Periodic Table of Elements l Bohr Model and

Periodic Table of Elements

l Bohr Model: and Valence electrons http: //youtu. be/trsln 8 RCEVo l Casseopia http: //youtu. be/5 MMWpe. J 5 dn 4 l l l Dan Radcliffe http: //youtu. be/r. SAai. YKF 0 cs Bald Guy: http: //youtu. be/nsb. Xp 64 YPRQ l Song: http: //youtu. be/af. Fw 91 fv. NJM l http: //youtu. be/z. GM-w. SKFBpo l

Dmitri Mendeleev l Russian chemist. l Created first version of the periodic table. l Predicted properties of elements yet to be discovered. Rockin the shaggy beard…

l Mendeleev arranged elements according to their atomic weights. l Today they are arranged by atomic number.

Periodic Law l Properties of elements are periodic functions of their atomic numbers. l In other words, properties tend to repeat as you start a new period.

Groups/Families l Columns on the table (1 -18). l Elements have the same number of valence electrons. l Similar chemical properties. l Tend to form bonds in a similar way to get stable octet.
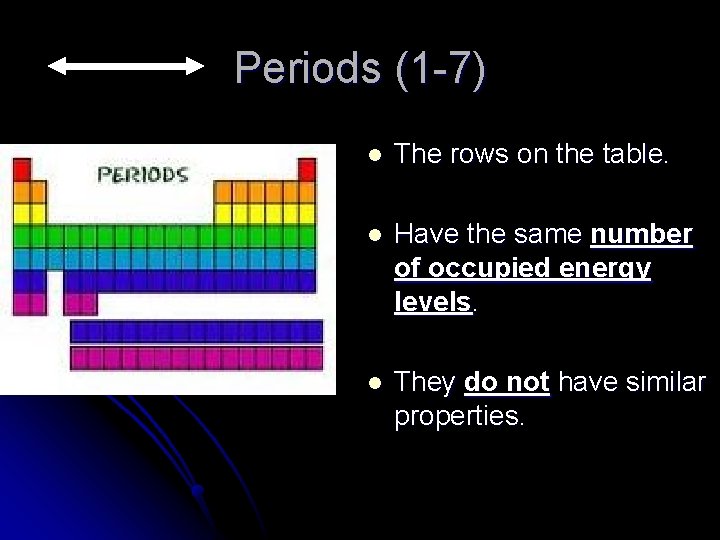
Periods (1 -7) l The rows on the table. l Have the same number of occupied energy levels. l They do not have similar properties.

Get to Know Your Groups

Alkali Metals l l 1 valence electron Form +1 Ions Very reactive Found only in compounds.

Rubidium

Comparing the Alkali Metals http: //youtu. be/uixx. Jt. JPVXk

Alkaline Earth Metals l l 2 valence electrons Form +2 ions Very reactive Found only in compounds


Halogens l l 7 valence electrons Form -1 ions Very reactive All nonmetals

Gas Liquid FLUORINE Extremely reactive gas Solid IODINE solid → gas (sublimes)

Noble Gases Inert Gases l Not reactive l Have stable octet (except He) l Monoatomic gases l

