Periodic Table Mendeleevs Periodic Table In 1869 Dmitri
Periodic Table

• • • Mendeleev's Periodic Table In 1869, Dmitri Ivanovitch Mendeléev created the first accepted version of the periodic table. Mendeléev grouped elements according to their atomic mass, and similar chemical properties. The table contained gaps but Mendeleev predicted the discovery of new elements. In 1906, Mendeleev came within one vote of receiving the Nobel Prize in chemistry. Dimitri Mendeleev (1834 -1907)
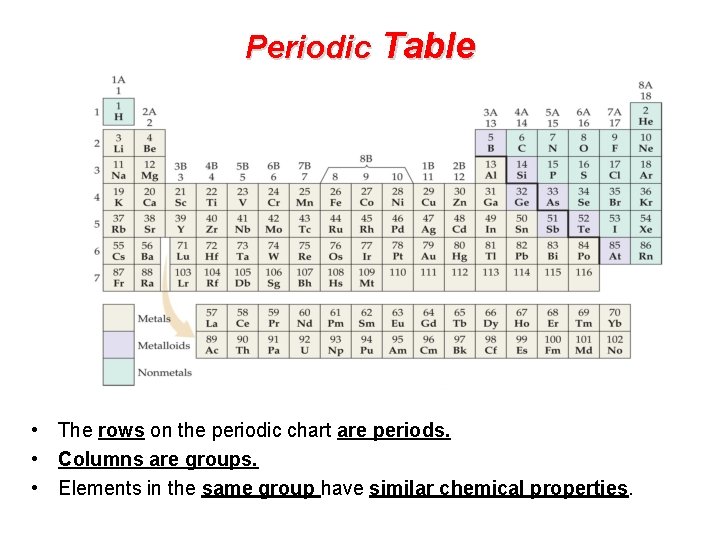
Periodic Table • The rows on the periodic chart are periods. • Columns are groups. • Elements in the same group have similar chemical properties.
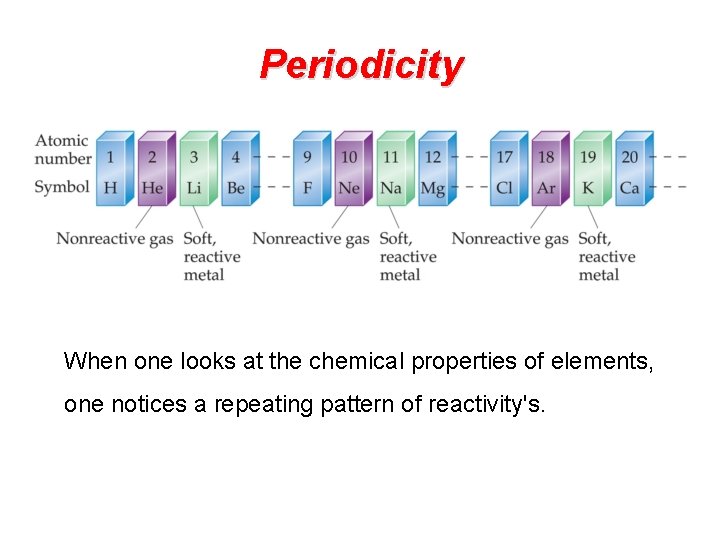
Periodicity When one looks at the chemical properties of elements, one notices a repeating pattern of reactivity's.

Periods • Each horizontal row of elements is called a period. • The elements in a period are not alike in properties. • The first element in a period is always an extremely active solid. The last element in a period, is always an inactive gas. • Each periods represents a major energy level. As you go across The Periodic Table horizontally in particular row, each element has one more electron in the same outer level. Example: Sodium has 1 (3 s 1), Magnesium has 2 (3 s 2), Aluminum has 3 (3 s 2, 3 p 1), Silicon has 4 (3 s 23 p 2 ), Phosphor has 5 (3 s 2, 3 p 3), Sulfur has 6 (3 s 2, 3 p 4), Chlorine has 7 (3 s 2, 3 p 5), and Argon has 8 (3 s 2, 3 p 6).

