Periodic Table of Elements https www youtube comwatch

Periodic Table of Elements https: //www. youtube. com/watch? v=z. GM-w. SKFBpo https: //www. youtube. com/watch? v=z. UDDi. Wt. Ft. EM

nitrogen chlorine gold helium silver oxygen mercury hydrogen sodium niobium neodymium carbon

Elements § Science has come along way since Aristotle’s theory of Air, Water, Fire, and Earth. § Scientists have identified 90 naturally occurring elements, and created about 28 others.

Elements § The elements, alone or in combinations, make up our bodies, our world, our sun, and in fact, the entire universe.

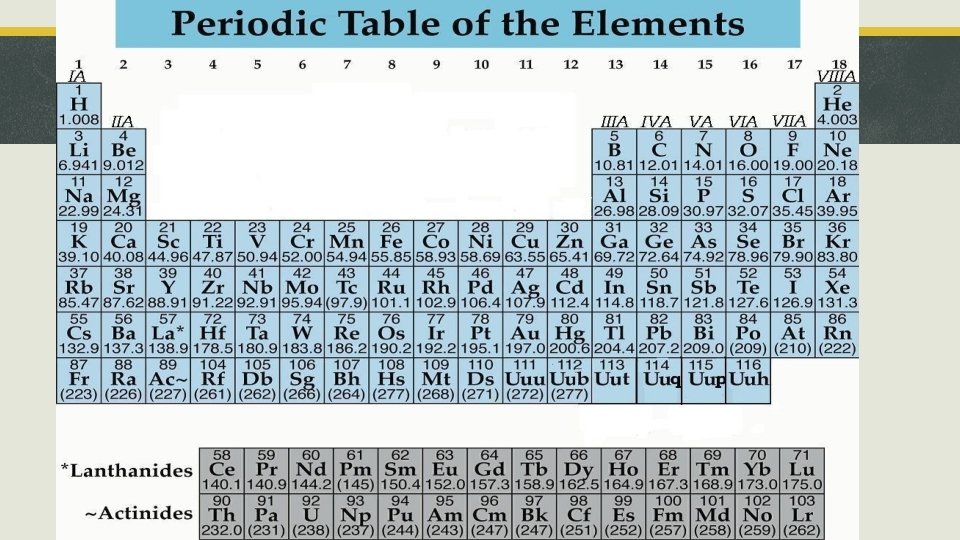
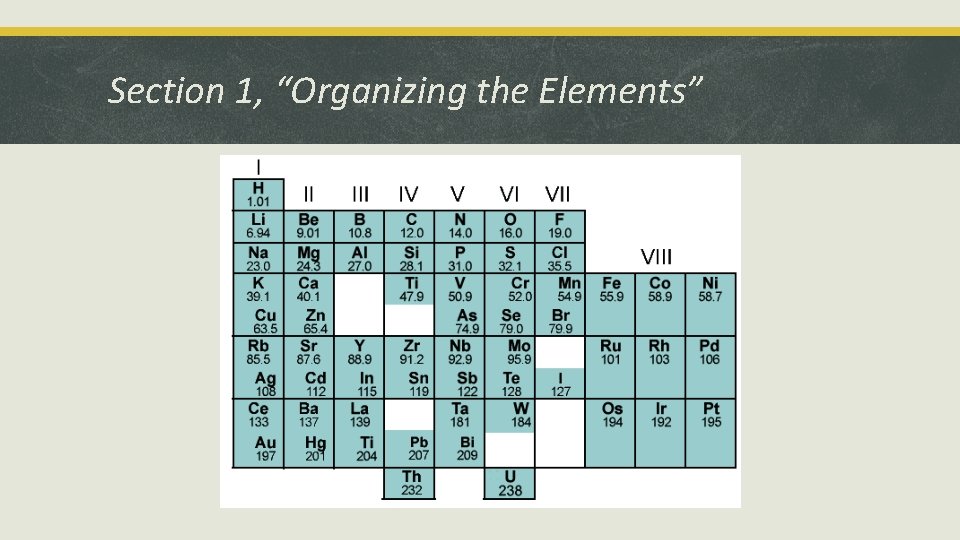
Section 1, “Organizing the Elements” § Dmitri Mendeleev created the periodic table. § He arranged elements in rows by increasing atomic mass.
Section 1, “Organizing the Elements”

