Periodic Table Notes Periodic Table An arrangement of

Periodic Table Notes
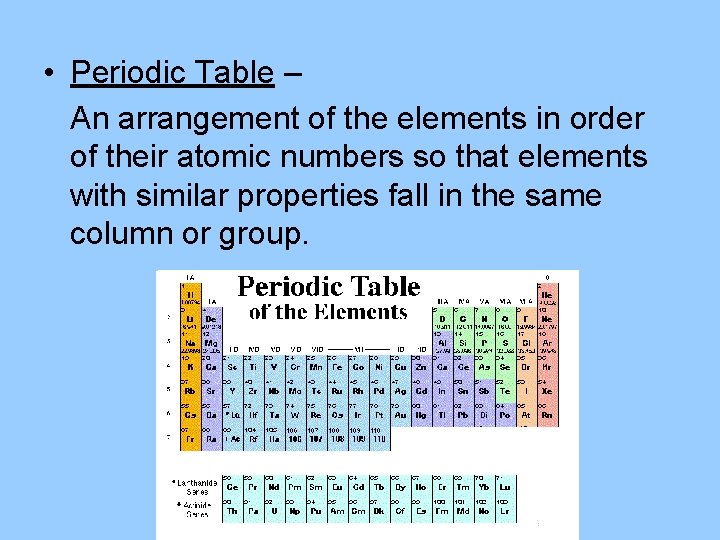
• Periodic Table – An arrangement of the elements in order of their atomic numbers so that elements with similar properties fall in the same column or group.
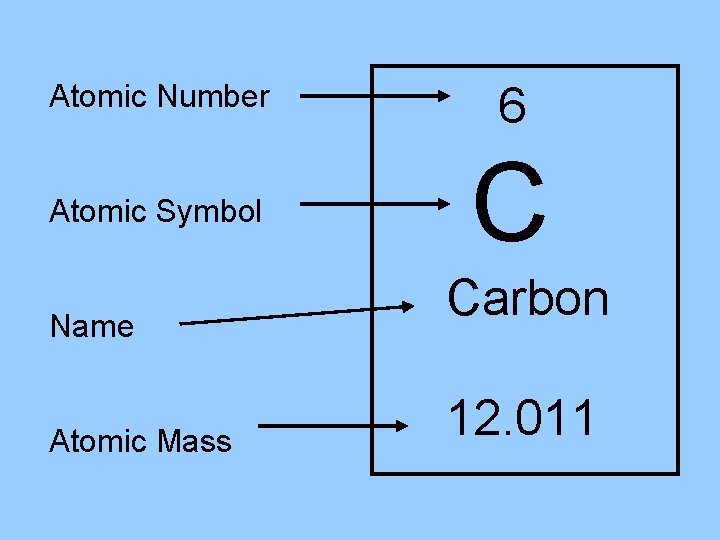
Atomic Number 6 Atomic Symbol C Name Atomic Mass Carbon 12. 011

• Atomic Radius – One half the distance between the nuclei of identical atoms that are bonded together. • Trend – Increases as you go down the groups – Decreases as you go across the periods

I N C R E A S E A. R. DECREASE

Trends Sample 1 Of the elements magnesium, Mg, chlorine, Cl, sodium, Na, and phosphorus, P, which has the largest atomic radius? Which has the smallest?
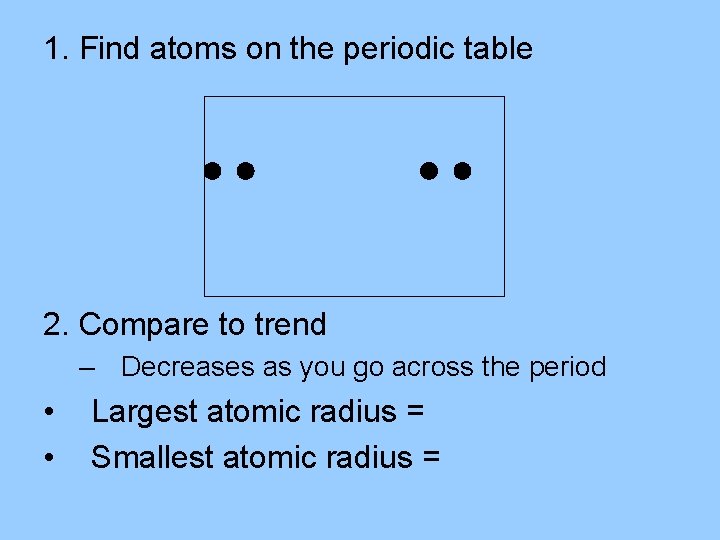
1. Find atoms on the periodic table 2. Compare to trend – Decreases as you go across the period • • Largest atomic radius = Sodium, Na Smallest atomic radius = Chlorine, Cl

