Periodic Table PT History of the Periodic Table

Periodic Table- PT

History of the Periodic Table n one of the major milestones in the history of chemistry n helped to predict the existence of elements that had yet to be discovered

JW Dobereiner n n n n 1829 - about 30 known elements Noticed that elements with similar atomic weights and their chemical properties Classified some elements in triads (sets of three) Li, Na, K Ca, Sr, Ba Cl, Br, I Look on the current periodic http: //www. juntadeandalucia. es table- notice a pattern?

JAR Newlands n n n 1865 - about 56 elements were known Noticed that if elements were arranged in increasing atomic mass, the properties of the 8 th element were like those of the 1 st, the 9 th like those of the 2 nd, the 10 th like those of the 3 rd, and so on Called this the Law of Octaves (nobody believed him) again check the current perodic table http: //falcon. sbuniv. edu

Dimitri Mendeleev n n n 1869 Published his periodic table arranging elements by their atomic mass Also arranged the table so that elements in the same column have similar properties Left blank spots in his periodic table Predicted the existence of elements yet to be discovered http: //www. rit. ac. th

Mendeleev’s Periodic Table “I began to look about and write down the elements with their atomic weights and typical properties, analogous elements and like atomic weights on separate cards, and this soon convinced me that the properties of elements are in periodic dependence upon their atomic weights. ” --Mendeleev, Principles of Chemistry, 1905, Vol. II

n one of the predicted elements he named ekasilicon n eka - first located below silicon on the periodic table n 1886 germanium was discovered –very close to the properties that Mendeleev predicted for ekasilicon n

HGJ Moseley 1913 – HGJ Moseley developed the concept of atomic number ( he was working in Rutherford’s lab who discovered protons) n the correct way to arrange the elements is by atomic number NOT atomic mass n http: //dbhs. wvusd. k 12. ca. us

Mendeleev’s table is close – because generally atomic mass increases with atomic number n Can you find the exceptions to the rule? n Periodic Law – the physical and chemical properties of the elements are periodic functions of their atomic numbers n Today we give credit for the periodic table to Mendeleev and Moseley (think M&M) n

The Modern Periodic Table

n n n Elements are found in squares Different periodic tables have different information in the squares but they may include: Symbol, name, atomic number, either atomic mass or mass number. Some periodic tables even include electron configurations and oxidation numbers (charge of element) The squares are then arranged into rows & columns The rows are called periods The columns are called groups or families

↔ n 7 periods n 18 groups ↕

Periods Ø Ø Ø Horizontal rows Arranged by atomic number As you go down the table each period has more and more elements Ø 1 st row has only 2 elements- Hydrogen & Helium Ø 2 nd and 3 rd rows have 8 elements- Li Ne, Na Ar Ø 4 th and 5 th rows have 18 elements Ø How many does the 6 th row have? Ø 32

Groups/Families labeling and naming of the groups n IUPAC method just numbers each group 1 -18 n American method- uses numbers 1 -8 and the letters A & B (some PTs use roman numbers IVII) n 1 -8 A elements in the S & P blocks n 1 -8 B elements in the D & F blocks n This method is more useful to identifying the charges of elements n

n European Method – uses the numbers 1 -8 and the letters A & B only A is for metals & B is for non metals. (Not very helpful method) n Hydrogen- can be found in different locations n is usually not connected to the table because in terms of reactivity, it is more like the halogens (group 17), but its electron configuration is like that of the alkali metals (group 1)

Metal n n n characteristic luster or shine good conductors of heat and electricity typically solids at room temperature malleable – can be hammered into thin sheets many are ductile – can be pulled into thin wires http: //www. webelements. com

n PT also seperates the metals from the nonmetals this can be done by colors

Non-metal n n no luster poor conductors not malleable or ductile many are gases at room temperature, others are solids n n Br - liquid varied properties n n colored, colorless soft solids, hard solids http: //www. webelements. com

Semi-metals or metalloids n some properties of metals and non-metals or intermediate n Si - principle component of computer chips http: //www. webelements. com

Electron Configuration and the Periodic table electron configuration for the first 3 elements in group 1 A : n H 1 s 1 n Li 1 s 22 s 1 n Na 1 s 22 p 63 s 1 n the highest energy electron is in the ‘s’ orbital n

electrons occupying the highest principle energy level are the atoms’ outermost electrons n ****Valence electrons**** n located in ‘s’ and ‘p’ orbitals n these valence electrons are the electrons that are going to interact with other elements n