Section 1, “Organizing the Elements” § The modern periodic table is arranged by atomic number. § Periodic law states that the repeating chemical and physical properties of elements change periodically with the atomic numbers of the elements.

Section 2, “Exploring the Periodic Table” § The periodic trends in the periodic table are the result of electron arrangement. The energy level can be determined depending on the period the element is in and the orbital can be determined depending on its location in the table.
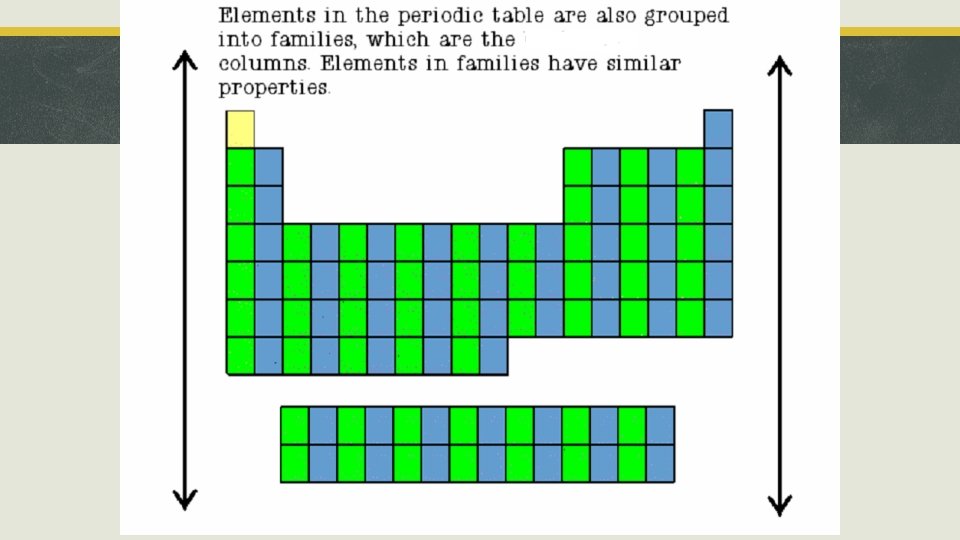


Section 1, “Organizing the Elements” § When arranged by atomic number, all elemental properties aligned in columns called groups. § § Atomic size increases as you go from top to bottom of the periodic table. Reactivity increases as you go from top to bottom.
How many groups are on the Periodic Table? 18
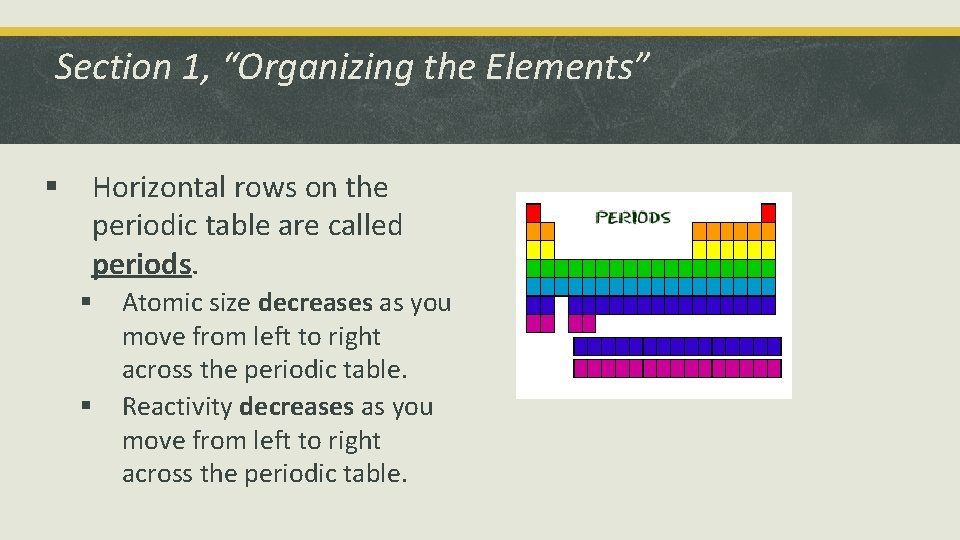
Section 1, “Organizing the Elements” § Horizontal rows on the periodic table are called periods. § § Atomic size decreases as you move from left to right across the periodic table. Reactivity decreases as you move from left to right across the periodic table.

