PERIODIC TABLE Periodic Table J W Dbereiner q

PERIODIC TABLE

Periodic Table J. W. Döbereiner q 1829—TRIADS, groups of the elements q similar in chemical and physical properties q properties based on atomic mass q Useful q Triads were useful b/c they grouped elements together that had similar properties q For the first time a pattern was observed based on atomic mass

Periodic Table Dmitri Mendeleev (the big dawg of the Periodic Table) q Chem. Prof. In St. Petersberg, 1869 q Noticed patterns within the elements that Döbereiner described q Organized the elements further q Published the first chart in 1869 q elements increased in size from top to bottom (groups) q elements that shared properties went in same rows from left to right (periods)

Periodic Table Dmitri Mendeleev q a second table was published later q more elements q further speculation q patterns repeated each period
Periodic Table

Periodic Table Dmitri Mendeleev q a second table was published later q more elements q further speculation q patterns repeated each period q vertical columns (groups) had similar properties
Periodic Table

Periodic Table Dmitri Mendeleev q a second table was published later q more elements q further speculation q patterns repeated each period q vertical columns (groups) had similar properties q horizontal rows (periods) had patterns of changing properties
Periodic Table

Periodic Table Dmitri Mendeleev q a second table was published later q more elements q further speculation q patterns repeated each period q vertical columns (groups) had similar properties q horizontal rows (periods) had patterns of changing properties

Periodic Table Dmitri Mendeleev q His table was first to show Periodicity q tendency to reoccur, i. e. —moon every month or Halley’s comet every 76 years q True Brilliance q left spaces for undiscovered elements q all based on properties q even speculated (correctly) on those properties

Periodic Table Modern Table q Very similar to Mendleev’s q periodicity still seen q Arranged by atomic number (# of protons) rather than atomic mass q except for first period, all periods start with a metal and end with a noble gas

Periodic Table Three types of elements… ATOM Smallest unit of an element that has the same properties as the element ION An atom that has lost or gained one or more electrons—it is charged (+/-) ISOTOPE Atom with the same number of PROTONS but different number of NEUTRONS than other atoms of the same element (different atomic mass)

Periodic Table Ions • TRANSFER of electrons • Atoms that transfer electrons become IONS • GAIN electron = NEGATIVE ion/ or anion • LOSE electron = POSITIVE ion or cation

Periodic Table Why Do Ions have a charge? • Sodium atom 11 electrons (-) 11 protons (+) 0 (+/-) • Sodium ion 10 electrons (-) 11 protons (+) 1+

Periodic Table Why Do Ions have a charge? • Nitrogen atom 7 electrons (-) 7 protons (+) 0 (+/-) • Nitrogen ion 10 electrons (-) 7 protons (+) 3–

Periodic Table Why Do Ions have a charge? Oxidation Number • Nitrogen ion 10 electrons (-) 7 protons (+) 3– IONIZATION: process of becoming an ion

Periodic Table Ions • You can use the periodic table to determine the charge an ion will have • It’s all about valence electrons and the octet rule • 1, 2, 3 valence electrons—give them away “cheaper” = +1, +2, +3 • 5, 6, 7 valence electrons—take in new e-s “cheaper” = -3, -2, -1

Periodic Table Ions—Bonding Capacity • Once you have determined the oxidation number you can determine how many bonds • • +1, bonds once -1, bonds once +3, bonds 3 times -3, bonds 3 times

Periodic Table � There are several important trends in the PT: 1. 2. 3. � Ionization Energy Electronegativity (electron affinity) Atomic Radius Periodicity is the study of the trends of the periodic table.

Ionization Energy � Ionization energy, E 1, is the energy needed to remove the most loosely held electron from a neutral gaseous atom. • As we go across the P. T. , E 1 increases because you have more protons (more + charges) • As we go down the P. T. , E 1 decreases because the e- is further away from the nucleus
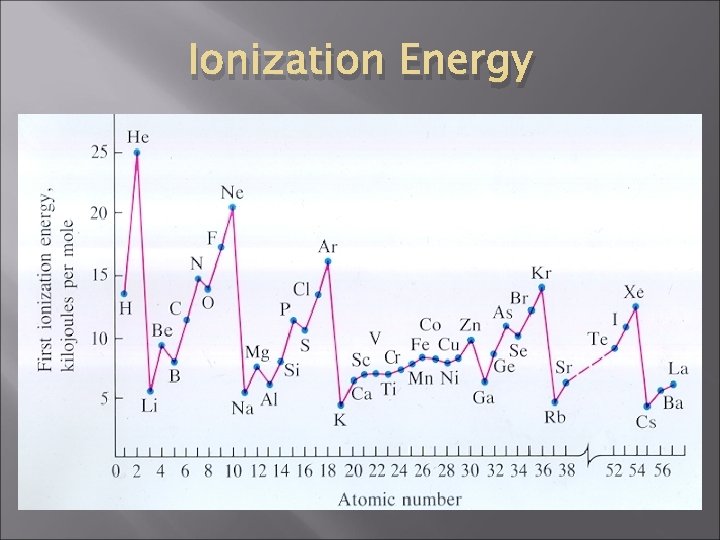
Ionization Energy