Abbreviated Electron Configuration the atom’s inner electrons are represented by the symbol for the nearest noble gas – with a lower atomic number n K - 1 s 22 p 63 s 23 p 64 s 1 n [Ar] 4 s 1 n argon’s electron configuration of electrons after that n
![n n n n H 1 s 1 Li [He] 2 s 1 Na n n n n H 1 s 1 Li [He] 2 s 1 Na](http://slidetodoc.com/presentation_image_h2/a18ff649a6970cb6ca460f095a38e32e/image-23.jpg)
n n n n H 1 s 1 Li [He] 2 s 1 Na [Ne] 3 s 1 K [Ar] 4 s 1 Rb [Kr] 5 s 1 Cs [Xe] 6 s 1 all elements in group 1 A have a single valence electron in the ‘s’ orbital the principal quantum number of this ‘s’ orbital is the same as the element’s period or row number
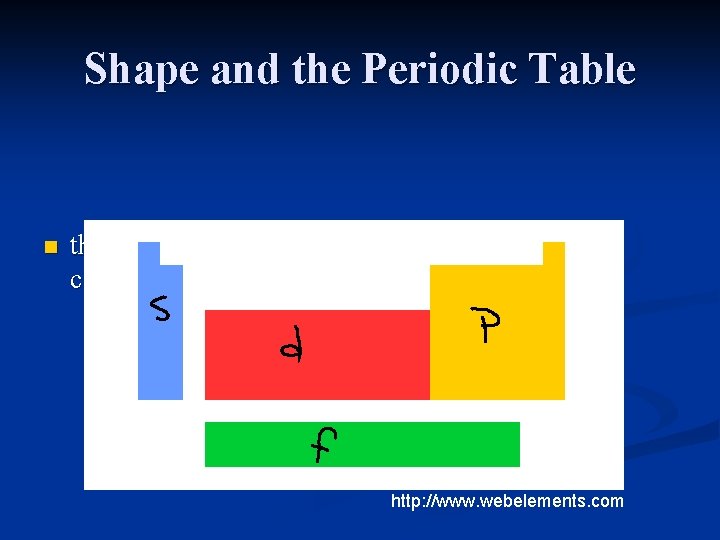
Shape and the Periodic Table n the shape of the table is related to the electron configuration of the elements http: //www. webelements. com

s block H, He and the elements in group 1 A and 2 A n valence electrons are in ‘s’ orbitals n the ‘s’ block is 2 elements wide n an ‘s’ orbital can only hold 2 electrons n the elements in group 1 A have 1 valence electron n the elements in group 2 A have 2 valence electrons n

p block group 3 A – 8 A n the p block is 6 elements wide n p orbitals can hold a maximum of 6 electrons n

d block takes up most of the middle of the table n 10 elements wide n

f block n the 28 elements below the main body of the periodic table
http: //www. webelements. com

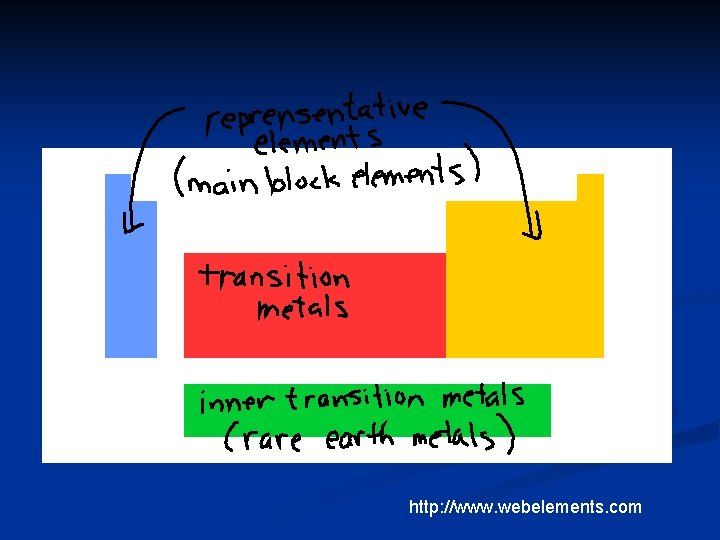
the s and p blocks are also called the representative elements or the main block elements n d block is also called the transition metals n f block is also called the inner transition metals n