How many periods are on the Periodic Table? 7
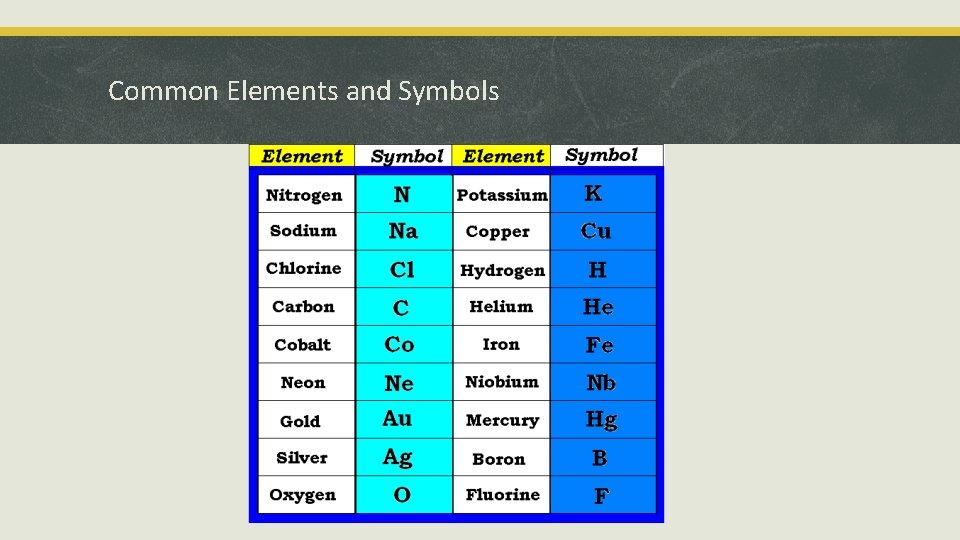
Common Elements and Symbols

