Periodic Table Trends History of the Periodic Table
Periodic Table & Trends

History of the Periodic Table • Dimitri Mendeleev was the first scientist to publish an organized periodic table of the known elements. • Considered an “enemy” of the Orthodox Church in Russia for his scientific work • Brilliant young genius
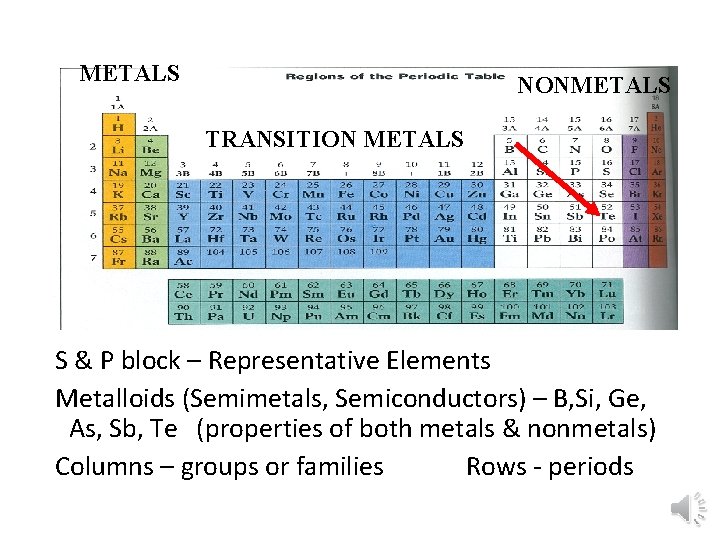
METALS NONMETALS TRANSITION METALS S & P block – Representative Elements Metalloids (Semimetals, Semiconductors) – B, Si, Ge, As, Sb, Te (properties of both metals & nonmetals) Columns – groups or families Rows - periods

Metals • Metals are lustrous (shiny), malleable, ductile, and are good conductors of heat and electricity. • They are mostly solids at room temp. • What is one exception?

Nonmetals • Nonmetals are the opposite. • They are dull, brittle, nonconductors (insulators). • Some are solid, but many are gases, and Bromine is a liquid.

Metalloids • Metalloids, aka semi-metals are just that. • They have characteristics of both metals and nonmetals. • They are shiny but brittle. • And they are semiconductors.

Periodic Groups • Elements in the same column have similar chemical and physical properties • These similarities are observed because elements in a column have similar e- configurations (same amount of electrons in outermost shell) • VALENCE ELECTRONS

What are Periodic Trends? • Periodic Trends – patterns in the behavior and traits of elements • Trends we’ll be looking at: 1. Atomic Radius 2. Ionization Energy 3. Electronegativity
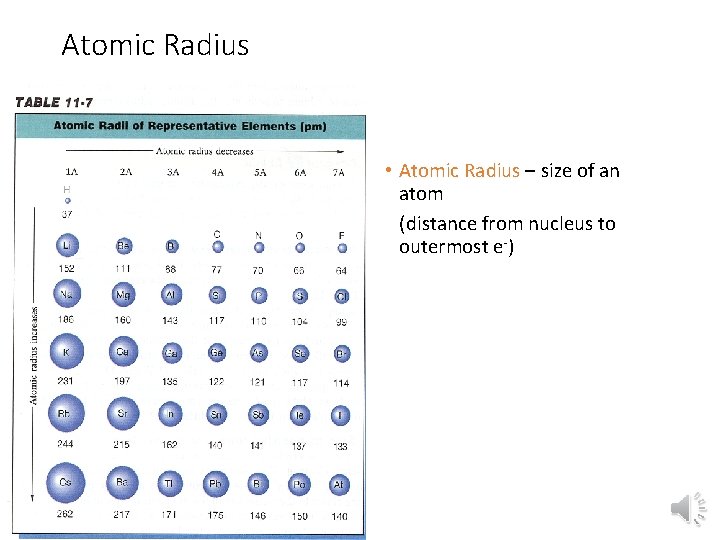
Atomic Radius • Atomic Radius – size of an atom (distance from nucleus to outermost e-)