Periodic Trends

Periodic Trends the properties of elements change in a predictable way as you move through the periodic table n nuclear charge n atomic radius n shielding n ionization energy n electronegativity n

Nuclear Charge n as more protons are added to the nucleus the charge on the nucleus increases

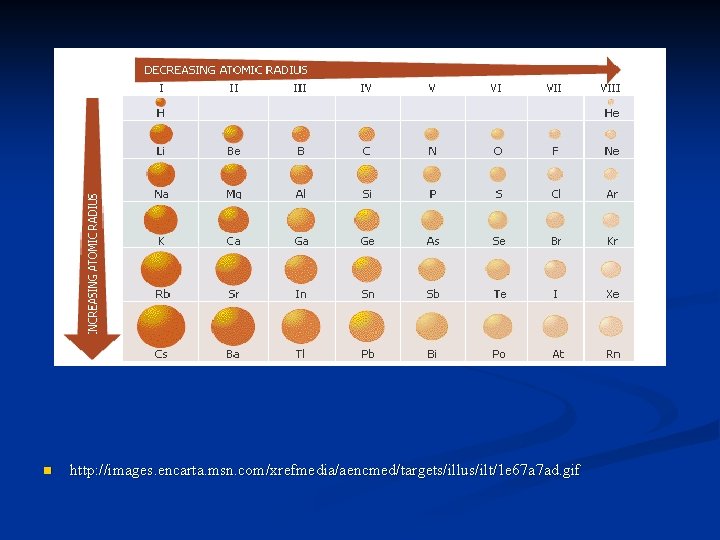
Atomic radius n n n radius – center of nucleus to outermost electrons as you move down a group the principal quantum number of the outermost electrons increases 1 A 1 s 1 -2 s 1 -3 s 1 -4 s 1 -5 s 1 electrons with a larger principal quantum number are found in orbitals that extend farther and farther from the nucleus – which make the atomic radius larger

n n n as you move left to right across the table the atomic radius decreases in any period as you move from left to right, the atoms’ nuclei gain more protons n n more positive charge a stronger pull is exerted on the electrons in a given principal quantum level electrons are pulled in closer to the nucleus the atom becomes smaller
n http: //images. encarta. msn. com/xrefmedia/aencmed/targets/illus/ilt/1 e 67 a 7 ad. gif

Shielding effect comes into play as you go down the group n as electrons are added to successively higher principal energy levels, the innermost electrons shield the outer electrons from the pull of the positive charge from the nucleus, making the atom larger n

Ionic Size when an atom gains or loses an electron, it forms an ion n if it loses an electron n positive ion (cation) n it becomes smaller n n 1 less electron reduces the electron – electron repulsion, allowing the electrons to be pulled closer to the nucleus

n if it gains an electron n negative ion it becomes larger greater number of electrons increases the repulsion forces among the electrons Group 1 A generally form 1+ ions Group 2 A generally form 2+ ions Group 7 A generally form 1 - ions Group 6 A generally form 2 - ions Noble gases ? n do not form ions

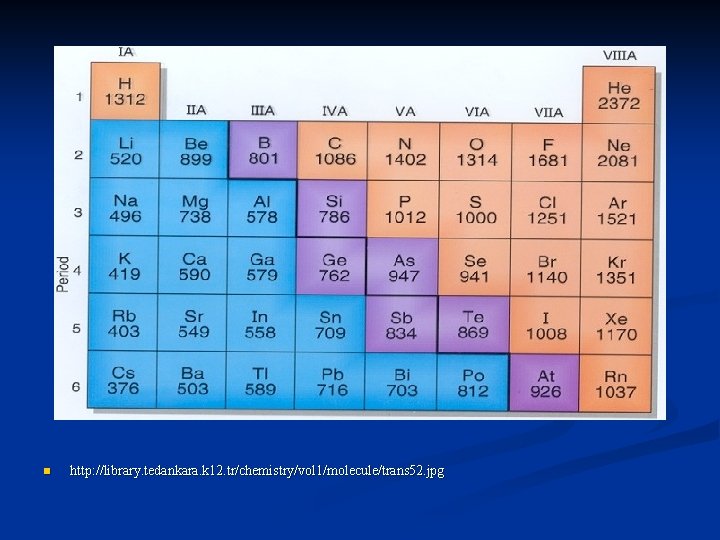
Ionization Energy energy needed to remove an electron n measured in k. J/mole n reflects how strongly an atom holds onto its outermost electrons n

