Periodic Table Symbols Element Symbol Oxygen O Chromium

Periodic Table
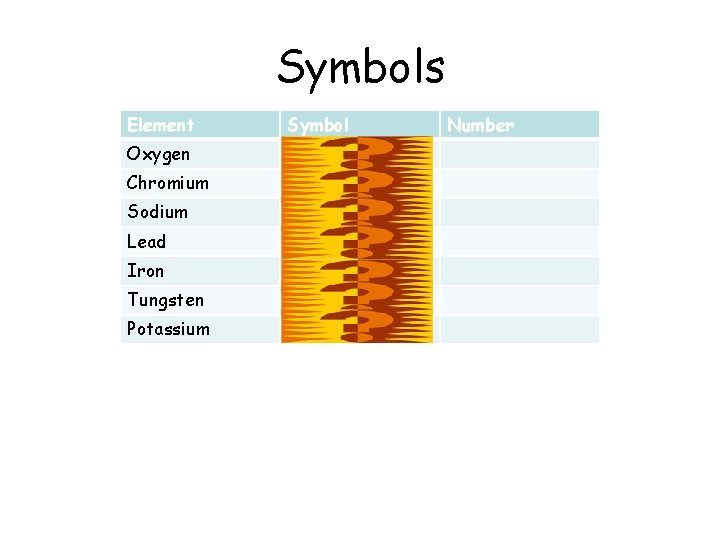
Symbols Element Symbol Oxygen O Chromium Cr Sodium Na Lead Pb Iron Fe Tungsten W Potassium K Number
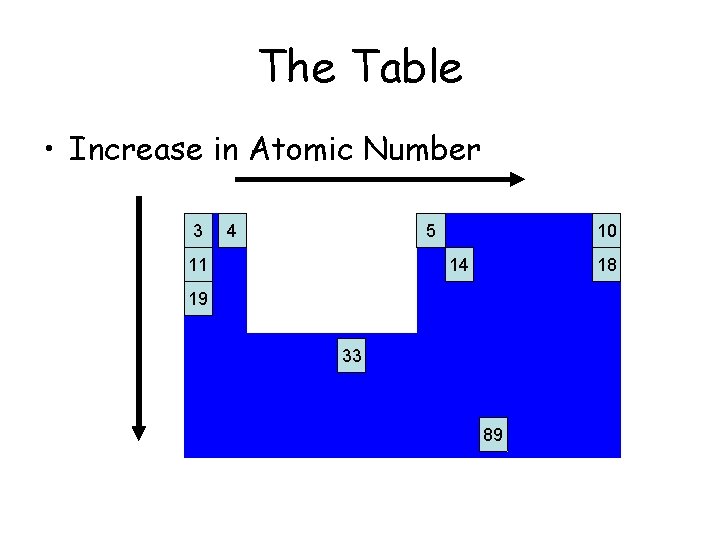
The Table • Increase in Atomic Number 3 4 5 11 10 14 18 19 33 89

Notice how the first few start a new row every 8 atoms
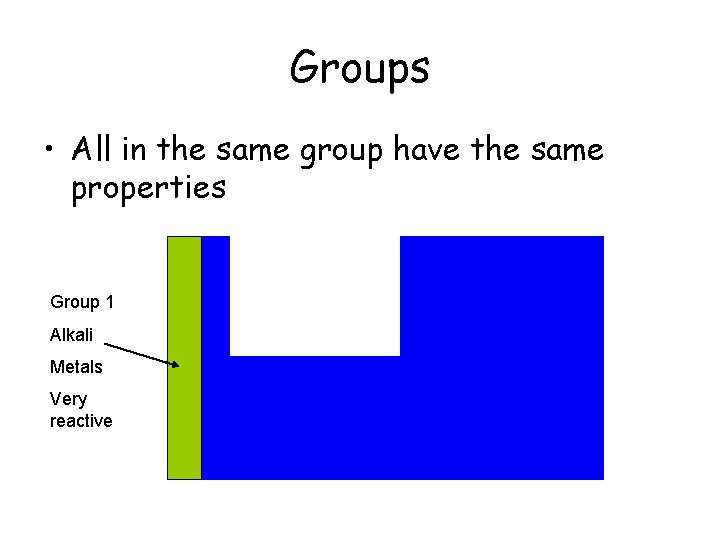
Groups • All in the same group have the same properties Group 1 Alkali Metals Very reactive
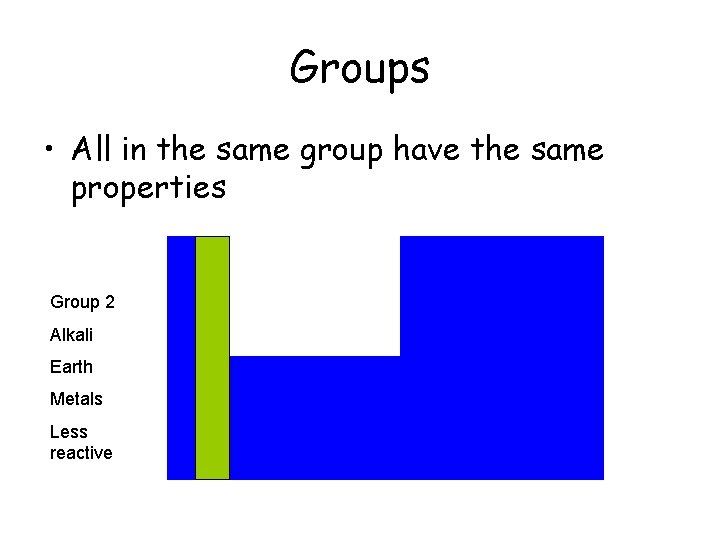
