Periodic Table of Elements Dmitri Mendeleev l Created

Periodic Table of Elements

Dmitri Mendeleev l Created first version of periodic table. l Predicted properties of elements yet to be discovered. Ted Talk: https: //www. youtube. com/watch? v=O 48 zn. Ag 7 VE Rockin the shaggy beard…

l Arranged elements according to atomic weights. l Today they are arranged by atomic number. Development of the Table: 3: 39 http: //youtu. be/nsb. Xp 64 YPRQ

Periodic Law l “Properties of elements are periodic functions of their atomic numbers. ” l In other words, properties tend to repeat as you start a new period.

Groups/Families (Columns 1 -18) l Elements have: l l Same number of valence electrons. Similar chemical properties. l Form bonds in same way to get stable octet.
Periods (Rows 1 -7) l Elements have: l l Same number of occupied energy levels. Do not have similar properties.
Group 1: Alkali Metals l l l 1 valence electron Form +1 Ions Very reactive l Found only in compounds.

Rubidium http: //youtu. be/uixx. Jt. JPVXk http: //www. youtube. com/watch? feature=player_embe dded&v=HY 7 m. TCMvp. EM

Group 2: Alkaline Earth Metals l l l 2 valence electrons Form +2 ions Very reactive l Found only in compounds

Group 2: Some Alkaline Earth Metals

Group 17: Halogens l l 7 valence electrons Form -1 ions Very reactive All nonmetals

Gas Liquid FLUORINE Extremely reactive gas Solid IODINE solid → gas (sublimes) 3: 00 http: //www. youtube. com/watch? feature=player_embedded&v=u 2 og. MUDBaf 4
Group 18: Noble Gases l Inert Gases l l l Not reactive Have stable octet (except He) Monoatomic gases
Transition Metals l Metals in the “lower” columns. (d-block metals) l “Inner” transition metals are the two bottom rows (f-block metals)

Transition Metals Form colored compounds and solutions. l Often have multiple “oxidation states” (charges) l Honors: May involve d-sublevel electrons in forming bonds l
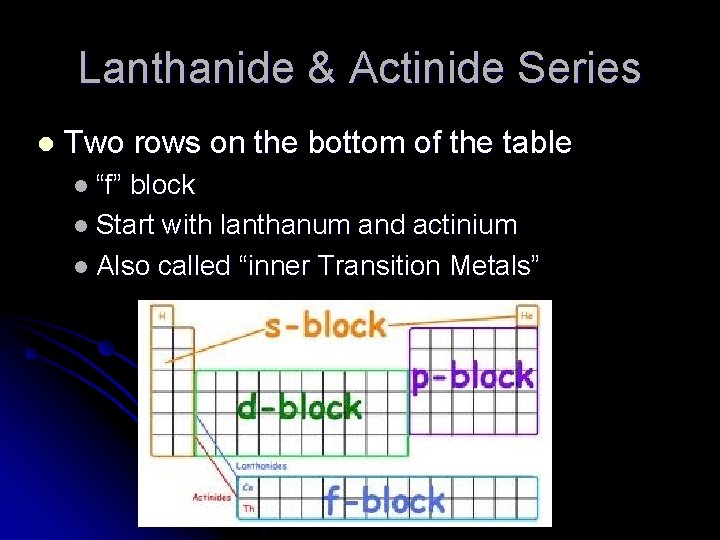
Lanthanide & Actinide Series l Two rows on the bottom of the table l “f” block l Start with lanthanum and actinium l Also called “inner Transition Metals”

l Man Made Elements: (“Trans Uranium”) Elements above Uranium (Atomic #92) l Not found naturally on earth l Must be created through nuclear bombardment using particle accelerators. l l Radioactive Elements: l All atoms of elements higher than Bismuth (Atomic #83) are radioactive.