Electronegativity (E- affinity) Electronegativity is the attraction of an atom for ein the covalent bond. The most EN element is fluorine(F) with a value of 4. 0 As we go across the P. T. , EN goes up, and as we go down the P. T. , EN goes down.
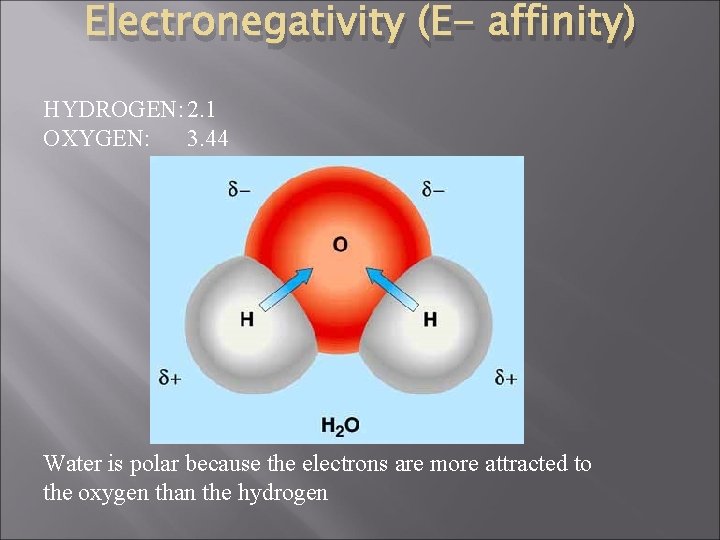
Electronegativity (E- affinity) HYDROGEN: 2. 1 OXYGEN: 3. 44 Water is polar because the electrons are more attracted to the oxygen than the hydrogen

Atomic Radius � � Atomic radius is the distance between the nucleus of an atom and its valence shell when that atom has formed a covalent bond. Size of atomic radius determined by: 1. 2. � Nuclear charge (number of protons) Shielding of outer shell electrons by inner shell electrons Across PT, radius decreases because electrons are more strongly attracted to the nucleus and shielding remains same.
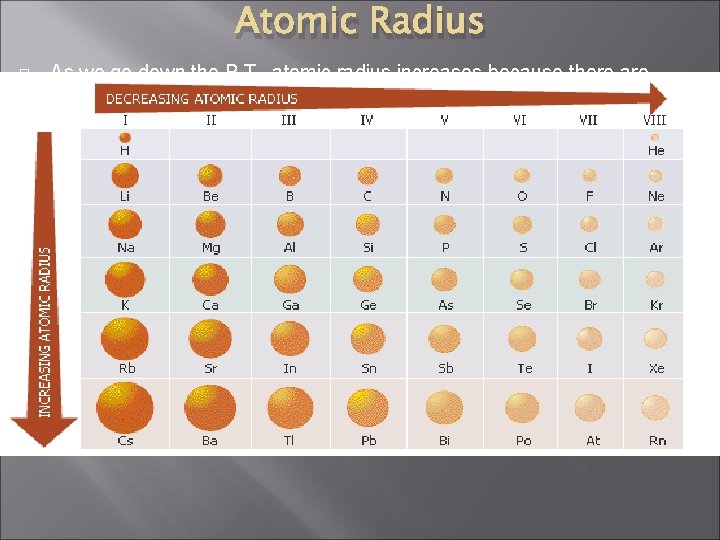
Atomic Radius � As we go down the P. T. , atomic radius increases because there are more energy levels � As we go across the P. T. , atomic radius decreases because the atoms are getting smaller (more p+, stronger attraction of electrons)
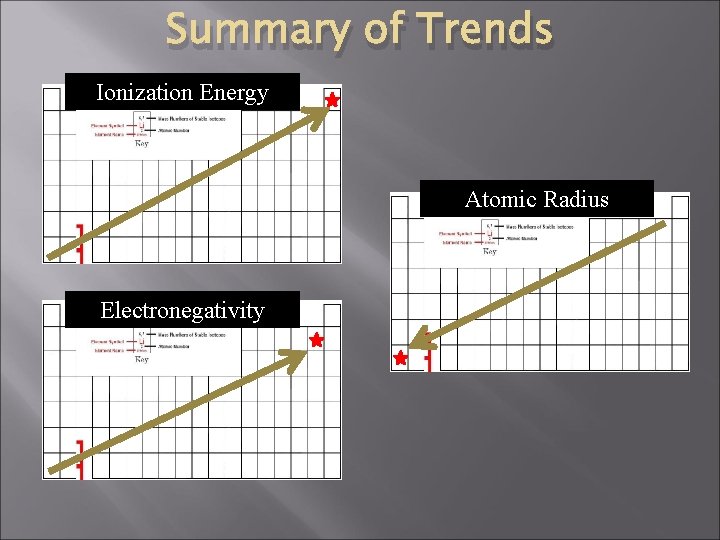
Summary of Trends Ionization Energy Atomic Radius Electronegativity

The End!
- Slides: 28