Groups • All in the same group have the same properties Group 2 Alkali Earth Metals Less reactive
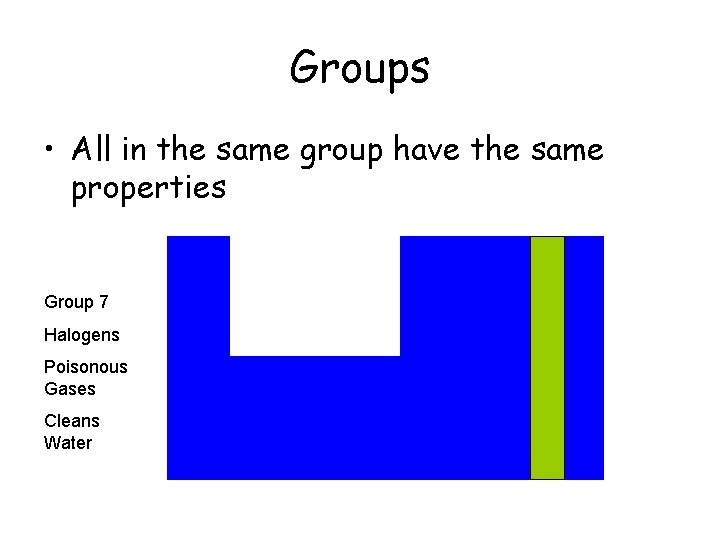
Groups • All in the same group have the same properties Group 7 Halogens Poisonous Gases Cleans Water
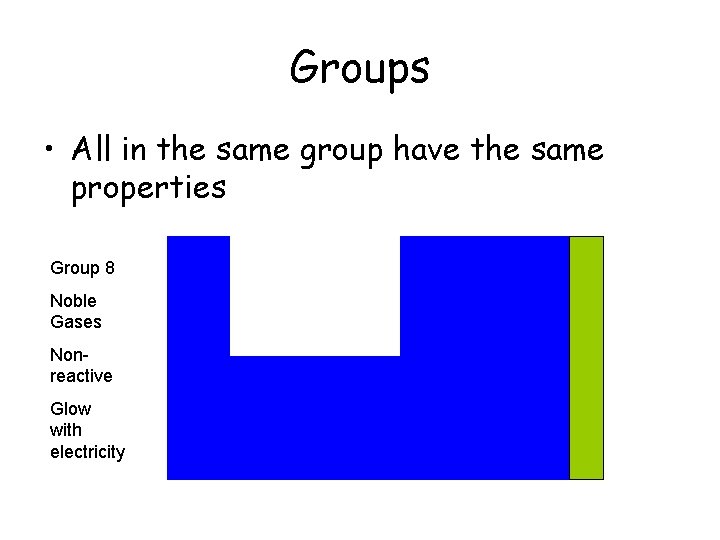
Groups • All in the same group have the same properties Group 8 Noble Gases Nonreactive Glow with electricity

Groups • All elements in the same group have the same properties Group 8 Group 2 Noble Gases Alkali Earth Group 1 Metals Alkali Less reactive Metals Very reactive Group 7 Halogens Poisonous Gases Cleans Water Nonreactive Glow with electricity