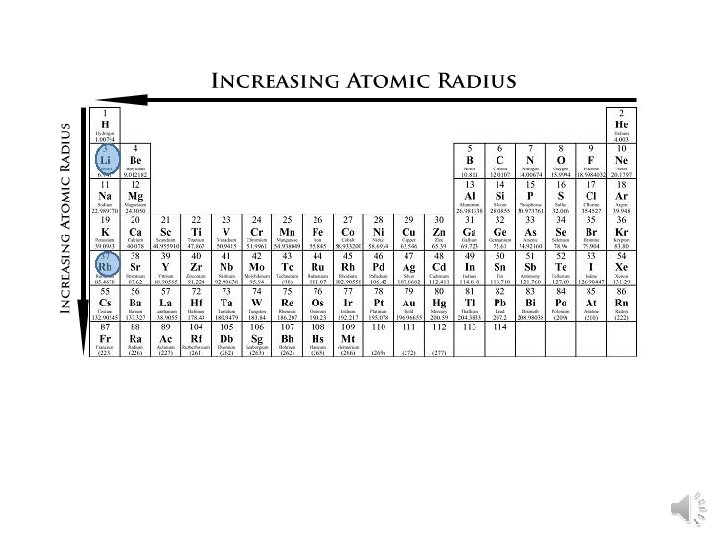
Atomic Radius Trend • Group Trend – As you go down a column, atomic radius increases As you go down, e- are filled into orbitals (energy levels) that are farther away from the nucleus (attraction not as strong) • Periodic Trend – As you go across a period (L to R), atomic radius decreases As you go L to R, e- are put into the same orbital, but more p+ (protons) and e- (electrons) (more attraction = smaller size)
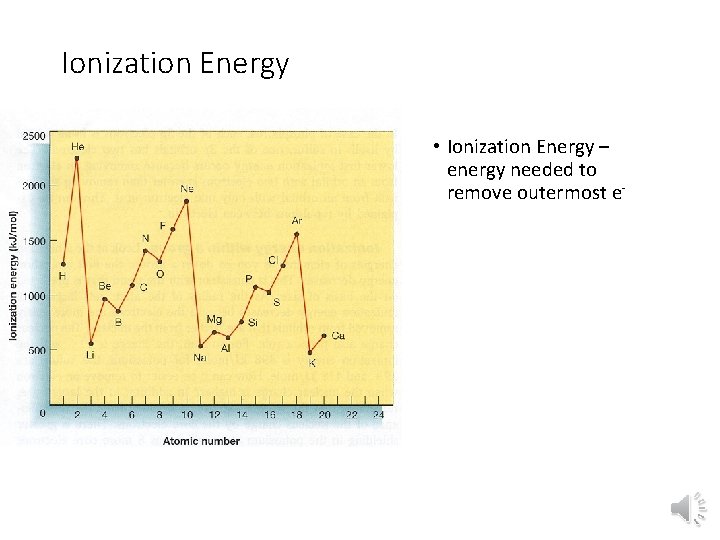
Ionization Energy • Ionization Energy – energy needed to remove outermost e-

