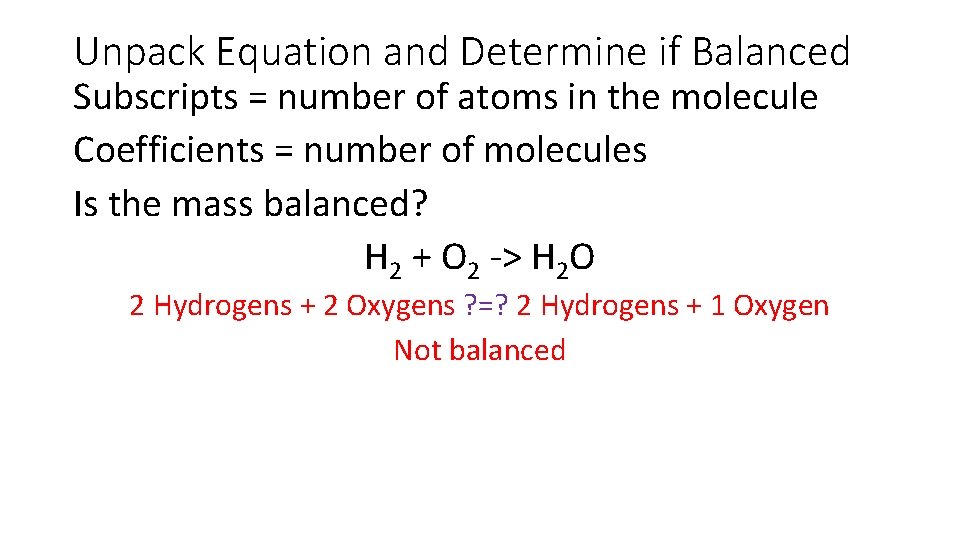
Unpack Equation and Determine if Balanced Subscripts = number of atoms in the molecule Coefficients = number of molecules Is the mass balanced? H 2 + O 2 -> H 2 O 2 Hydrogens + 2 Oxygens ? =? 2 Hydrogens + 1 Oxygen Not balanced

Unpack Equation and Determine if Balanced H 2 O 2 -> H 2 O + O 2

Unpack Equation and Determine if Balanced H 2 O 2 -> H 2 O + O 2 2 H + 2 O ? =? 2 H + 1 O + 2 O Combine like atoms on each side of the equation 2 H + 2 O ? =? 2 H +3 O Not balanced
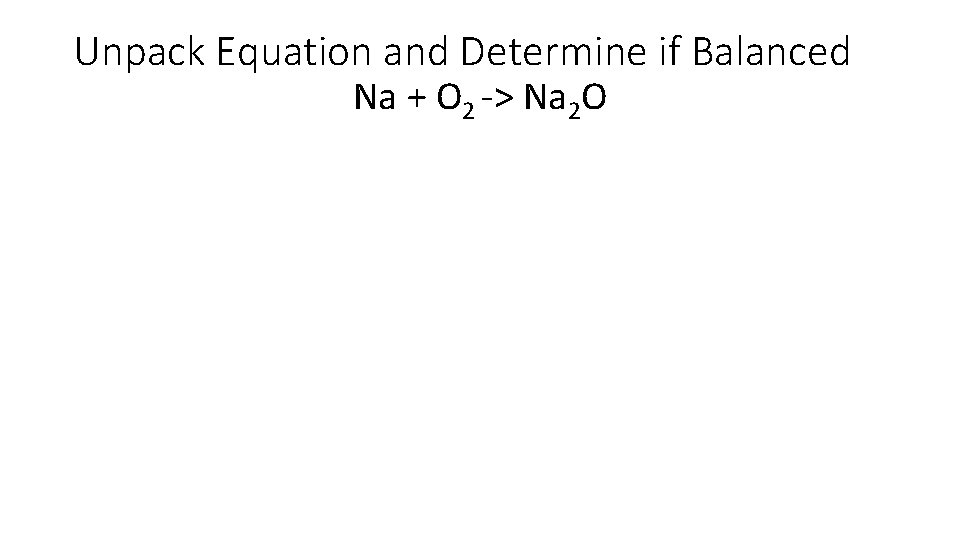
Unpack Equation and Determine if Balanced Na + O 2 -> Na 2 O
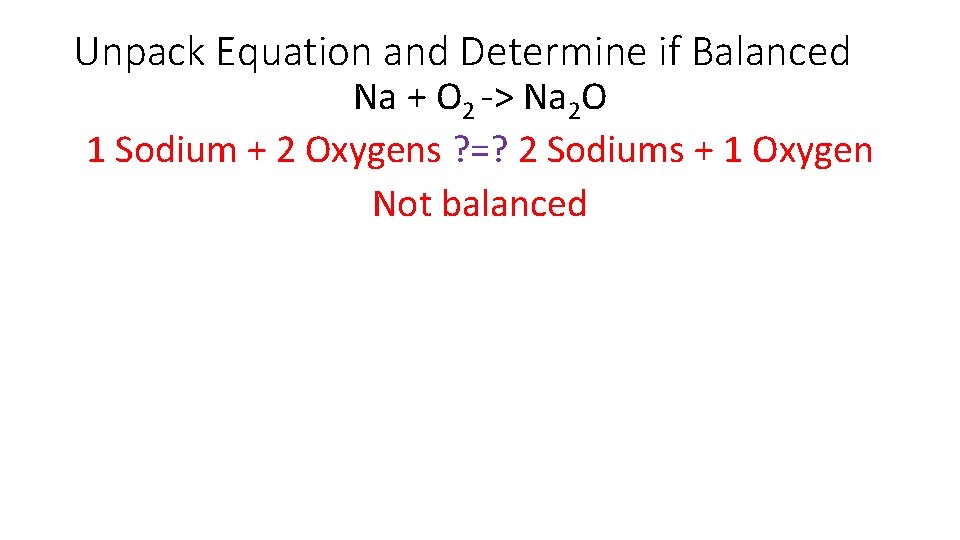
Unpack Equation and Determine if Balanced Na + O 2 -> Na 2 O 1 Sodium + 2 Oxygens ? =? 2 Sodiums + 1 Oxygen Not balanced

Unpack Equation and Determine if Balanced N 2 + H 2 -> NH 3

Unpack Equation and Determine if Balanced N 2 + H 2 -> NH 3 2 Nitrogen + 2 Hydrogens ? =? 1 Nitrogen + 3 Hydrogens Not balanced

Unpack Equation and Determine if Balanced P 4 + O 2 -> P 4 O 6

Unpack Equation and Determine if Balanced P 4 + O 2 -> P 4 O 6 2 Phosphorus’ + 2 Oxygens ? =? 4 Phosphorus’ + 6 Oxygens Not balanced

Unpack Equation and Determine if Balanced C + H 2 -> CH 4

Unpack Equation and Determine if Balanced Al 2 O 3 -> Al + O 2

Unpack Equation and Determine if Balanced Fe + H 2 O -> Fe 3 O 4 + H 2

Unpack Equation and Determine if Balanced C 2 H 6 + O 2 -> CO 2+ H 2 O

Unpack Equation and Determine if Balanced Na 2 SO 4 + Ca. Cl 2 -> Ca. SO 4 + Na. Cl
- Slides: 18