l Do we know our groups? l Elements Song Again! l http: //youtu. be/z. GM-w. SKFBpo l l Periodic Table Dating Game http: //www. youtube. com/watch? v=A 6 c. F 0 i. WC 3 J 4 Ted Talk: 4: 24 https: //www. youtube. com/watch? v=f. Pnw. BITSmg. U
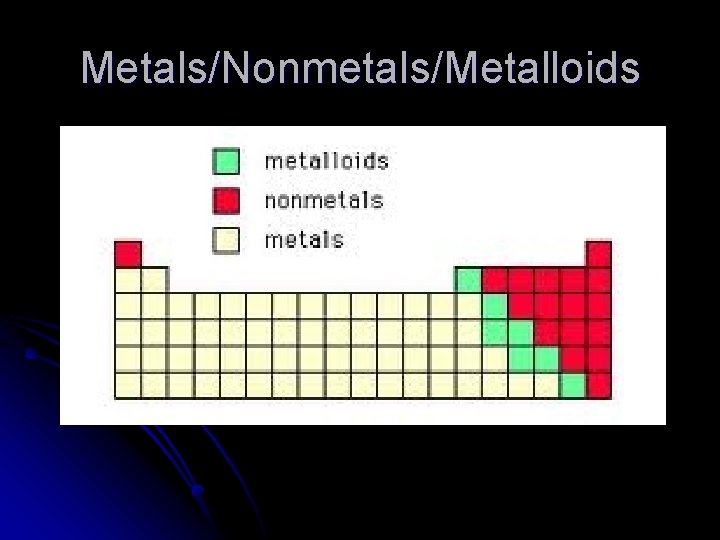
Metals/Nonmetals/Metalloids

Properties of Metals Luster l Malleable l Ductile l Conduct heat and electricity l Lose valence electrons, form positive ions l All solids (except Hg) l

Nonmetals l l l No luster Brittle (if solid) Don’t conduct Gain valence electrons, form negative ions Can be solid, liquid, gas Hydrogen is a nonmetal

Metalloids (Semimetals) l l l Most elements along the “staircase” Properties of both metals and nonmetals. B, Si, Ge, As, Sb, Te, At

Trends in the Periodic Table
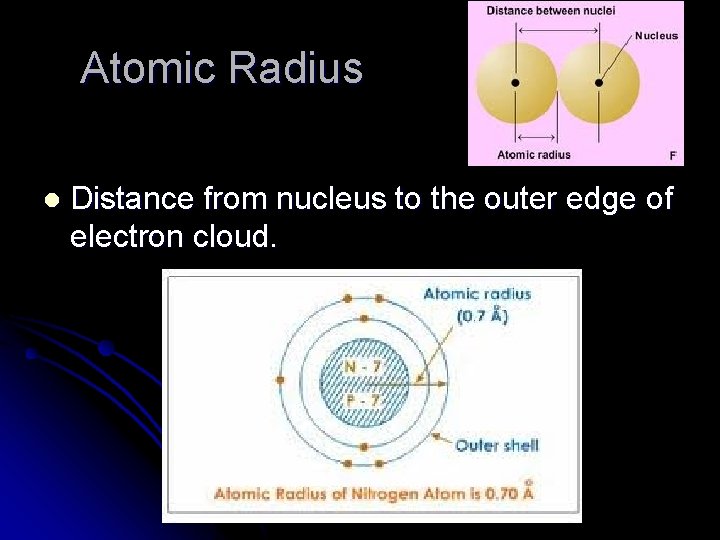
Atomic Radius l Distance from nucleus to the outer edge of electron cloud.

Trends in Radius (Use Table S) l Li Down a Group Na K Ex: Group 1 What happens? Rb Cs l Fr Across a Period Ex: Period 2 Li Be B What happens? C N O F Ne

Let’s Think: l Why does this trend happens? l How might it be related to electron shells and electron repulsion? l How might it be related to the pull of nuclear charge?

