Periodic Table of Elements Elements Scientists have identified
Periodic Table of Elements
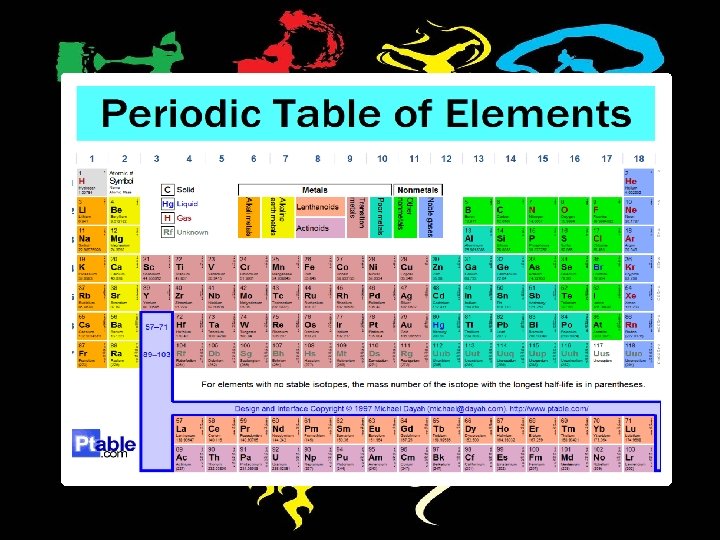

Elements *Scientists have identified 90 naturally occurring elements and created about 28 others.

The Elements, alone or in combinations, make up our bodies, our sun and in fact the entire universe.


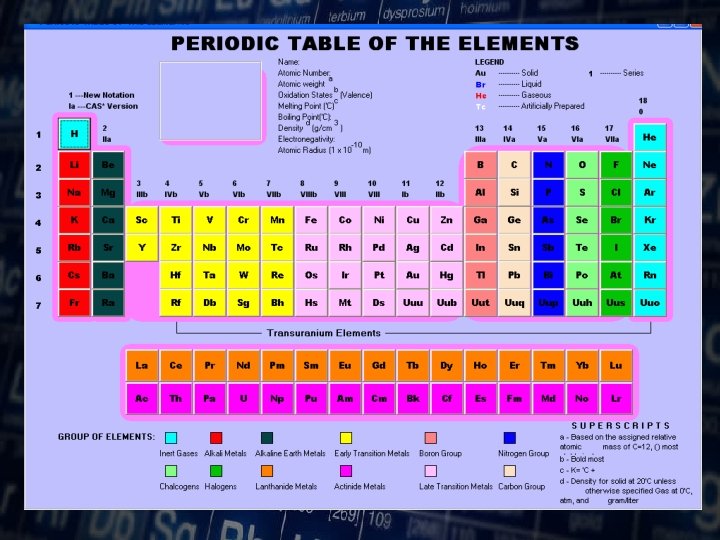
The periodic table organizes the elements in particular way. A great deals of information about an element can be gathered from its position in the periodic table