Families (Groups) • Columns of elements are called groups or families. • All elements in a family have the same number of valence electrons. • Elements in each family have similar but not identical properties. • For example, lithium (Li), sodium (Na), potassium (K), and other members of family IA (Alkali Metals). They are all soft, white, shiny metals. • Elements in the same column usually have the same formulas for their compounds. Example: When Beryllium (Be) in Group IIA bonds with Oxygen, it forms a compound with formula Be. O. Similarly, Magnesium (Mg) bonds with Oxygen to form Mg. O and Calcium (Ca) with Oxygen to form Ca. O.

What’s in a square? Different periodic tables can include various bits of information, but usually: – atomic number – symbol – atomic mass – number of valence electrons – state of matter at room temperature.

Elements are organized on the table according to their atomic number, usually found near the top of the square. – The atomic number refers to how many protons an atom of that element has. – For instance, carbon has 6 proton, so it’s atomic number is 6. – The atomic number is unique to that element. No two elements have the same atomic number.
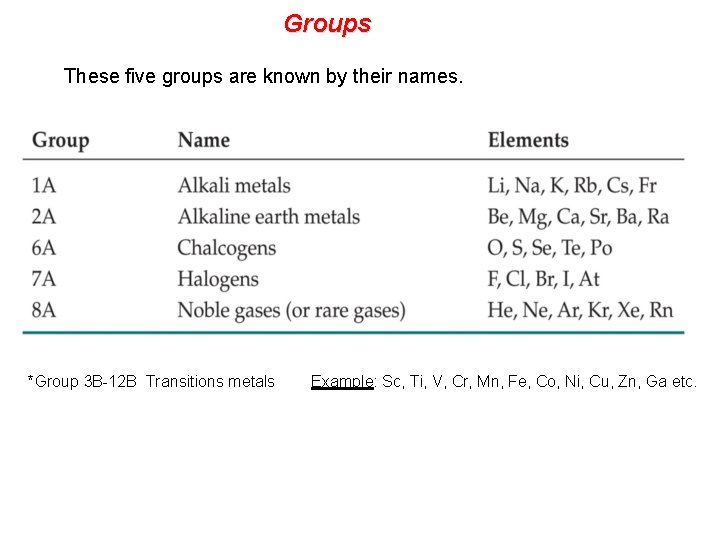
Groups These five groups are known by their names. *Group 3 B-12 B Transitions metals Example: Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga etc.

Periodic Table Trends • As you compare elements from left to right across the periodic table, you will notice a trend or regular change in a number of properties. • The same thing happens if you go up and down on the periodic table and compare the properties of the elements. • There are several important atomic characteristics that show predictable trends that you should know: 1. Atomic radius ( or atomic size) - half of the distance between the centers of two atoms that are touching each other 2. Ionization energy - tendencies to lose and gain electrons 3. Electron affinity (chemical reactivity) - ability to accept an electron 4. Electronegativity - measure of the attraction to form a chemical bond
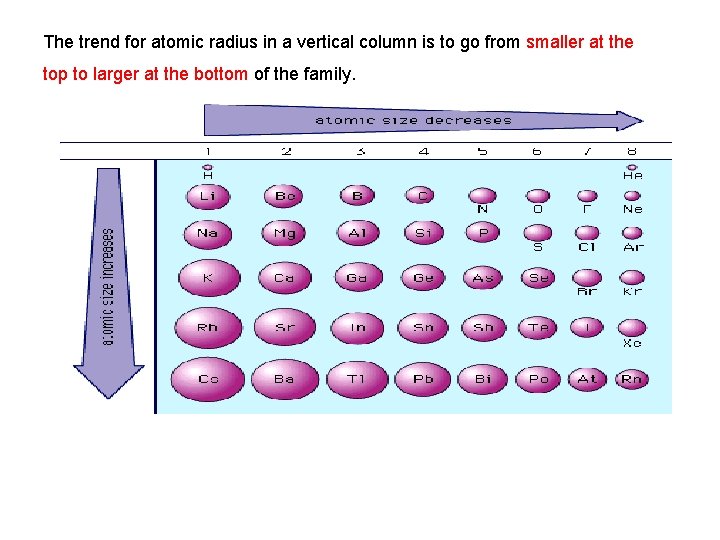
The trend for atomic radius in a vertical column is to go from smaller at the top to larger at the bottom of the family.
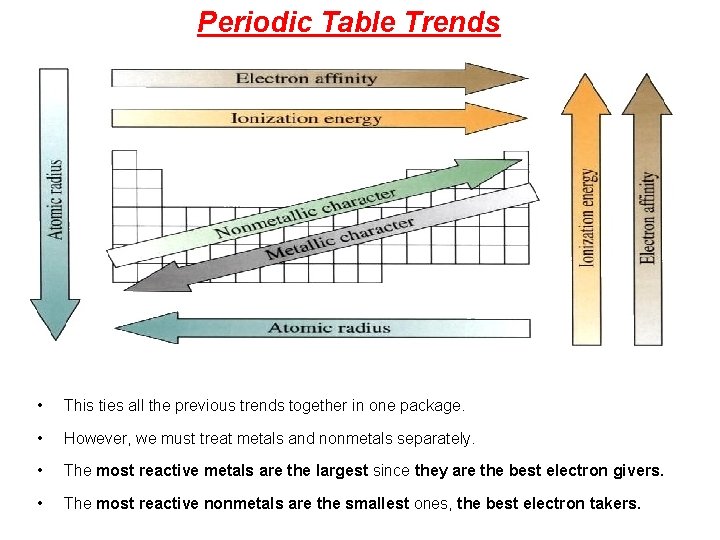
Periodic Table Trends • This ties all the previous trends together in one package. • However, we must treat metals and nonmetals separately. • The most reactive metals are the largest since they are the best electron givers. • The most reactive nonmetals are the smallest ones, the best electron takers.