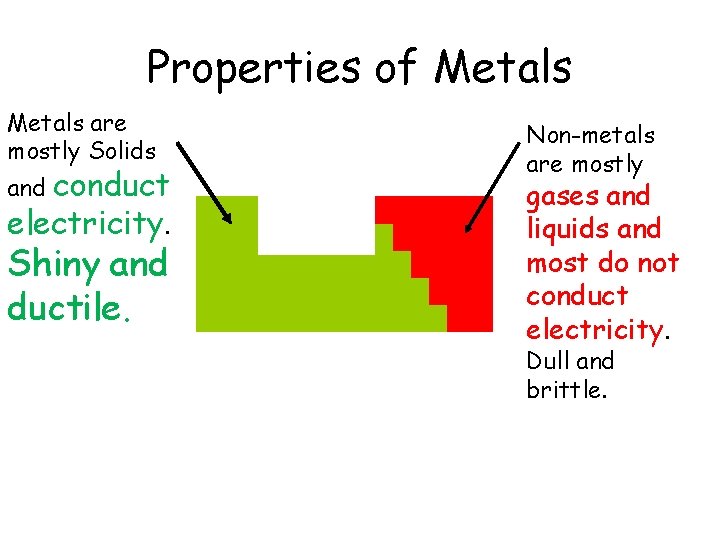
Properties of Metals are mostly Solids conduct electricity. and Shiny and ductile. Non-metals are mostly gases and liquids and most do not conduct electricity. Dull and brittle.

Alloys • • • Two metals mixed together Some of both properties Brass – Copper and Zinc Bronze – Copper and Tin Stainless Steel – Chromium and Iron (and carbon)
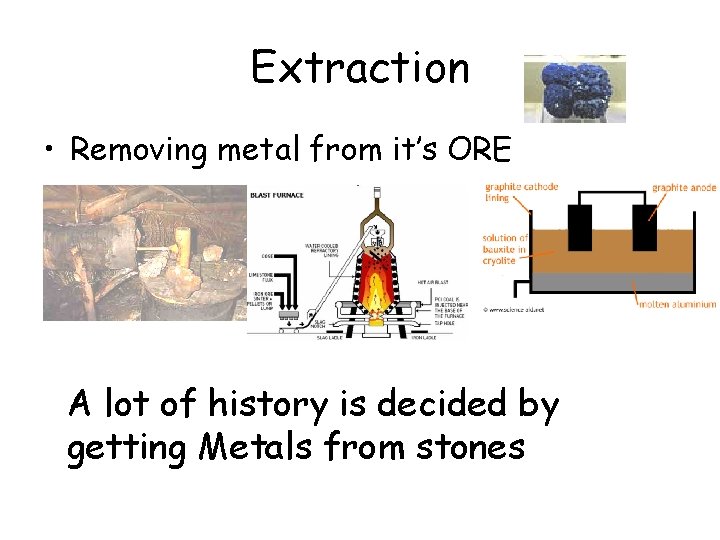
Extraction • Removing metal from it’s ORE A lot of history is decided by getting Metals from stones
