PERIODIC TABLE The periodic table is a tabular
PERIODIC TABLE

The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number, electron configuration, and recurring chemical properties, whose structure shows periodic trends. Generally, within one row (period) the elements are metals to the left, and non-metals to the right, with the elements having similar chemical behaviours placed in the same column. Table rows are commonly called periods and columns are called groups. Six groups have accepted names as well as assigned numbers: for example, group 17 elements are the halogens; and group 18 are the noble gases.

Russian chemist Dmitri Mendeleev was the first to publish a recognizable periodic table in 1869, developed mainly to illustrate periodic trends of then-known elements. He also predicted some properties of unidentified elements that were expected to fill gaps within the table. Most of his forecasts proved to be correct. Mendeleev's idea has been slowly expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behaviour.

Alkali metals The alkali metals are a group (column) in the periodic table consisting of the chemical elements lithium (Li), sodium (Na), potassium(K), [note 1] rubidium (Rb), caesium (Cs), [note 2] and francium (Fr). This group lies in the s-block of the periodic table of elements as all alkali metals have their outermost electron in an s-orbital: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of group trends in properties in the periodic table, with elements exhibiting well-characterised homologous behaviour.

Metallic and Nonmetallic Properties ü About 80% of the periodic table are metals. ü Metallic properties increase from top to bottom and from right to left in a periodic table. ü Nonmetallic properties increase from bottom to top and from left to righ in a periodic table.
3 Group Neutron Electron Proton

1 group. Polyglot IRTGONEN PASOTIMUS TALOCB IMUYEBRLL RLUFUS RCRUEMY MPLAUNIT NITROGEN POTASSIUM COBALT BERYLLIUM SULFUR MERCURY PLATINUM

2 GROUF. Identification • • Күкірт Қалайы Көміртек Алтын Сурьма Мышьяк Азот Nitrogen Gold Tin Sulfur Carbon Arsenic Stibium
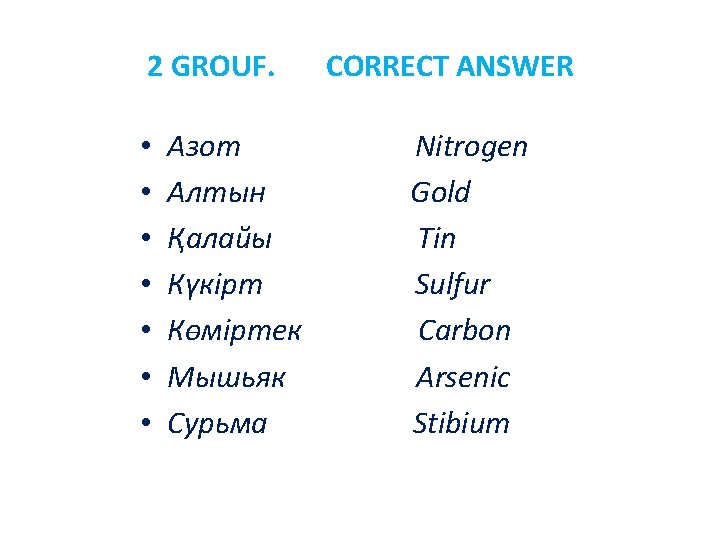
2 GROUF. • • Азот Алтын Қалайы Күкірт Көміртек Мышьяк Сурьма CORRECT ANSWER Nitrogen Gold Tin Sulfur Carbon Arsenic Stibium

3 GROUF. Reading part soft, charge, oxygen, salts, outermost electron, reactive, cations The alkali metals are all shiny, _______, highly _______ metals at standard temperature and pressure and readily lose their _______to form _____ with _______ +1. They can all be cut easily with a knife due to their softness, exposing a shiny surface that tarnishes rapidly in air due to oxidation by atmospheric moisture and _____ (and in the case of lithium, nitrogen). Because of their high reactivity, they must be stored under oil to prevent reaction with air, and are found naturally only in______ and never as the free elements. Caesium, the fifth alkali metal, is the most reactive of all the metals.

3 GROUF. Reading part The alkali metals are all shiny, soft, highly reactive metals at standard temperature and pressure and readily lose their outermost electronto form cations with charge +1. They can all be cut easily with a knife due to their softness, exposing a shiny surface that tarnishes rapidly in air due to oxidation by atmospheric moisture and oxygen (and in the case of lithium, nitrogen). Because of their high reactivity, they must be stored under oil to prevent reaction with air, and are found naturally only in salts and never as the free elements. Caesium, the fifth alkali metal, is the most reactive of all the metals.

Questions § § § § How Mendeleyev came to life? (1834) Date of birth? (February 8) How many children in the family? (7 ) When was the periodic law opened? (1869) How many elements are currently known? (118) What elements did he predict beforehand? (21, 32) What is the number of the element in honor of Mendeleev? (101)
- Slides: 12