Section 1, “Organizing the Elements” § The number of valence electrons accounts for many of the chemical properties of an element. This can be easily determined using the periodic table.
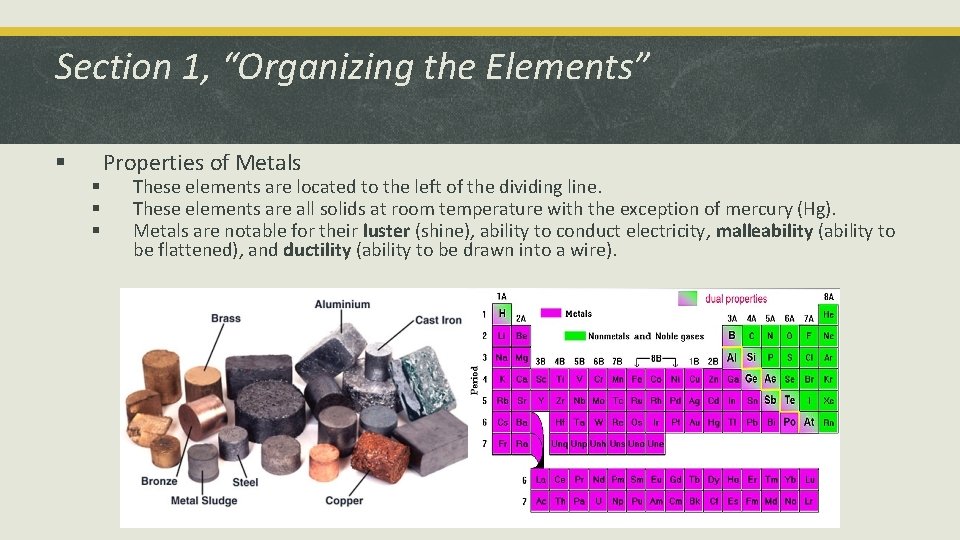
Section 1, “Organizing the Elements” § § Properties of Metals These elements are located to the left of the dividing line. These elements are all solids at room temperature with the exception of mercury (Hg). Metals are notable for their luster (shine), ability to conduct electricity, malleability (ability to be flattened), and ductility (ability to be drawn into a wire).




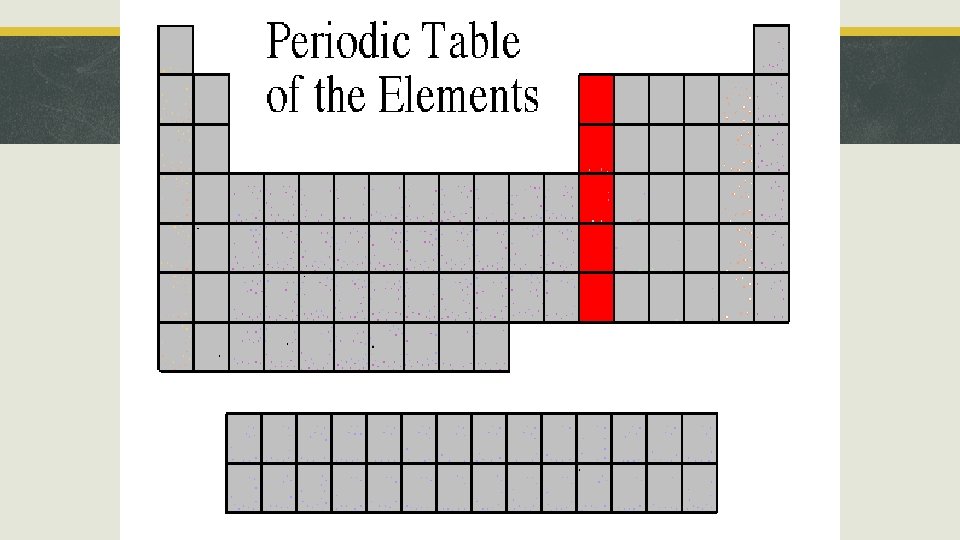




Section 3, “Families of Elements” § Alkali Metals § § § Location: Group 1 Ionic Charge: +1 Properties: soft, shiny, reacts violently with water. https: //www. youtub e. com/watch? v=RAF c. Zo 8 d. Tc. U Li, Na, K, Rb, Cs, Fr

Alkali Metals Lithium, Li Potassium, K Cesium, Ce Rubidium, Rb Francium, Fr

Section 3, “Families of Elements” § Alkaline Earth Metals § Location: Group 2 § Ionic Charge: +2 § Properties: Are harder, denser, stronger, and have higher melting points than alkali metals. § Examples: Be, Mg, Ca, Sr, Ba, Ra

Alkaline Earth Metals

Section 3, “Families of Elements” § Transition Metals § Location: Groups 3 -12 § Properties: Less reactive than alkali or alkalineearth metals. Are harder, more dense, and have higher melting points than the other two metals.

Precious Metals § The transition metals include precious metals such as copper (Cu), silver (Ag), gold (Au), and platinum (Pt).