Elements are organized on the table according to their atomic number. v The atomic number refers to how many proton an atom of that element has. v The atomic number is unique to that element. No two elements have the same atomic number.
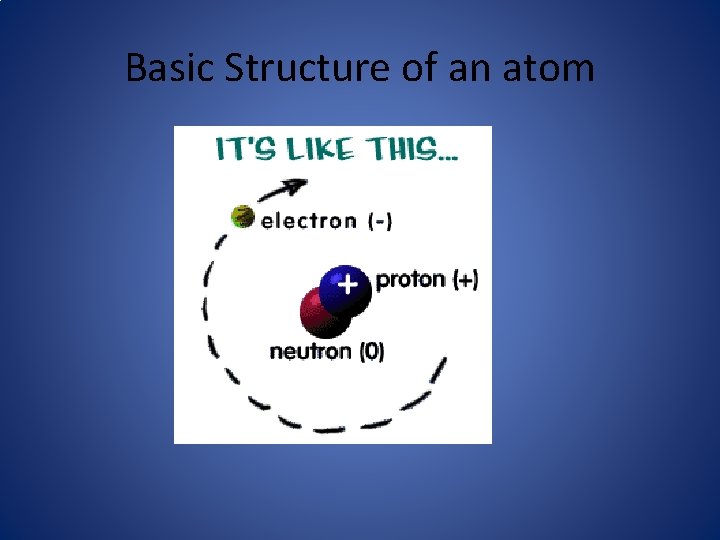
Basic Structure of an atom
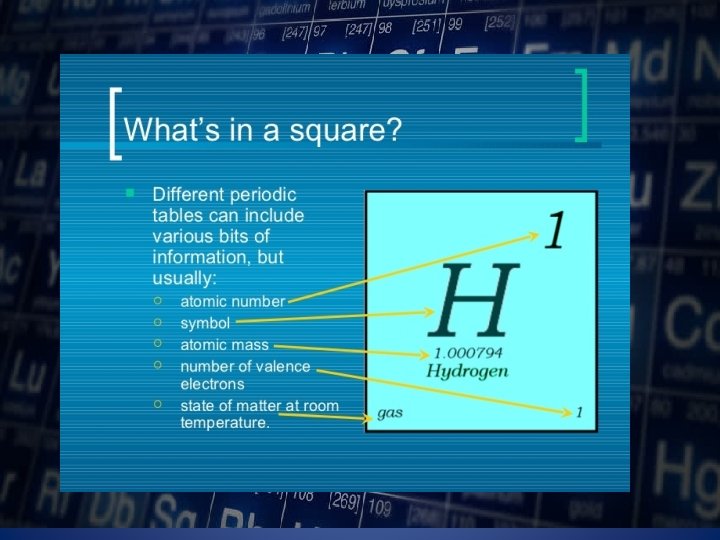

Atomic Number ØThis refers to how many proton an atom of that element has. ØNo two elements, have the same number of protons.
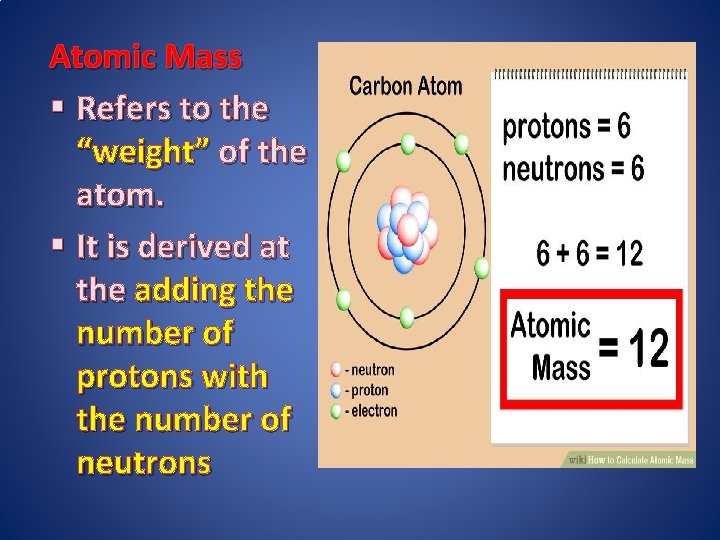
Atomic Mass § Refers to the “weight” of the atom. § It is derived at the adding the number of protons with the number of neutrons

Hydrogen • The Hydrogen square sits a top of family Al, but it is not a member of that family. • Hydrogen is in a class of it’s own. • It’s a gas at room temperature. • Hydrogen is a diatomic, reactive gas. • It has one proton and one electron in its one and only energy level • Hydrogen only needs 1 electrons to fill up its valence shell