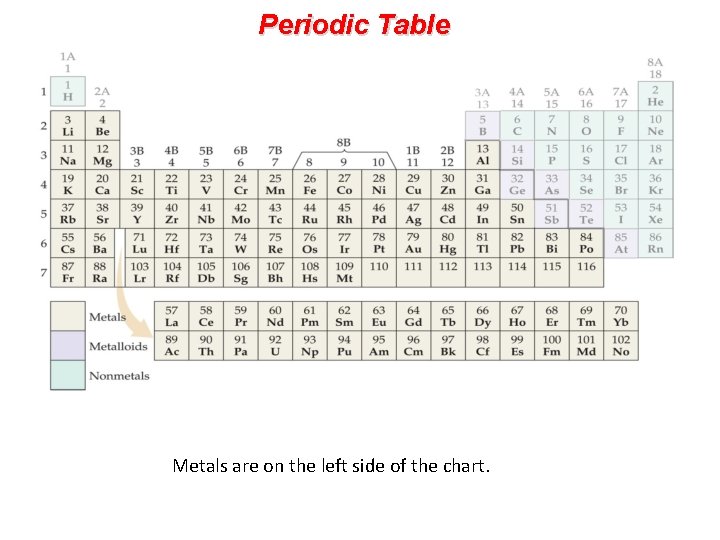
Periodic Table Metals are on the left side of the chart.

Properties of Metals • Metals are solids at room temperature, except for mercury which is liquid. • Gallium will melt in your hand. • Metals have a shiny luster. • Metals are good conductors of heat and electricity. • Metals are shiny. • Metals are ductile (can be stretched into thin wires). • Metals are malleable (can be pounded into thin sheets). • A chemical property of metal is its reaction with water and oxygen which results in corrosion. • In a metal, atoms readily lose electrons to form positive ions (cations). • Metals form ionic bonds with non-metals. • Metals generally have high melting points and high densities compare to nonmetals. • The least active metals are so-called “noble” metals, such gold (Au), silver(Ag), platinum (Pt), palladium (Pd) and sometimes copper (Cu)


Alkali Metals • The alkali family is found in the first column of the periodic table. • The word “alkali” means “base”, these metal are so named because they react with water to form a base and hydrogen gas Example: Na(s) + H 2 O(l) →Na. OH (aq) + H 2(g) • Atoms of the alkali metals have a single electron in their outermost level, in other words, 1 valence electron. • They are shiny, have the consistency of clay, and are easily cut with a knife. • They are the most reactive metals. • They react violently with water. • Alkali metals are never found as free elements in nature. They are always bonded with another element. Cesium (or Caesium)