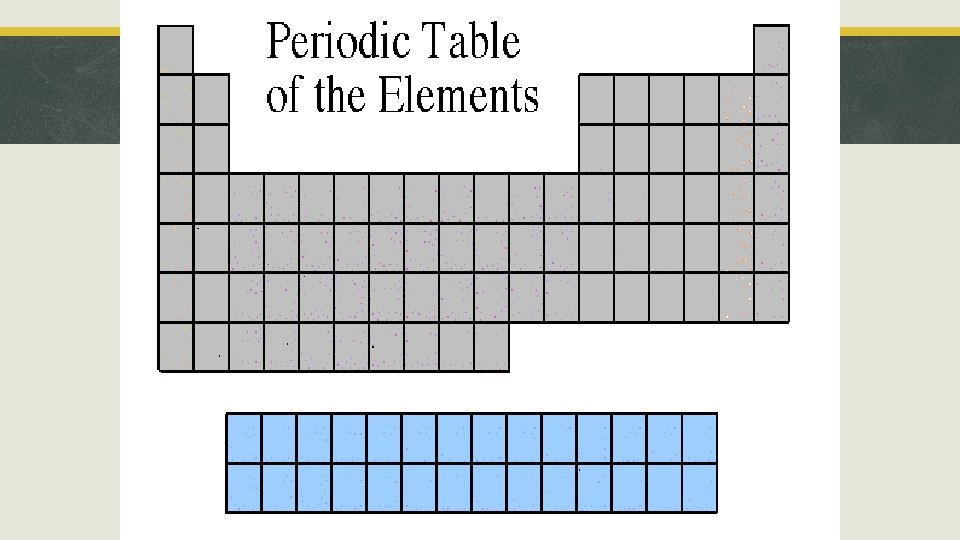
Section 3, “Families of Elements” § Lanthanide and Actinide Series § § Location: Within Periods 6 and 7. Represented at the bottom of the periodic table. Properties: Share properties with transition metals and are radioactive. Many are synthetic (made in a lab).

Section 1, “Organizing the Elements” § Properties of Metalloids or Semiconductors § Have both metallic and nonmetallic properties. § Able to conduct heat and electricity under certain conditions § These are solid at room temperature. § They are located between the metals and nonmetals and straddle the diagonal dividing line. § Metalloids are useful as electronic semiconductors. § Elements: B, Si, Ge, As, Sb, Te

Metalloids Tellurium, Te Arsenic, As Germanium, Ge Boron, B Antimony, Sb Silicon, Si Polonium, Po

Section 1, “Organizing the Elements” § § § Properties of Nonmetals These elements are located to the right. Nitrogen (N), oxygen (O), fluorine (F), chlorine (Cl), and the noble gases (in the last column) are gases at room temperature. Bromine (Br) is a liquid while all other nonmetals are solid. Nonmetals do not conduct electricity. Elements are brittle
Section 3, “Families of Elements” Boron, Carbon, Nitrogen, and Oxygen Families § § These families typically aren’t singled out because they are have a mix of nonmetals, metalloids, and metals.