Development of the Periodic table

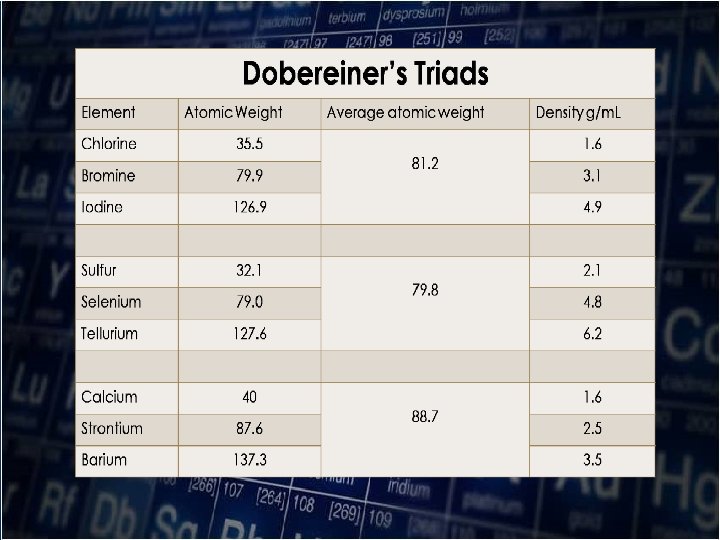
DOBEREINER’S TRIAD In 1829, the German chemist Johann Wolfgang Dobereiner(1780 -1849) classified the elements with similar properties into groups of three called “TRIAD”

Johann Wolfgang Dobereiner (1780 -1849)

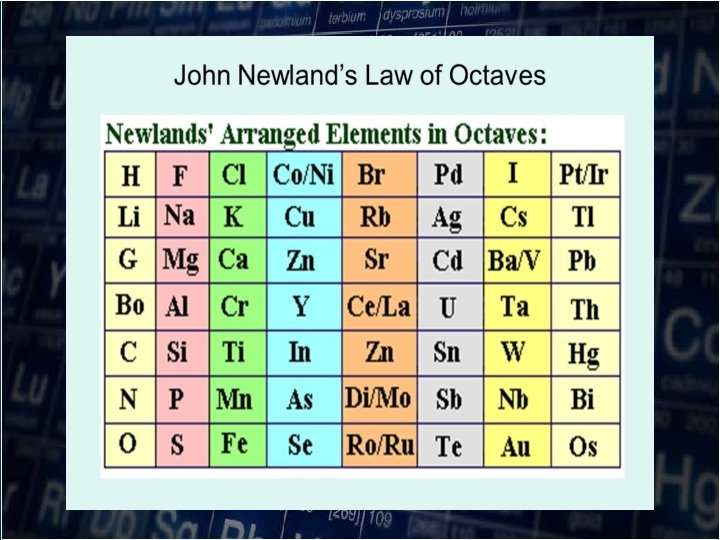
Newland’s LAW OF OCTAVES In 1864, an English Industrial chemist JOHN NEWLANDS(18371898) arrange the elements in increasing atomic weights. He noted that every eight element has similar properties

John Newland (1837 -1898)

Mendeleev’s Periodic Table • In 1869, the Russian chemist Dmitri Mendeleev (1834 -1907) published his arrangement of the known element in vertical columns. He noticed a regular (periodic)recurrence of physical and chemical properties. (Periodic Law)

Dmitri Mendeleev (1834 -1907)
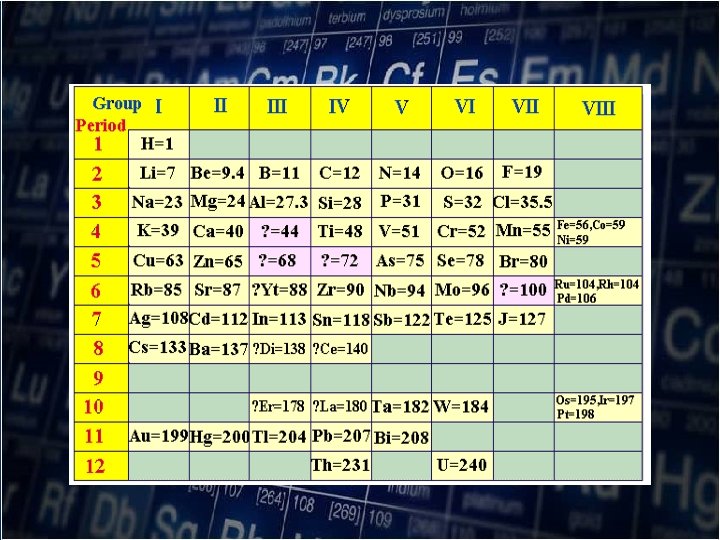

1 st he left spaces in his periodic table for undiscovered element 2 nd he corrected some atomic weight values of element

