Periodic Table of Elements Dmitri Mendeleev First Table

Periodic Table of Elements

Dmitri Mendeleev □ First Table made by arranging the 63 known elements in order of □ atomic weight □ grouping them by similarity of properties. □ Predicted □ the existence of new elements □ pointed out that some of the known atomic weights were wrong □ He left space on his table for new elements, and predicted yet-to-be-discovered elements

Hydrogen: the lightest Gaps where he knew that missing elements should be.


The Modern Periodic Table □ Periods – □ Horizontal Rows (label these from top to bottom #’s 1 through 7) □ Groups/Families – □ Vertical Columns (label these from left to right #’s 1 through 18) □ Know this: Elements in groups have similar physical and chemical properties
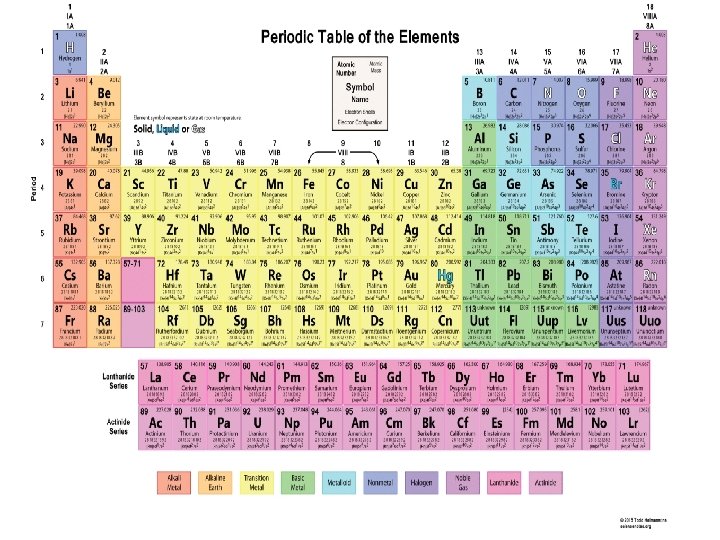
The elements get more metallic as you go down the table, and less metallic as you go across. Non-metals
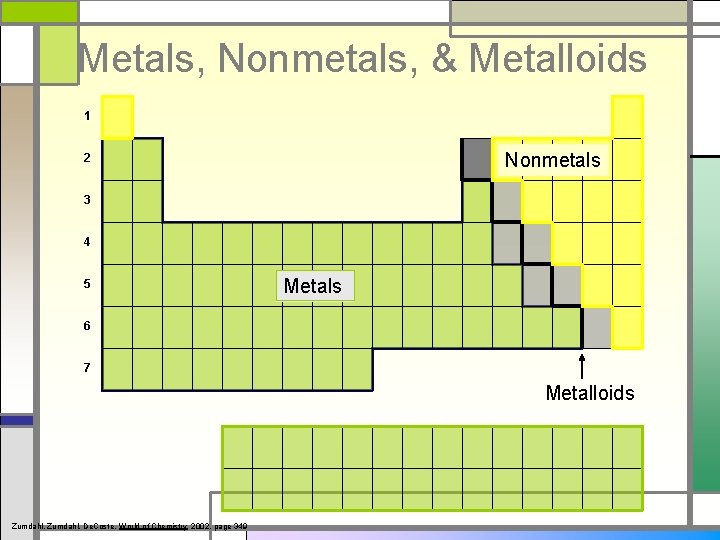
Metals, Nonmetals, & Metalloids 1 Nonmetals 2 3 4 5 Metals 6 7 Metalloids Zumdahl, De. Coste, World of Chemistry 2002, page 349

Properties of Metals, Nonmetals, and Metalloids METALS malleable, lustrous, ductile, good conductors of heat and electricity NONMETALS gases or brittle solids at room temperature, poor conductors of heat and electricity (insulators) METALLOIDS (Semi-metals) dull, brittle, semi-conductors (used in computer chips)
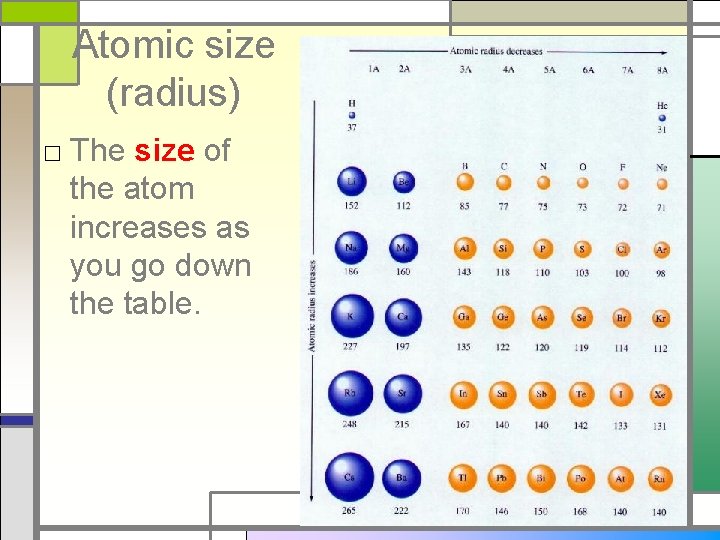
Atomic size (radius) □ The size of the atom increases as you go down the table.
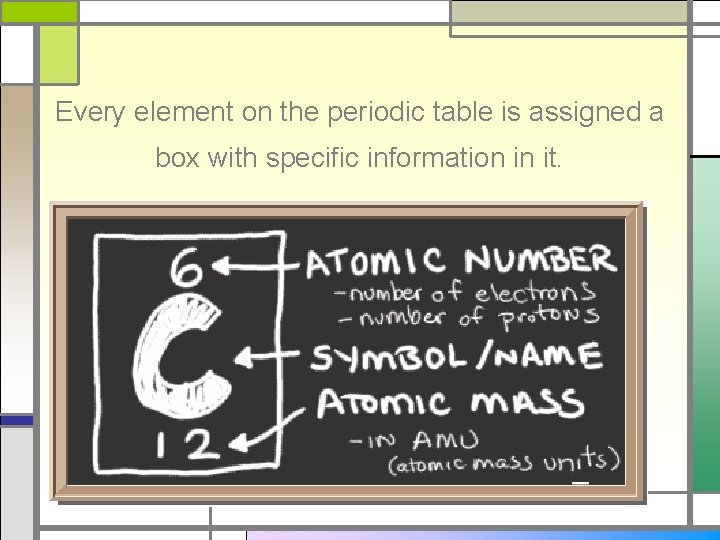
Every element on the periodic table is assigned a box with specific information in it.

Atomic Number □ The atomic number = #of protons in nucleus. □ The atomic number determines what the element is. • For example, any atom that contains exactly 47 protons in its nucleus is an atom of silver.
- Slides: 12