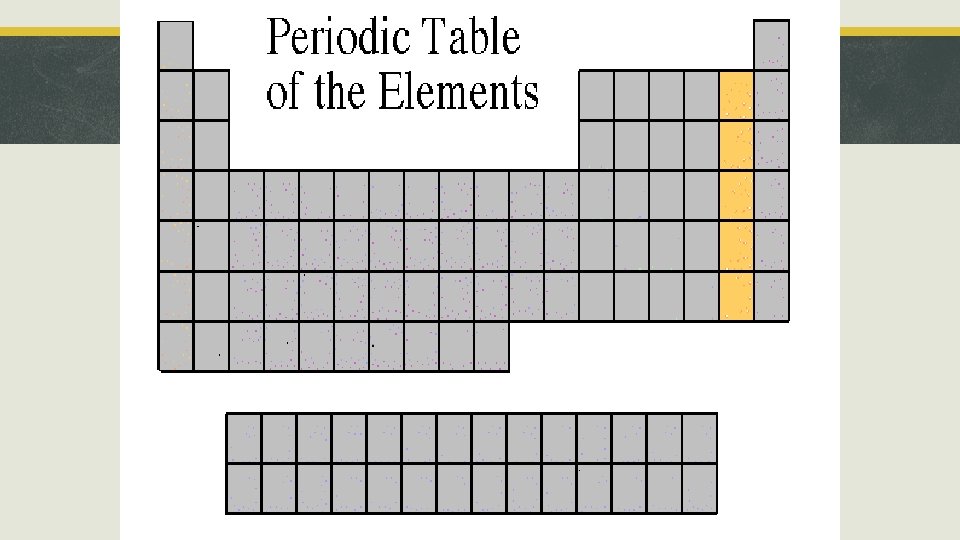
Section 3, “Families of Elements” § Halogens § § § Location: Group 17 Ionic Charge -1 Properties: Most reactive nonmetals. Combine with metals to make salts. Examples: F, Cl, Br, I, At Bromine=Liquid non-metal

Halogens Bromine, Br Iodine, I Fluorine, F Astatine, At Chlorine, Cl

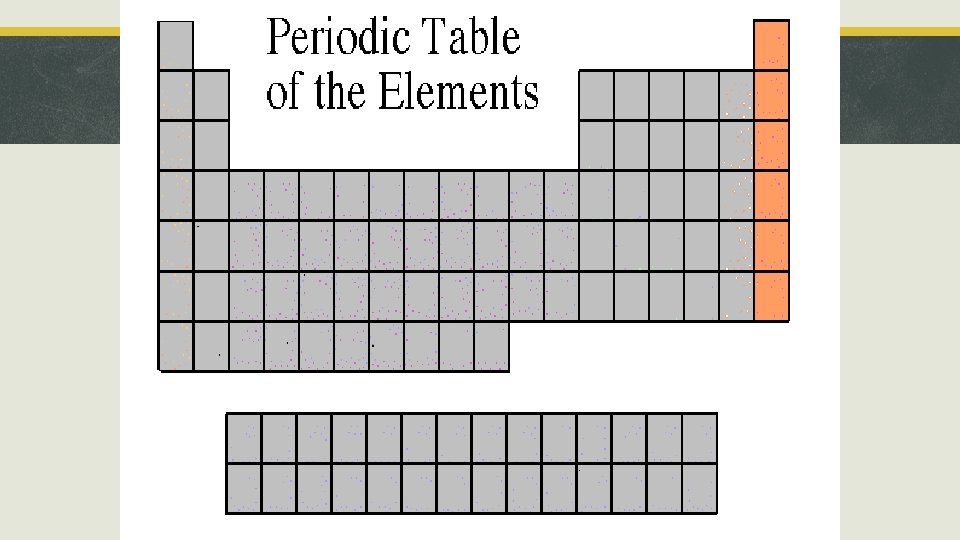
Section 3, “Families of Elements” § Noble Gases § Location: Group 18 § Ionic Charge: Will not ionize! § Properties: inert (non reactive), gaseous elements. § Examples: He, Ne, Ar, Kr, Xe, Rn
Noble Gases Neon, Ne Krypton, Kr Helium, He Xenon, Xe Argon, Ar Radon, Ra

Section 3, “Families of Elements” § Hydrogen is classified by itself because it does not behave like any of the other elements. It is the lightest of all elements, a gas at its normal state, and highly flammable.

Lewis Structures § Show the valence electrons for each atom. § Look at the Group # to HELP you determine valence electrons § Place “dots” around the element symbol to represent the valence electrons. N § We will cover this next week in a Bohr and Lewis Structure Lesson. 5 Valence Electrons
- Slides: 47