Periodic Table Organization and Trends Periodic Trends If

Periodic Table Organization and Trends

Periodic Trends If you look at the Periodic Table, you will notice several trends… take a look at it now (pg. 172 in your text) and see if you can notice any
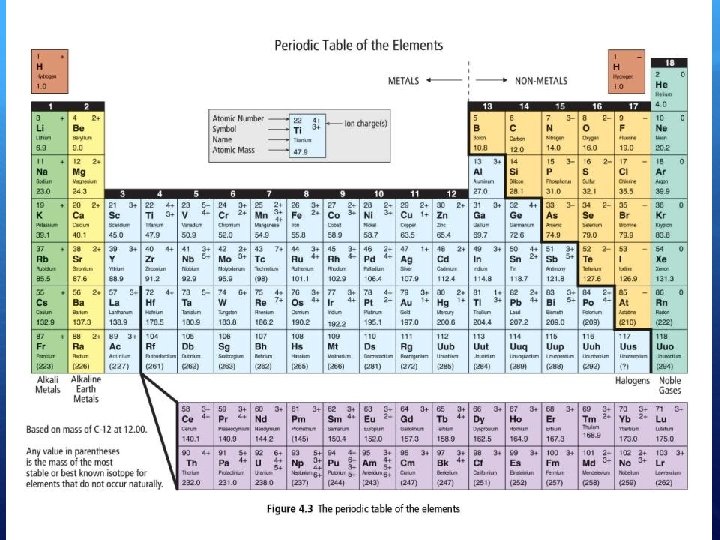

Color-coded to show similar elements Alkali metals Ex: Sodium (Na) Shiny, soft, silvery, highly reactive metals Alkaline earth metals Ex: Calcium (Ca) Shiny, silvery-white, somewhat reactive metals Transition metals Ex: Copper (Cu) Hard, high BP and MP, barely reactive metals Halogens Highly reactive non-metals Ex: Flourine (F) Noble gases Ex: Helium (He) Odorless, colorless, very non-reactive non-metals
Atomic number increases L R, top to bottom
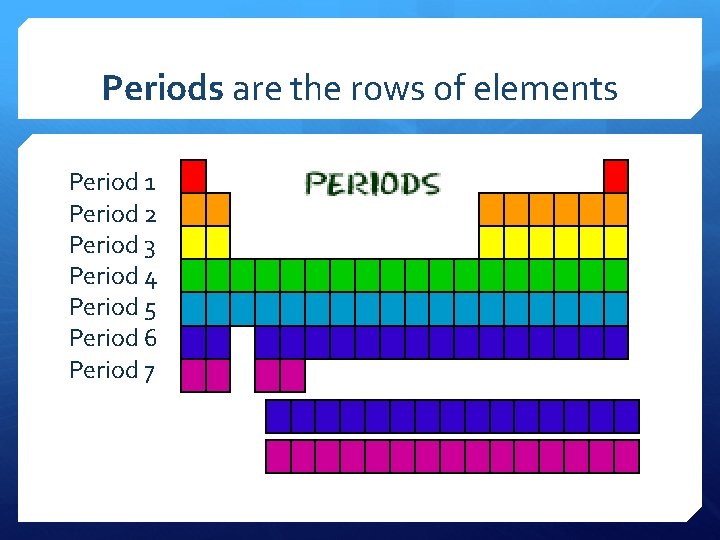
Periods are the rows of elements Period 1 Period 2 Period 3 Period 4 Period 5 Period 6 Period 7
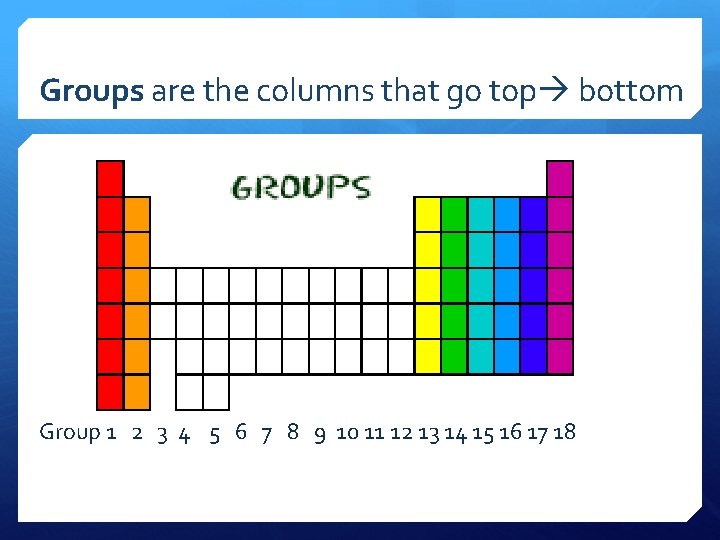
Groups are the columns that go top bottom Group 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18

Metal elements decrease in reactivity from L R; Non-metals increase in reactivity from L R Less Reactive R (Halogens are very non reactive, and are not shown LR here) Most Reactive | Least Most Reactive

Organization Summary Elements are listed by their atomic number from 1 – 118 from LEFT to RIGHT, top to bottom PERIODS = the rows that go from L R Group/ Family = the columns that go from TOP BOTTOM Reactivity ranges from very reactive (L side) non- reactive (R side) for both metals and non-metals

Element’s state of matter Elements can exist in various states of matter (solid, liquid, gas)
- Slides: 10