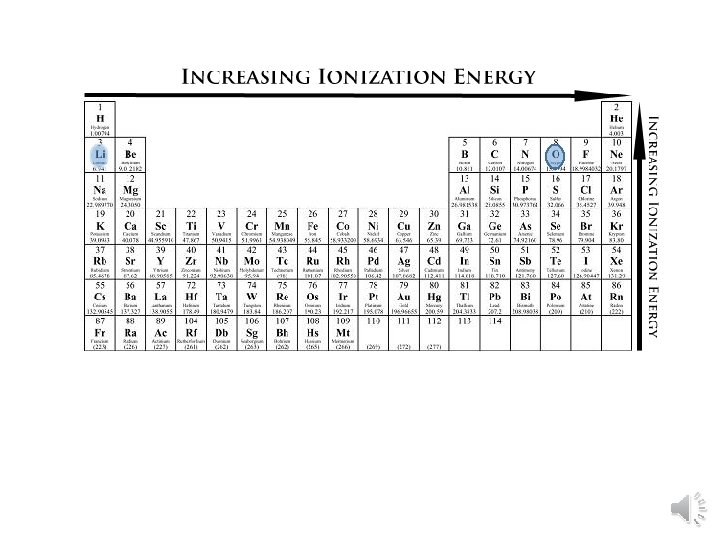
Ionization Energy • Group Trend – As you go down a column, ionization energy decreases As you go down, atomic size is increasing (less attraction), so easier to remove an e • Periodic Trend – As you go across a period (L to R), ionization energy increases As you go L to R, atomic size is decreasing (more attraction), so more difficult to remove an e(also, metals want to lose e-, but nonmetals do not)
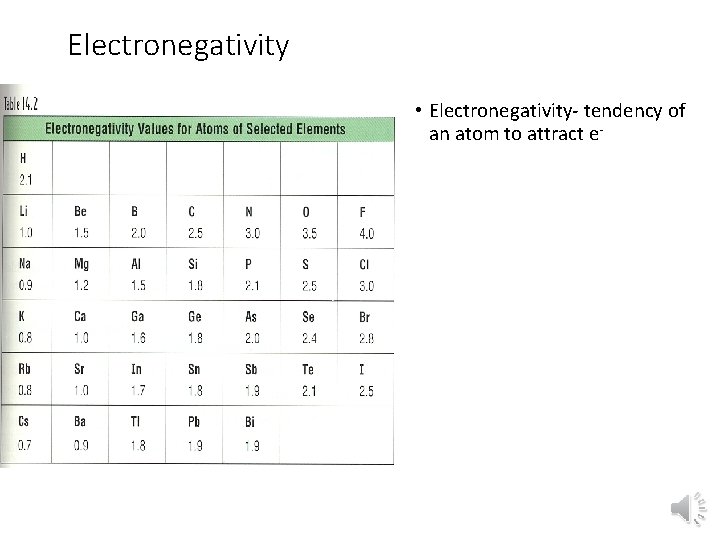
Electronegativity • Electronegativity- tendency of an atom to attract e-

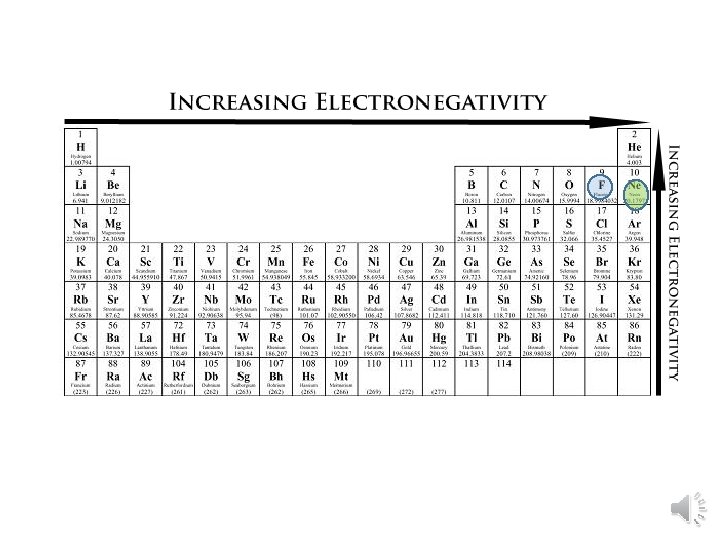
Electronegativity Trend • Group Trend – As you go down a column, electronegativity decreases As you go down, atomic size is increasing, so less attraction to its own e- and other atom’s e • Periodic Trend – As you go across a period (L to R), electronegativity increases As you go L to R, atomic size is decreasing, so there is more attraction to its own e- and other atom’s e-

Reactivity • Reactivity – tendency of an atom to react • Metals – lose e- when they react, so metals’ reactivity is based on lowest Ionization Energy (bottom/left corner) Low I. E = High Reactivity • Nonmetals – gain e- when they react, so nonmetals’ reactivity is based on high electronegativity (upper/right corner) High electronegativity = High reactivity

In conclusion…. • Describe the differences between: • • Metals Non-Metals Metalloids And provide an example for each • Define 3 Periodic Trends • (Psstt: Make sure your Periodic Table is LABELED!!!!)
- Slides: 19