l Down a Group: l Size increases l Add new energy levels l Greater shielding from core electrons l Outer electrons further from nucleus so less attractive force
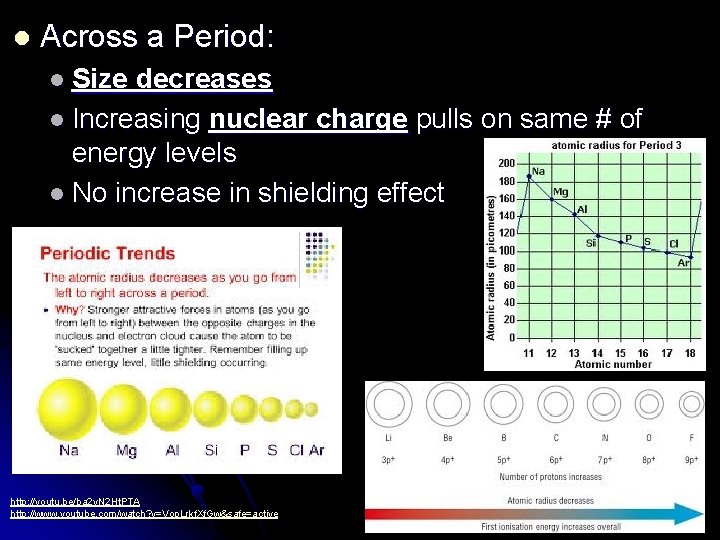
l Across a Period: l Size decreases l Increasing nuclear charge pulls on same # of energy levels l No increase in shielding effect http: //youtu. be/ba 2 y. N 2 Ht. PTA http: //www. youtube. com/watch? v=Vop. Lrkf. Xf. Gw&safe=active
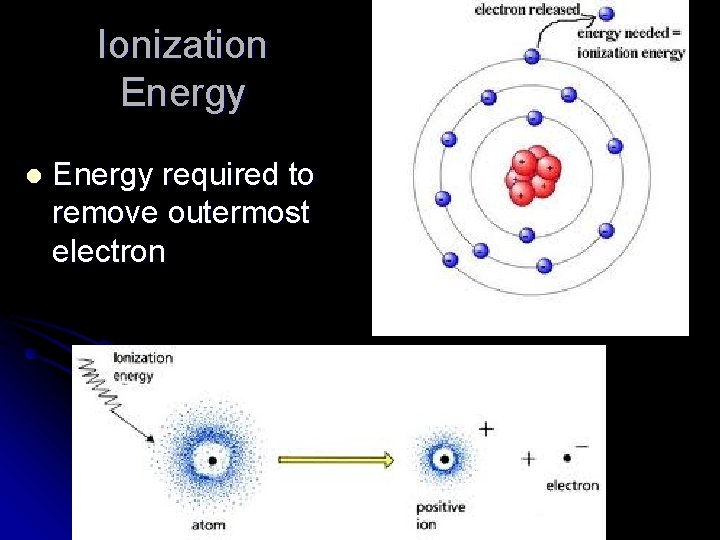
Ionization Energy l Energy required to remove outermost electron