Alkaline Earth Metals • They are never found uncombined in nature. • They have two valence electrons. • The alkaline earth metals contain beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba) and radium (Ra). • The alkaline earth metals are silver colored, soft metals, which react readily with halogens to form ionic salts, and with water, though not as rapidly as the alkali metals, to form strong alkaline (basic) hydroxides. • Beryllium is an exception: It does not react with water or steam, and its halides are covalent calcium

Transition Metals • These are the metals you are probably most familiar: copper, tin, zinc, iron, nickel, gold, and silver. • They are good conductors of heat and electricity. • The compounds of transition metals are usually brightly colored and are often used to color paints. The formation of compounds whose color is due to d - d electronic transitions • Transition elements have 1 or 2 valence electrons, which they lose when they form bonds with other atoms. Some transition elements can lose electrons in their next-to-outermost level. • Transition elements have properties similar to one another and to other metals, but their properties do not fit in with those of any other family. • Many transition metals combine chemically with oxygen to form Cobalt - hard lustrous gray metal compounds called oxides due to the relatively low reactivity of unpaired d electrons. • Transition metals can form of many paramagnetic compounds due to the presence of unpaired d electrons Cobalt(II) chloride hexahydrate
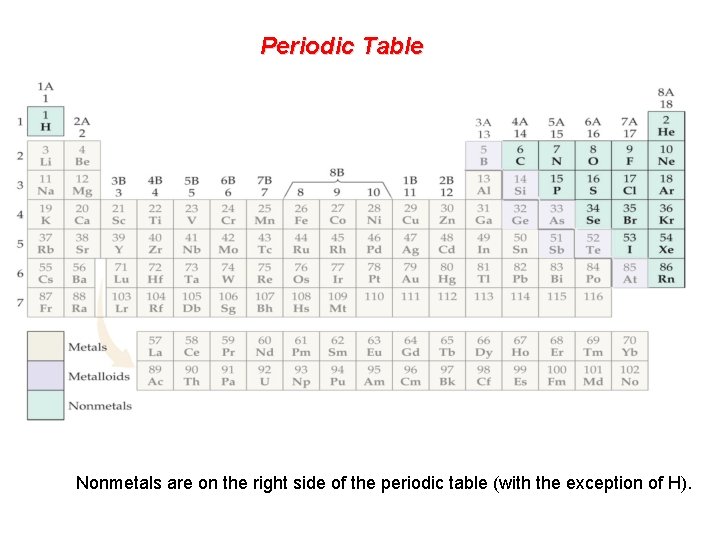
Periodic Table Nonmetals are on the right side of the periodic table (with the exception of H).

Properties of Non-Metals • Nonmetals can exist as all three state: solids (like carbon, phosphorus, sulfur), liquids (like bromine) and gases (like hydrogen, oxygen, nitrogen, chlorine, helium etc. ) • Nonmetals are poor conductors of heat and electricity. • Nonmetals in solid form are dull • Solid non-metals are brittle and break easily. • Usually have lower densities than metals • They have significantly lower melting points and boiling points than metals (with the Sulfur exception of Carbon) • Non-metals have high electronegativity, and thus the tendency to form negative ions. • Non-metals will combine with metals to form salts. Phosphorus Example: Na. Cl, KBr etc. • Non-metal combines with oxygen can form oxide (ex. CO 2, SO 3, N 2 O 4); if non-metal combines with sulfur can form sulfide (ex. CS 2); if nonmetal combines with element from GROUP VII A of the Periodic Table can form halide (ex. NF 3, PCl 5). • Fluorine is the most reactive nonmetal Bromine


Boron Family (Group) • The Boron Family is named after the first element in the family. • Atoms in this family have 3 valence electrons. • The boron group consists of boron (B), aluminium (Al), gallium (Ga), indium (In), thallium (Tl), and ununtrium (Uut). • This family includes the most abundant metal in the earth’s crust (aluminum). • All of the elements in group 13 are pretty reactive at moderate temperatures except boron, which is only pretty reactive at very high temperatures. • One characteristic that all do have in common is that all have three electrons in their valence shells. • Boron, being a metalloid, is an insulator at room temperature, but a good conductor at high temperatures. Unlike boron, the metals in the group are good conductors at usual conditions. The exact same can go for electricity because boron is not good at conducting electricity at cool temperatures but is good at high temperatures, and the other metals conduct it.