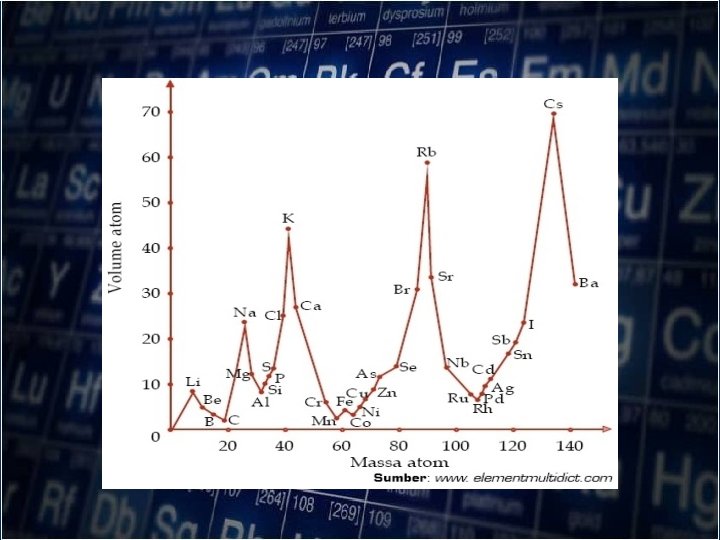
The German chemist , Lothar Meyer(1830 -1895) prepared a periodic table that closely resembles the tale of Mendeleev. But presented his results graphically

Lothar Meyer (1830 -1895)

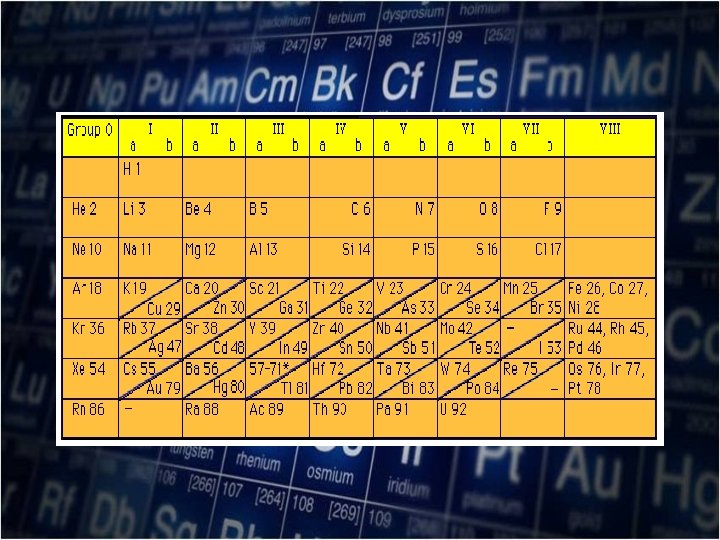
In 1913, Henry Mosely (18871915) a young British physicist determined the atomic number of the element. Mosely findings led to the modification of the periodic table by order of increasing Atomic number.