Trends in Ionization Energy (Use Table S) l Down a Group Ex: Group 1 What happens? l Li Na K Rb Cs Fr Across a Period Ex: Period 2 Li Be B C What happens? N O F Ne
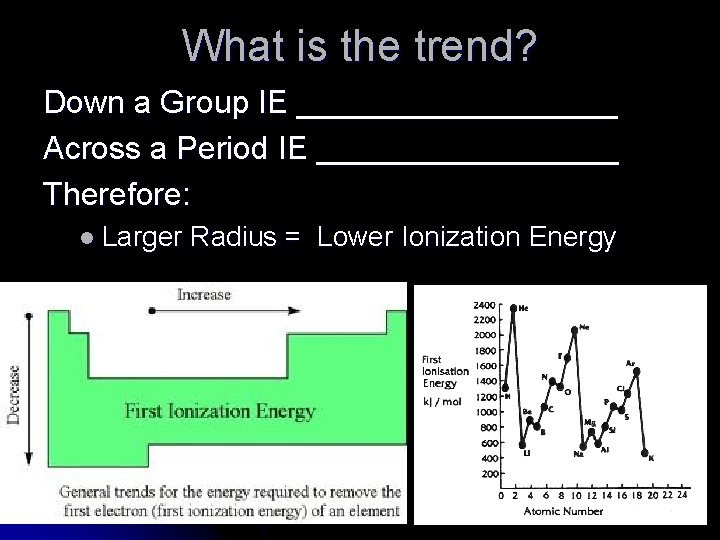
What is the trend? Down a Group IE _________ Across a Period IE _________ Therefore: l Larger Radius = Lower Ionization Energy
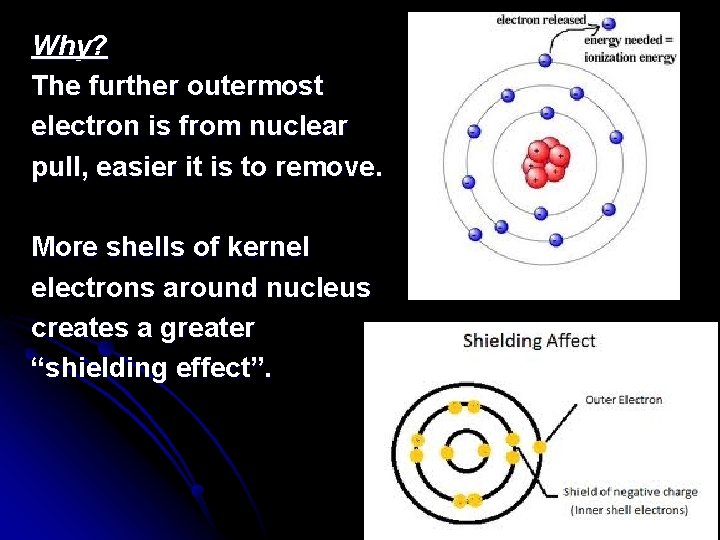
Why? The further outermost electron is from nuclear pull, easier it is to remove. More shells of kernel electrons around nucleus creates a greater “shielding effect”.

Electronegativity l Attraction an atom has for electrons involved in bond formation.

Electronegativity l l The higher the EN value the more the atom “pulls” on electrons involved in bond. “Tug of war” for electrons in bond

l Fluorine has the highest EN value = 4 l l EN scale was created by comparing other elements to Fluorine Nonmetals generally have much higher EN values than metals http: //youtu. be/93 G_Fqp. GFGY http: //www. youtube. com/watch? v=YZ 8 izz. Eq 6 z. I&safe=active

How does this Affect Bonds? l Polar Bond: l Unequal electron sharing l Atoms have different EN l Nonpolar Bond: l Equal electron sharing l Atoms have same EN

Trends in Electronegativity (Use Table S) l Down a Group Ex: Group 1 What happens? l Li Na K Rb Cs Fr Across a Period Ex: Period 2 Li Be B C What happens? N O F Ne

l What was the trend in electronegativity? l Down a group EN _________ l Across a period EN ________ l What is the relationship to atomic radius? l As size gets bigger EN gets _______ l As size gets smaller EN gets _______ l What element has the highest EN value? l What elements don’t have really have electronegativity values? Why is that?

Metallic/Non. Metallic Character l More “Metallic”: l l Large radius Low IE Low EN More “Non Metallic”: l l l Small radius High IE High EN http: //www. youtube. com/watch? feature=player_embedded&v=h. A NWof. St. Hrk

Links and Songs l l l (Honors) Intro to the Table http: //youtu. be/5 MMWpe. J 5 dn 4 Dan Radcliffe Knows his elements! http: //youtu. be/r. SAai. YKF 0 cs Elements Song: Periodic Table of Videos Crew: l http: //youtu. be/af. Fw 91 fv. NJM l A different Song http: //www. youtube. com/watch? v=z. UDDi. Wt. Ft. EM&safe=active l l l LIST OF COOL LINKS http: //www. nclark. net/Periodic. Table. html

l l l l l Crash Course Video(13 minutes) http: //www. youtube. com/watch? v=0 RRVV 4 Diomg&safe=active Interactive Table http: //ny. pbslearningmedia. org/resource/phy 03. sci. phys. matter. ptable/period ic-table-of-the-elements/ Interactive Table http: //www. chemicool. com/ Sortify Game https: //www. brainpop. com/games/sortifyelementsoftheperiodictable/ Go React Periodic Table http: //www. msichicago. org/play/goreact/
- Slides: 41