Periodic Table Organization Discovering Elements In 1750 only
Periodic Table: Organization

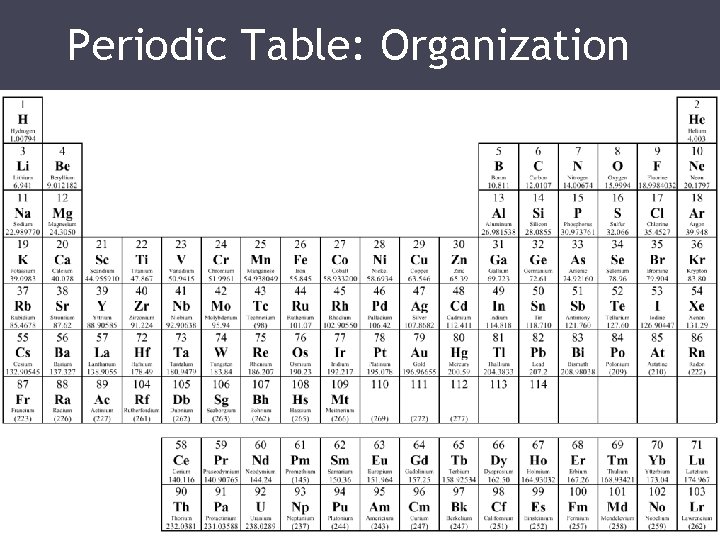
Discovering Elements • In 1750, only 17 elements had been discovered by scientists. • Systematic thinking increased the number of the elements being discovered • In 1789, Antoine Lavoisier grouped the elements into categories that called: metals, nonmetals, gases, and earths.

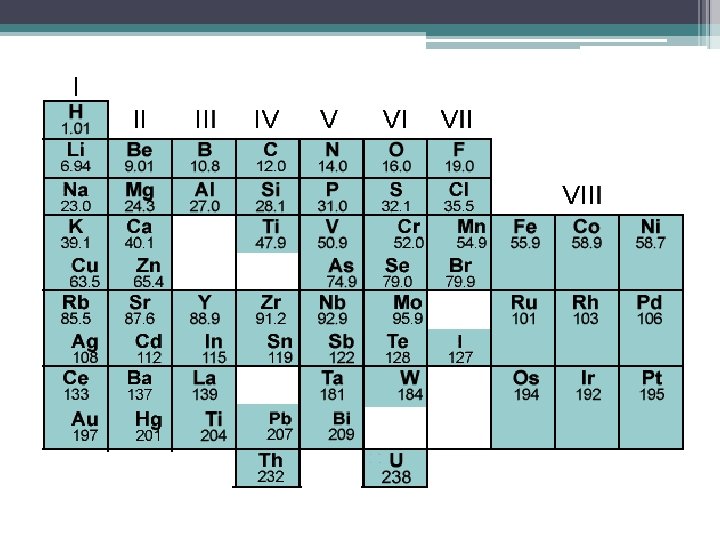
Mendeleev’s Periodic Table • He needed a way to organize the 63 elements because we writing a textbook for chemistry students. • His idea stemmed from his favorite card game, a version of solitaire. ▫ Cards must be sorted by suit and value

Mendeleev’s Periodic Table • Proposal ▫ Created a “deck of cards” of the elements ▫ Each card contained the elements mass, name and properties • He arranged the elements into rows in order of increasing mass so the elements with similar properties were in the same column. ▫ Final arrangement was similar to the wining hand of solitaire except that the columns were properties and not suits. ▫ Within a column, the mass increased from top to bottom • Periodic table is an arrangement of elements in columns based on a set of properties that repeat from row to row.
Mendeleev’s Periodic Table

Questions • Describe how Mendeleev organized the elements into rows and columns in his periodic table. • What characteristics of solitaire did Mendeleev use as a model for his periodic table? • Why did Mendeleev leave spaces in his table? • Define periodic table.
- Slides: 6