Carbon Family (Group) • This family includes a non-metal (carbon), metalloids, and metals. The carbon group consisting of carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and ununquadium (Uuq). • The element carbon is called the “basis of life. ” There is an entire branch of chemistry devoted to carbon compounds called organic Germanium chemistry. • Each of the elements in this group has 4 electrons in its outer energy level. Carbon alone forms negative ions, in the form of carbide (C 4−) ions. Silicon and germanium, both metalloids, each can form +4 ions. Tin and lead both are metals while ununquadium is a synthetic short-lived radioactive metal. Tin and lead are both capable of forming +2 ions • Except for germanium and ununquadium, all of these elements are familiar in daily life either as the pure element or in the form of compounds. Diamond and Graphite

Nitrogen Family (Group) • The nitrogen family is named after the element that makes up 78% of our atmosphere. • This family includes non-metals, metalloids, and metals. • The nitrogen group consisting of nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi) and ununpentium (Uup) (unconfirmed). • Atoms in the nitrogen family have 5 valence electrons. They tend to share electrons when they bond. • These elements are also noted for their stability in compounds due to their tendency forming double and triple covalent bonds. • This is the property of these elements which leads to their potential toxicity, most evident in phosphorus, arsenic and antimony. • All the elements in the group are a solid at room temperature except for nitrogen which is a gas at room temperature.

Oxygen Family (Group) Chalcogen Group (“"copper-ore former”) • Oxygen Group consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), the radioactive element polonium (Po), and the synthetic element ununhexium (Uuh). • Atoms of this family have 6 valence electrons. • Most elements in this family share electrons when forming compounds. • Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements. • Oxygen and sulfur are nonmetals, and selenium, tellurium, and polonium are metalloid semiconductors. Nevertheless, tellurium, as well as selenium, is often referred to as a metal when in elemental form. • Metal chalcogenides are common as minerals. For example, pyrite (Fe. S 2) is an iron ore. The rare mineral calaverite is the ditelluride Au. Te 2. • The formal oxidation number of the most common chalcogen compounds is − 2. The highest formal oxidation number +6 is found in sulfates, selenates and tellurates, such as in sulfuric acid or sodium selenate (Na 2 Se. O 4).

Halogen Family (Group) • The elements in this family are fluorine (F), chlorine (Cl), bromine(Br), iodine(I), and astatine(At). The artificially created element 117, provisionally referred to by the systematic name ununseptium, may also be a halogen. • Halogens have 7 valence electrons, which explains why they are the most active non-metals. They are never found free in nature • In their elemental forms, the halogens exist as diatomic molecules. • At room temperature and pressure, fluorine and chlorine are gases, bromine is a liquid and iodine and astatine are solids. • Halogen atoms only need to gain 1 electron to fill their outermost energy level. • They react with alkali metals to form salts. • Halogens are highly reactive, and as such can be harmful or lethal to biological organisms in sufficient quantities.
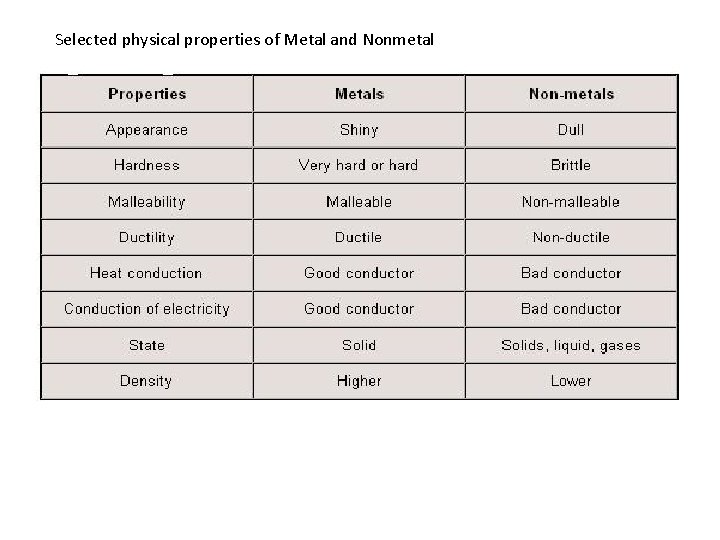
Selected physical properties of Metal and Nonmetal
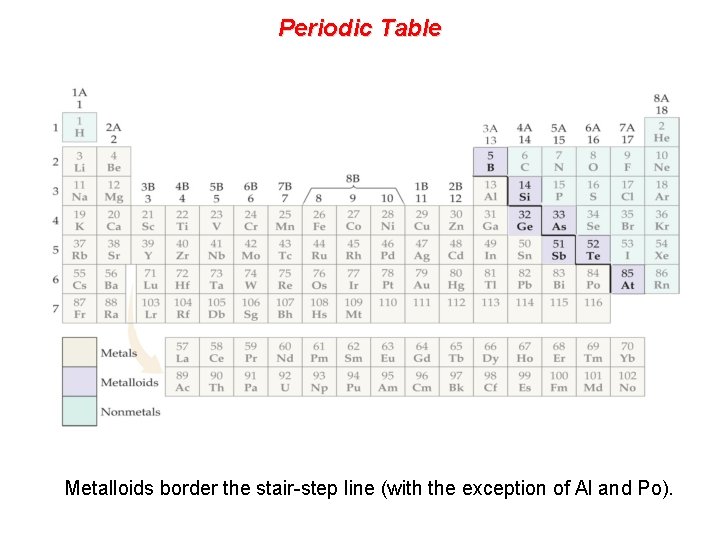
Periodic Table Metalloids border the stair-step line (with the exception of Al and Po).