Henry Mosely (1887 -1915)
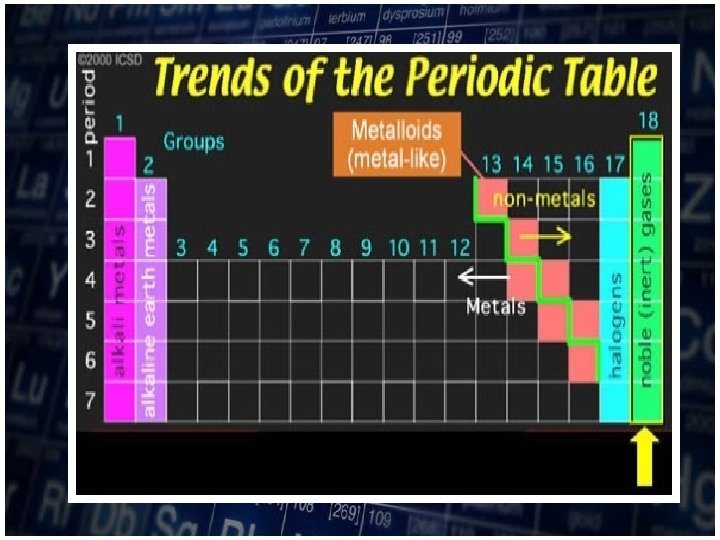
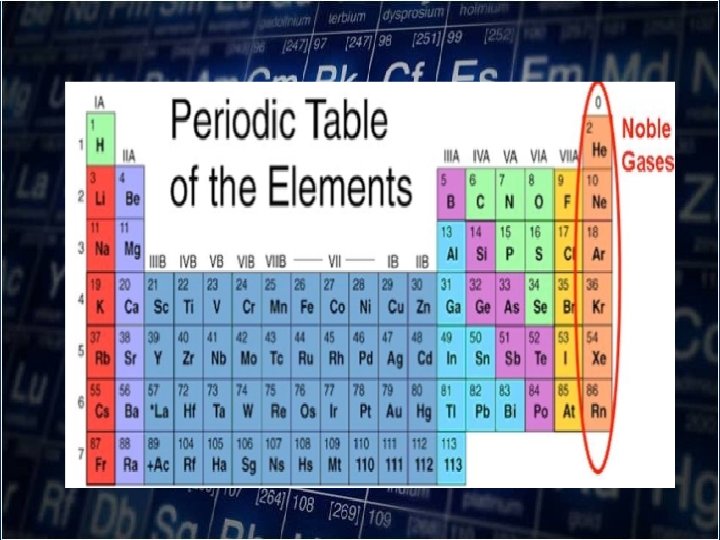
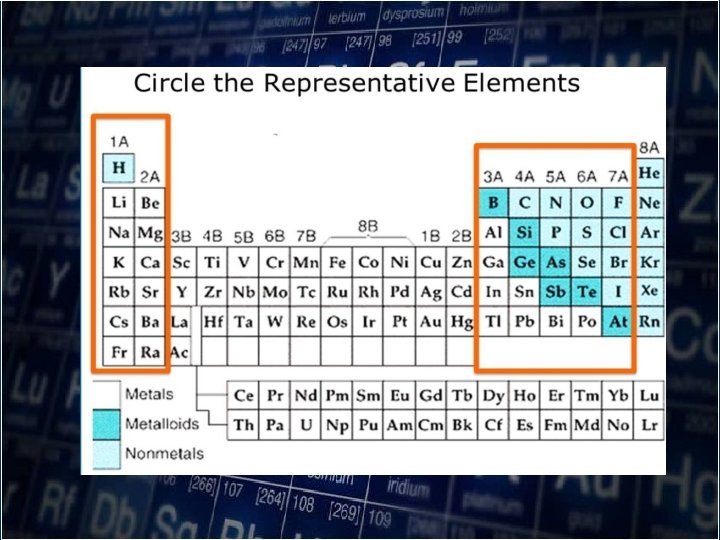
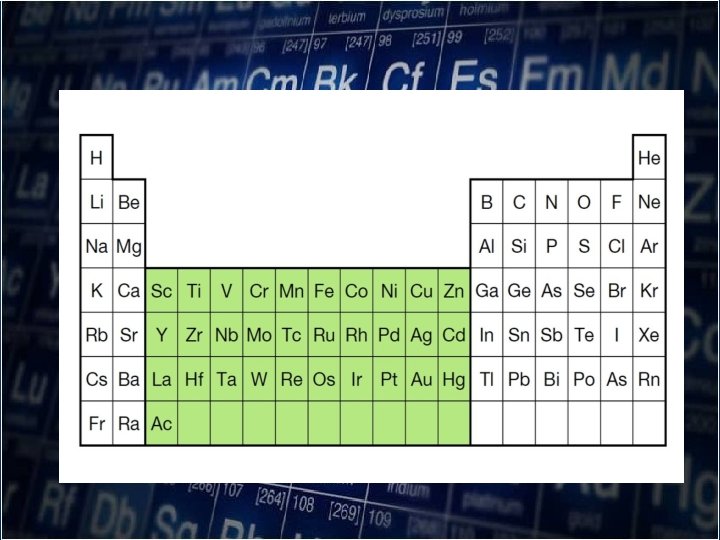
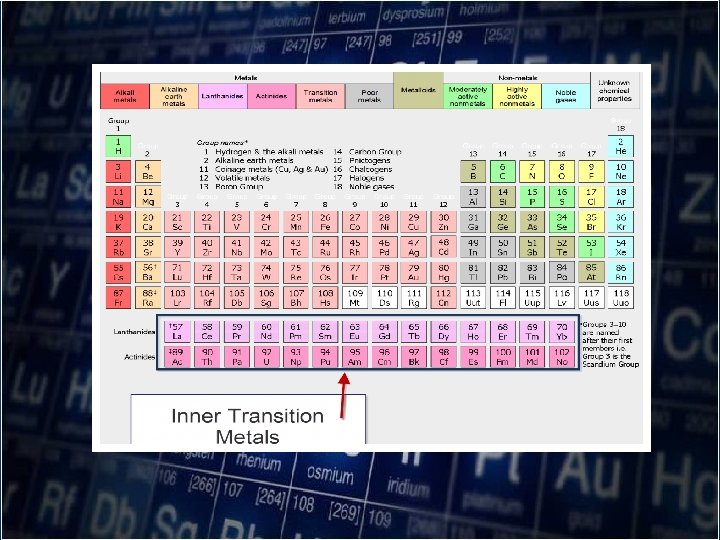