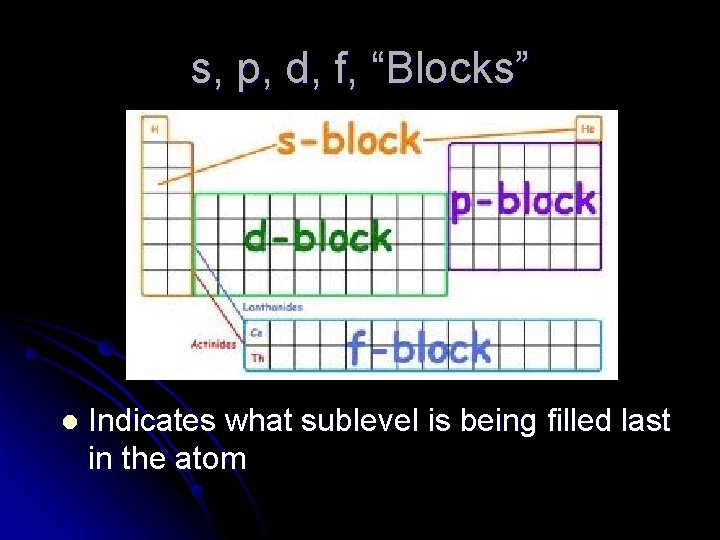
s, p, d, f, “Blocks” l Indicates what sublevel is being filled last in the atom
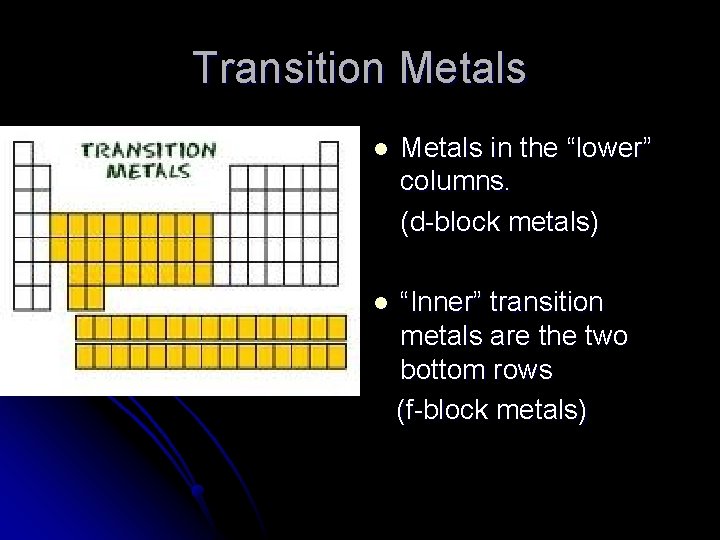
Transition Metals l Metals in the “lower” columns. (d-block metals) l “Inner” transition metals are the two bottom rows (f-block metals)

Transition Metals l Form colored compounds and solutions.
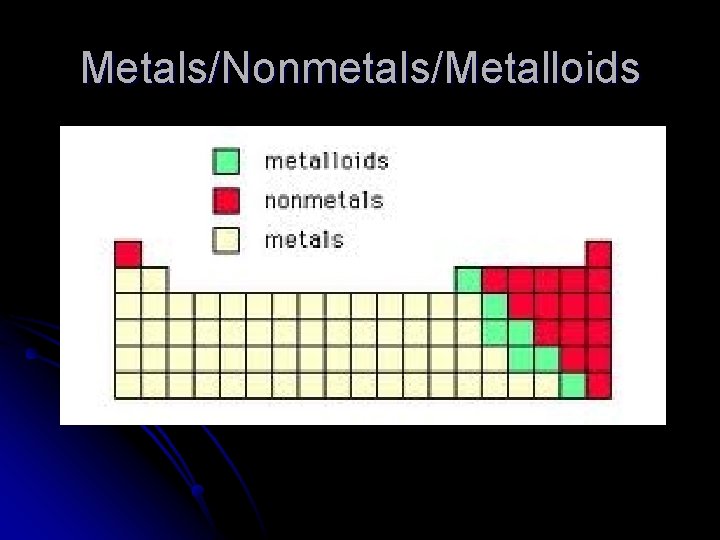
Metals/Nonmetals/Metalloids

Metals Have luster l Malleable l Ductile l Conduct heat and electricity l Tend to lose valence electrons and form positive ions l All solids (except Hg) l


Nonmetals l l l No luster Brittle (if solid) Don’t conduct Tend to gain valence electrons and form negative ions Can be solid ( I, C, P, S) , liquid (Br), or gases (F, Cl, N, O) Hydrogen is considered a nonmetal

Metalloids (Semimetals) l Most elements along the “staircase” l Can have properties of both metals and nonmetals. l B, Si, Ge, As, Sb, Te, At

Trends in the Period Table

Atomic Radius l Distance from the nucleus of an atom to the outer edge of it’s electron cloud.