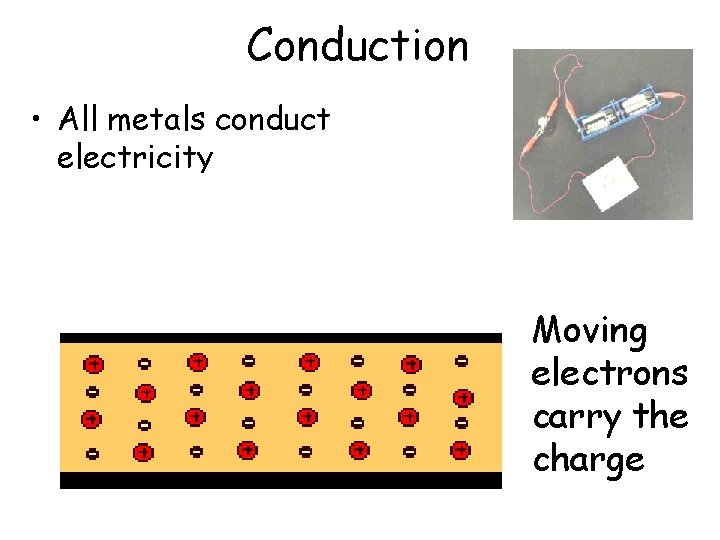
Conduction • All metals conduct electricity Moving electrons carry the charge
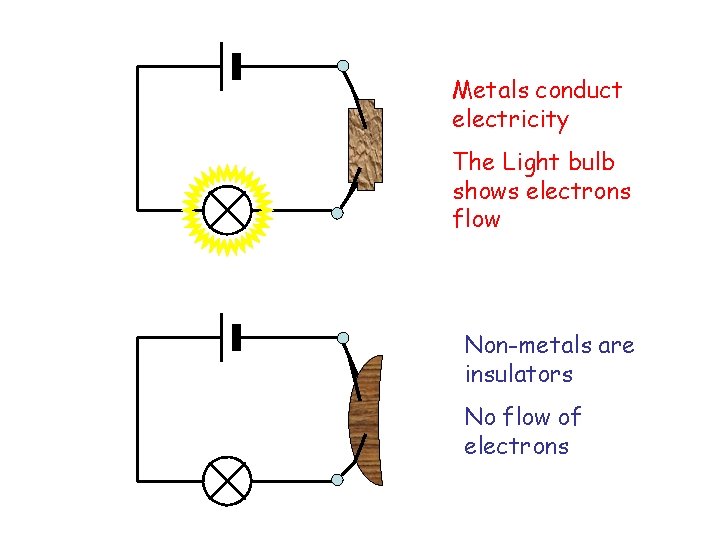
Metals conduct electricity The Light bulb shows electrons flow Non-metals are insulators No flow of electrons
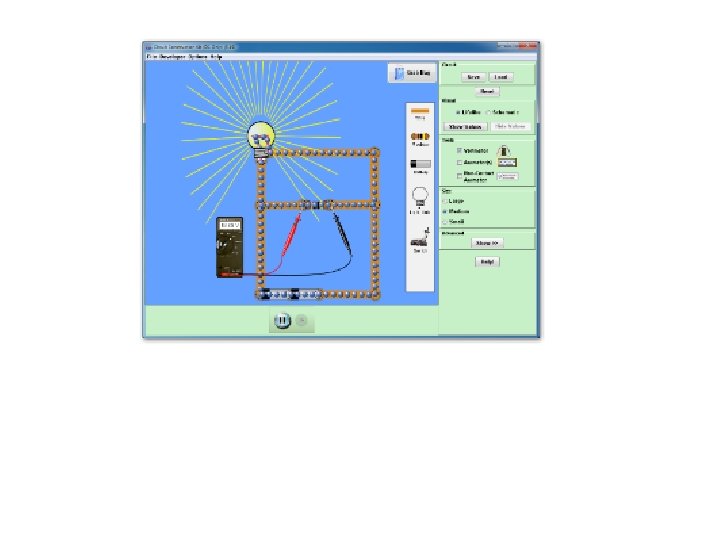

Summary • • • Groups all same reactions Names of groups and reactions Metals and non-metals Conduction of electricity Metals and alloys

H/W Explain to your parents what the periodic table actually is and then write down what you said.
- Slides: 17