high ionization energy holds onto electrons very tightly n low ionization energy more likely to lose one or more of its outermost electrons n to ionize a mole of magnesium atoms it takes 738 k. J of energy n

ionization energies decrease as you move down a group n larger atoms electrons are held less strongly – less energy needed to remove an electron n

ionization energies increase as you move left to right across a period n smaller atoms hold electrons more strongly - so more energy is required to remove an electron n n *** opposite to atomic radius trends***
n http: //library. tedankara. k 12. tr/chemistry/vol 1/molecule/trans 52. jpg

Equations Mg (g) Mg 1+(g) + en measured in the gas state – electrons must be far apart for an accurate measurement to be made n

Successive ionization energies required to remove electrons beyond the first electron n 1 st n Mg (g) Mg 1+(g) + en 2 nd Mg 1+ (g) Mg 2+(g) + e- n 738 k. J/mol 1450 k. J/mol 3 rd Mg 2+ (g) Mg 3+(g) + e- 7730 k. J/mol

the large jump in energy between the 2 nd and 3 rd ionization energy is due to the fact that the third electron removed from the magnesium atom is part of the noble gas inner core n these electrons are very difficult to remove n

the increase in ionization energy is also due in part to the reduced electron – electron repulsion n as each electron is removed from the atom the remaining electrons are pulled closer and tighter to the nucleus n

Electron Affinity the energy change that occurs when an electron is gained by an atom n represented in k. J/mole n Equation: n F (g) + e- F- (g) -328 k. J/mol n fluorine has a negative affinity because energy is released when a mole of F atoms gains electrons n exothermic n

in general, non-metals have more negative electron affinities than do metals n except noble gases n n n positive electron affinities electron affinity is related to the number of electrons needed to fill its outer energy level

group 7 A elements have a very strong electron affinity n by looking at ionization energies and electron affinities – we can derive an important principle about atoms n Octet rule n n atoms tend to gain, lose or share electrons in order to acquire a full set of valence electrons
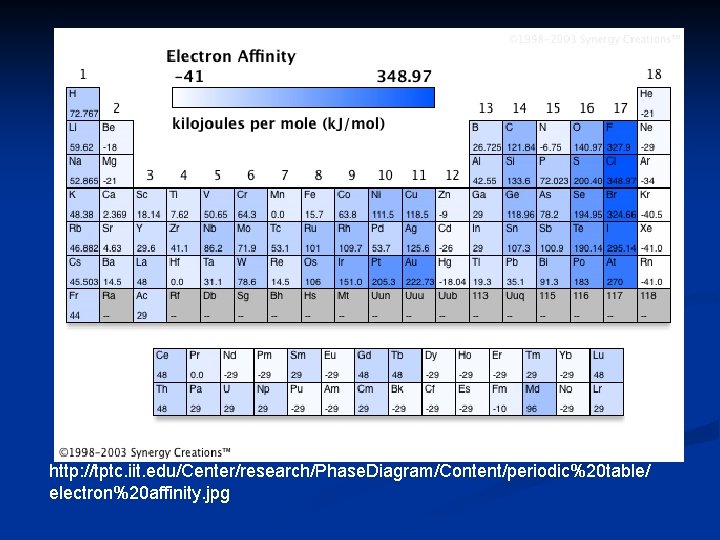
http: //tptc. iit. edu/Center/research/Phase. Diagram/Content/periodic%20 table/ electron%20 affinity. jpg

Electronegativity a measure of the ability of an atom in a chemical compound to attract electrons n fluorine is the most electronegative element n
n n n atomic radius decreases ionization energy increases electronegativity increases nuclear charge increases shielding is constant

n n n atomic radius increases ionization energy decreases electronegativity decreases nuclear charge increases shielding increases

n http: //www. webelements. com/webelements/properties/media/tables/balls/electroneg-pauling. gif
- Slides: 56