Periodic Table Trends Atomic Radii Density Ionization Energy

Periodic Table Trends Atomic Radii Density Ionization Energy Electronegativity Ionic Radii

Atomic Radii n Decreases from left to right in a period n b/c shell shrinks as protons added to nucleus n Increases from top to bottom in a group n b/c each higher energy shell is farther from the nucleus

Atomic Radii Decreases from left to right in a period n Increases from top to bottom in a group n Which is smaller atom? Mg, Na, N or P Ans: Nitrogen is smaller than phosphorus which is smaller than magnesium which is smaller than sodium n

Density n METALS-Increases from left to right in period n NONMETALS-Decreases from left to right in period n Increases going down a group metals nonmetals ARROWS GIVE INCREASING DENSITY

Ionization Energy The energy required to remove an e’ from a (gaseous) atom n Unique for each e’, depends on distance from nucleus n L to R more e’ but all in same shell. As nuclear charge increases so does IE. n Top to Bottom – each shell further from nuclear attraction. IE decreases.
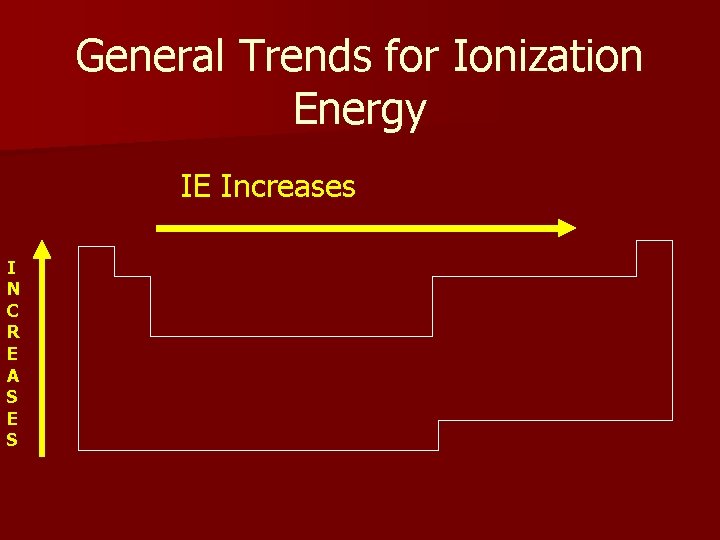
General Trends for Ionization Energy IE Increases I N C R E A S E S
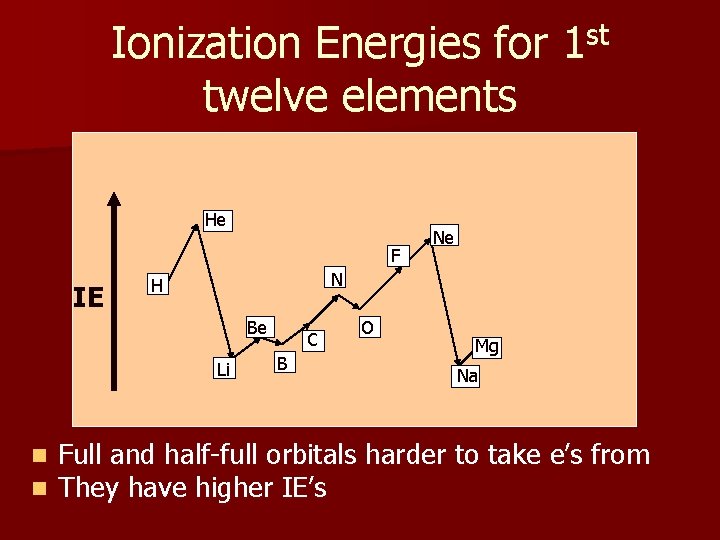
Ionization Energies for 1 st twelve elements He He Ne FF IE N N H H Be Be Li Li n n Ne C C B O O Mgg Mg B Na Na Full and half-full orbitals harder to take e’s from They have higher IE’s
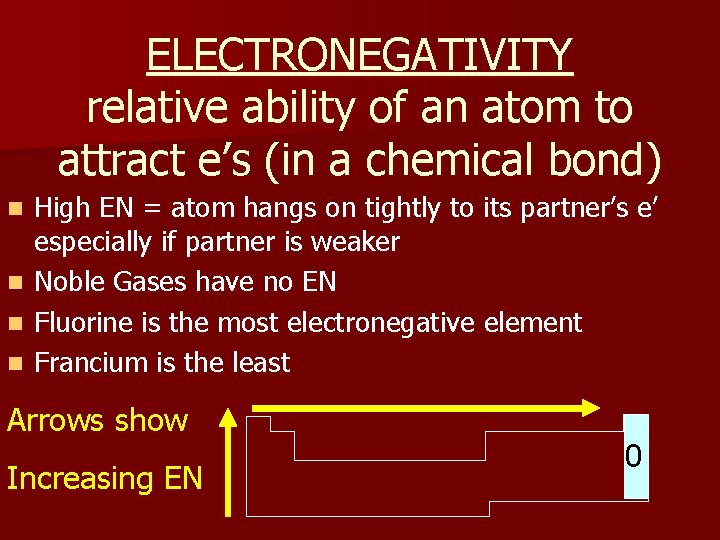
ELECTRONEGATIVITY relative ability of an atom to attract e’s (in a chemical bond) n n High EN = atom hangs on tightly to its partner’s e’ especially if partner is weaker Noble Gases have no EN Fluorine is the most electronegative element Francium is the least Arrows show Increasing EN 0

IONIC RADII n+ ions (cations) are smaller than their uncharged atoms due to loss of e’s from outer shell n - ions (anions) are larger than their noncharged atoms due to gain of e’s in outer (valence) shell. n Ex: Na 1 s 2 2 p 6 3 s 1 goes to Na+ 1 s 2 2 p 6 n Shell 3 has been vacated leaving smaller shell 2 as the outermost shell

Trends for Ionic Radii n Across a period- ionic radii decrease until column 5 switch to -3 anion = big increase in size n Begin decreasing again as ion charge lessens (-2, -1)

Ionic Radii for Representative Elements

Atomic & Ionic Radii Practice Which is larger? n. K atom or K+ cation? n F atom or F- anion? n Ca 2+ cation or K+ cation? n N 3 - anion or F- anion?

Electronegativity Practice n Which atom has the strongest attraction for an electron when a chemical bond is formed: n Oxygen or beryllium n Lithium or potassium n Sodium or nitrogen

- Slides: 14