• As you move down a group, atomic radii increase as electrons are added to successively higher energy level. • The size of the orbitals also increases with the number of energy level.

Ionization Energy -when the atom loses or gains electron from ions.
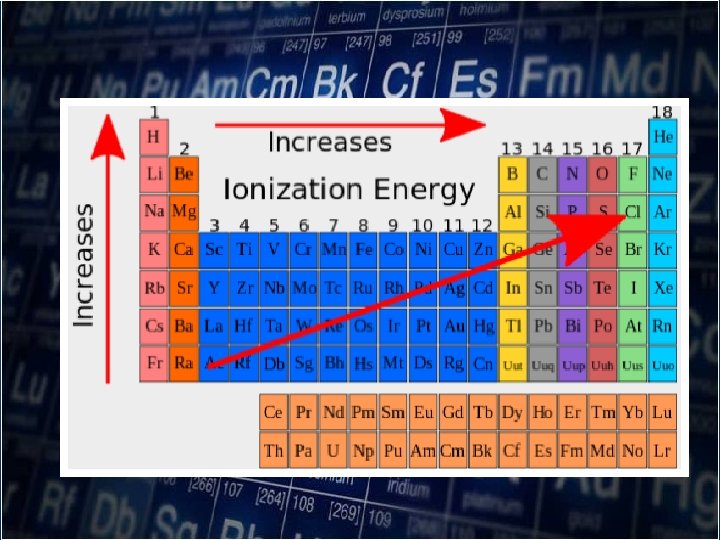

• Ionization energies measure how tightly bound of the electrons are to atoms. • The representative elements, the first ionization energy generally increases from left to right across the period

Remember: Ionization energy is always positively. Low ionization energy indicates easy removal of electrons to become cation

• Small atoms have high ionization energies while big atoms have low ionization energies

