Periodic Table Review Periodic Table structured arrangement of

Periodic Table Review

Periodic Table • • structured arrangement of elements explains physical and chemical properties of elements

What is an element? A pure substance that cannot be broken down into smaller substances.

What is an atom? • smallest particle of an element that retains all the properties of the element electrically neutral o made of subatomic particles: § protons, neutrons, and electrons o

Charge of Subatomic Particles Proton positively charged (1+), in nucleus Neutron neutral charge (charge of 0), in nucleus Electron negatively charged (-1), surrounds nucleus • • •

Atom is neutral, so # of protons = # of electrons

Information on the Periodic Table for each Element - see handout

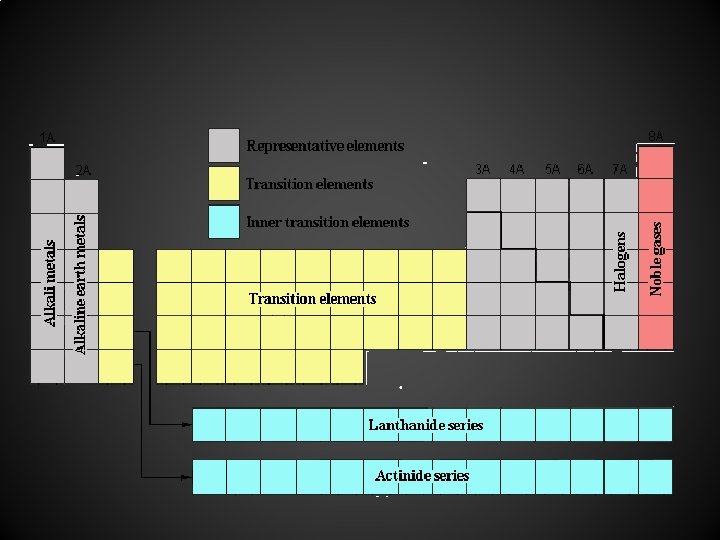
Elements and the Periodic Table Metals are found on the left side of the periodic table. Non-metals are found on the right side of the periodic table. Metalloids - metallic staircase properties of both metals and non-metals, typically dull gray, malleable solids exception aluminum • •

Groups are the columns of the periodic table (ex. calcium is group 2) **Families Periods are the horizontal rows of the periodic table (ex. calcium is period 4)

(continued) Chemical Families = groups of elements in the same vertical column of the periodic table. Group 1 - Alkali metals shiny, silvery metals that are very soluble in water • Group 2 - Alkaline earth metals shiny, silvery metals that are less reactive; used in alloys •

continued Group 17 - Halogens poisonous, nonmetallic elements, F, Cl are yellowish-green gases, Br is a dark red liquid, I is a purple-black solid • Group 18 - Noble Gases non-reactive gases; used in 'neon' signs •

Transition & inner-transition elements found in the middle of the periodic table They are usually high melting point metals. They have several oxidation states. They usually form colored compounds. They are often paramagnetic. • •
Periodic Table Use the given periodic table and identify (colour/outline) the following: • • • metals non-metals metalloids Group 1 Group 2 Group 17 Group 18 Periods Families
- Slides: 14