Trends Sample 2 Of the elements calcium, Ca, beryllium, Be, barium, Ba, and strontium, Sr, which has the largest atomic radius? Which has the smallest?
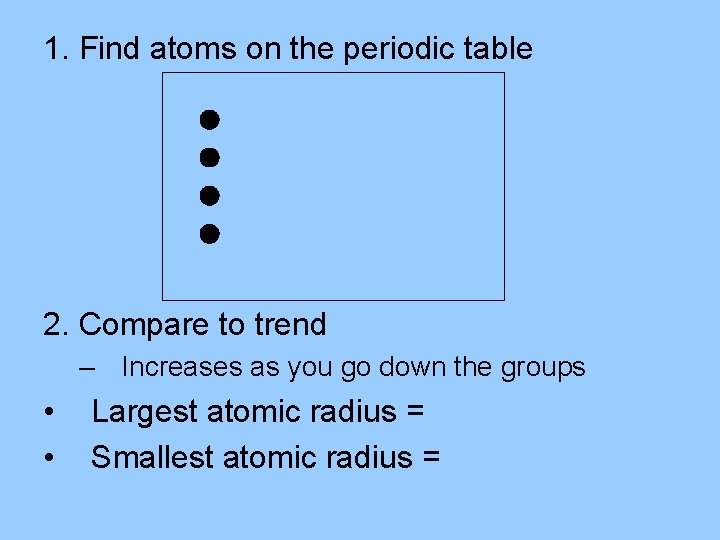
1. Find atoms on the periodic table 2. Compare to trend – Increases as you go down the groups • • Largest atomic radius = Barium, Ba Smallest atomic radius = Beryllium, Be

• Ion – An atom or group of bonded atoms that has a positive or negative charge. • Ionization Energy – The energy required to remove one electron from a neutral atom of an element. • Trend – Decreases as you go down the groups – Increases as you go across the periods

D E C R E A S E I. E. INCREASE

• Cation – Positively charged ion. • Anion – Negatively charged ion. • Valence Electrons – The electrons available to be lost, gained, or shared in the formation of chemical compounds.
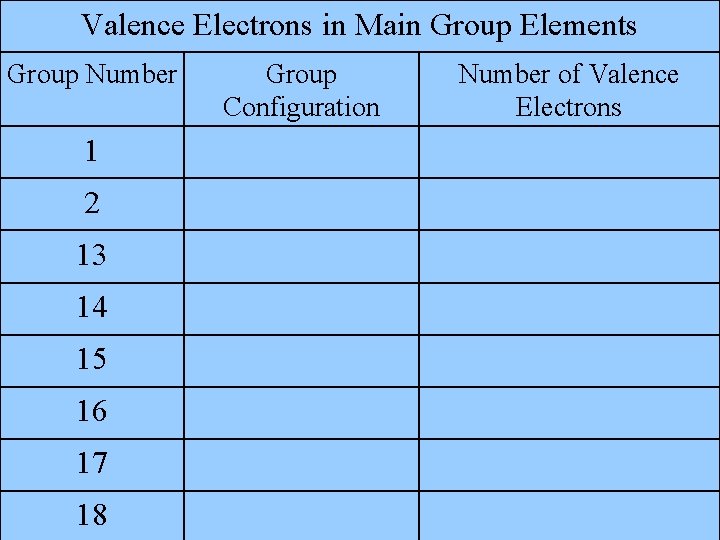
Valence Electrons in Main Group Elements Group Number Group Configuration Number of Valence Electrons 1 1 2 s 2 2 13 s 2 p 1 3 14 s 2 p 2 4 15 s 2 p 3 5 16 s 2 p 4 6 17 s 2 p 5 7 18 s 2 p 6 8

• Electronegativity – The ability of an atom in a chemical compound to attract electrons. • Trend – Decreases as you go down the groups – Increases as you go across the periods – Nobel gases have no electronegativity (they do not want electrons)

D E C R E A S E EN INCREASE

• Fluorine is the most electronegative element (wants an electron the most) and is therefore assigned the highest value of four.
- Slides: 16