Electron Affinity(AE) -the amount of energy released when an electron is added to a gaseous atom
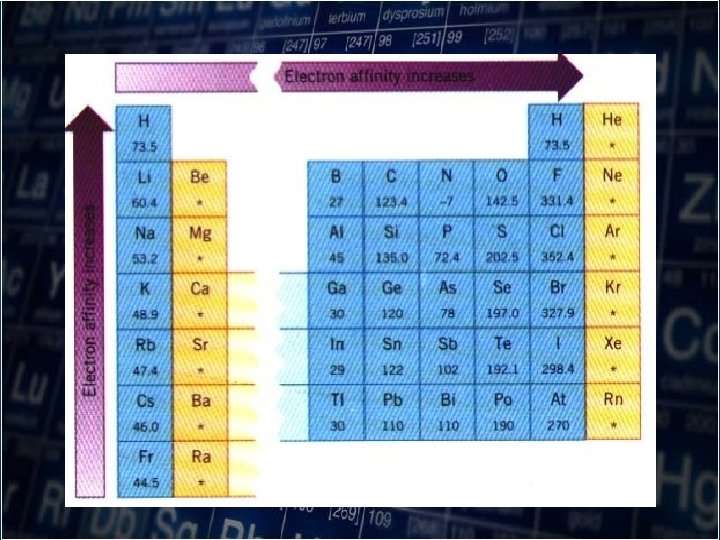

• Elements with the very negative electron affinities gain electrons readily to form negative ions. • The elements show greater attraction for an extra electron from the left to right across the row in the periodic table, excluding noble gases

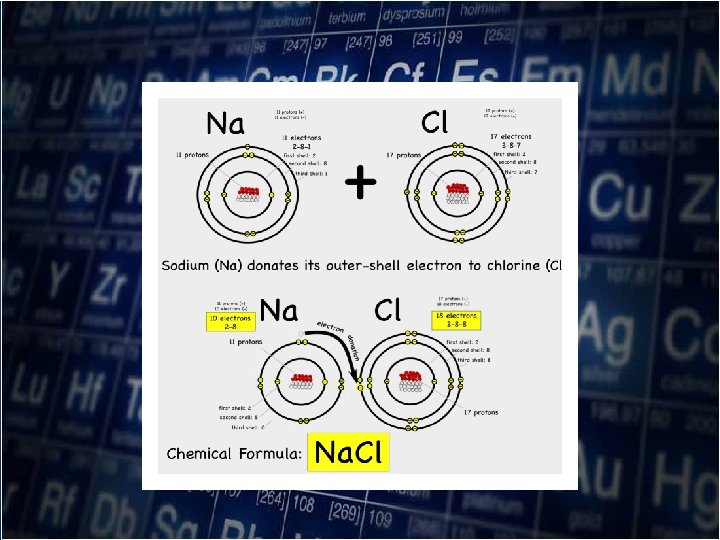
• The octet rule states that atoms tend to gain , lose, or share electrons in order to acquire a full setoff valence electrons



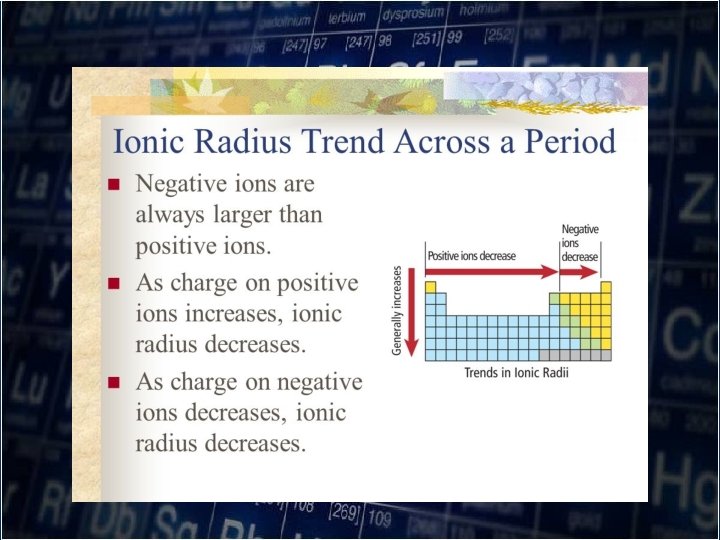
Ionic Radius -Metals have low ionization energies so they form positive ions readily. The loss of the outermost electrons results in increased attraction by the nucleus for the remaining electrons.

How to locate an element?



- Slides: 57