Get out Reference Table S

Trends in Radius l Down a Group Li Na K Ex: Group 1 What happens? Rb Cs l Fr Across a Period Ex: Period 2 Li Be B What happens? C N O F Ne

l Down a Group l Size increases l Why? You add new energy levels l Across a Period l Size decreases l Why? Increasing nuclear charge pulls on the energy levels

http: //youtu. be/ba 2 y. N 2 Ht. PTA
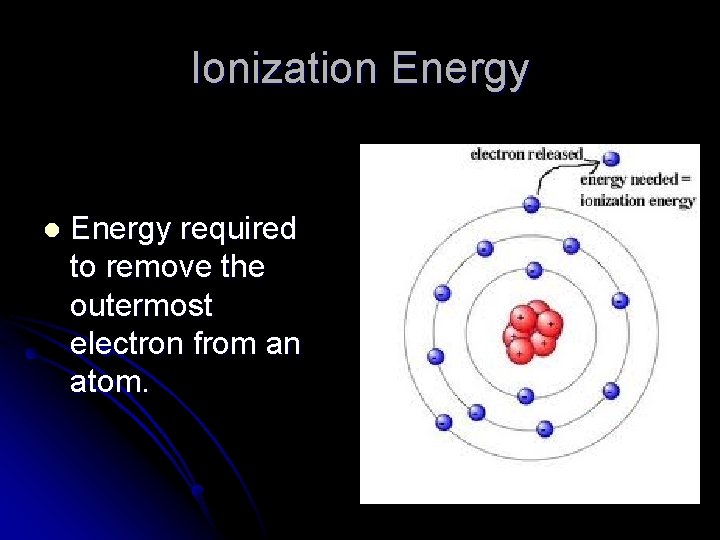
Ionization Energy l Energy required to remove the outermost electron from an atom.

Get out Reference Table S

Trends in Ionization Energy l Down a Group Ex: Group 1 What happens? l Li Na K Rb Cs Fr Across a Period Ex: Period 2 Li Be B C What happens? N O F Ne

What is the trend? Directly related to Atomic Radius. l Large Radius = Lower Ionization Energy Why? The further the outmost electron to the nuclear pull the easier to remove it.

l http: //youtu. be/8 F 9 n. A 4 Fg 3 Rw

Electronegativity l The relative attraction an atom has for electrons involved in bond formation.

l The higher the EN value the more the atom “pulls” on electrons involved in a bond. l This can result in “polar” and “nonpolar” bonds.

l Fluorine has the highest EN value = 4 (The EN scale was created by comparing other elements to Fluorine)

Get out Reference Table S

Trends in Electronegativity l Down a Group Ex: Group 1 What happens? l Li Na K Rb Cs Fr Across a Period Ex: Period 2 Li Be B C What happens? N O F Ne

l http: //youtu. be/93 G_Fqp. GFGY
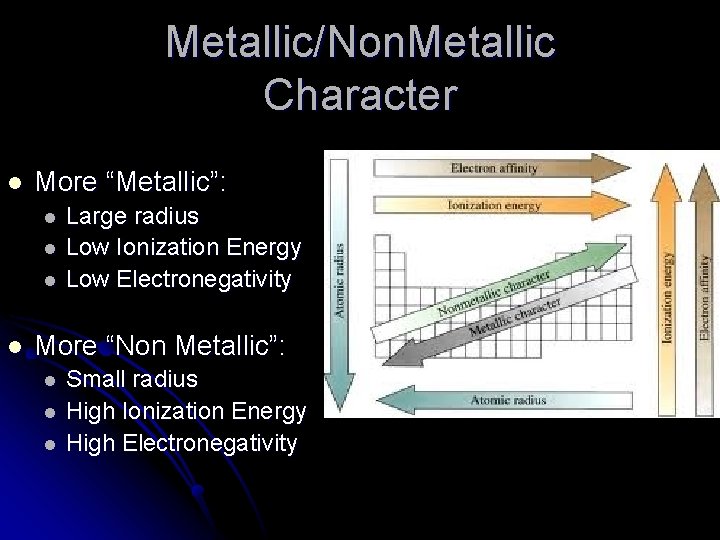
Metallic/Non. Metallic Character l More “Metallic”: l l Large radius Low Ionization Energy Low Electronegativity More “Non Metallic”: l l l Small radius High Ionization Energy High Electronegativity
- Slides: 43