Properties of Metalloids • Metalloids (metal-like) have properties of both metals and non-metals. • They are solids that can be shiny or dull. • They conduct heat and electricity better than non -metals but not as well as metals. • They are ductile and malleable. • Boron (B) • Silicon (Si) • Germanium (Ge) • Arsenic (As) • Antimony (Sb) • Tellurium (Te) Silicon


Noble Gases • Under standard conditions are colorless, odorless, . , monatomic gases that are extremely unreactive. • One important property of the noble gases is their inactivity. They are inactive because their outermost energy level is full. • Because they do not readily combine with other elements to form compounds, the noble gases are called inert. • The family of noble gases includes helium, neon, argon, krypton, xenon, and radon. • All the noble gases are found in small amounts in the earth's atmosphere.

Diatomic molecules are molecules composed only of two atoms. About 99% of the Earth's atmosphere is composed of diatomic molecules, specifically oxygen and nitrogen at 21 and 78%, respectively. The natural abundance of hydrogen (H 2) in the Earth's atmosphere is only on the order of parts per million, but H 2 is, in fact, the most abundant diatomic molecule seen in nature. Know: Hydrogen H 2 Nitrogen N 2 Oxygen O 2 Fluorine F 2 Chlorine Cl 2 Bromine Br 2 Iodine I 2 Astatine At 2 N 2

Rare Earth Elements • The thirty rare earth elements are composed of the lanthanide and actinide series. • One element of the lanthanide series and most of the elements in the actinide series are called trans-uranium, which means synthetic or man-made. • This group of 17 elements is critical to the production of automotive components, communications technologies, clean energy sources, weapons systems, traditional fuel refineries, and countless other technologies. • The rare earths are silver, silvery-white, or gray metals. • The metals have a high luster, but tarnish readily in air. • The metals have high electrical conductivity. • The rare earths share many common properties. This makes them difficult to separate or even distinguish from each other. • There are very small differences in solubility and complex formation between the rare earths. • The rare earth metals naturally occur together in minerals (e. g. , monazite is a mixed rare earth phosphate). • Rare earths are found with non-metals, usually in the 3+ oxidation state. There is little tendency to vary the valence. (Europium also has a valence of 2+ and cerium also a valence of 4+. ) These rare-earth oxides are used as tracers to determine which parts of a watershed are eroding. Clockwise from top center: praseodymium, cerium, lanthanum, neodymium, samarium, and gadolinium.

Hydrogen • Hydrogen was discovered by Henri Cavendish in 1766 and was named “hydrogen” (“water former”) by Lavoisier • Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of 1. 00794 u • The hydrogen square sits a top Family IA, but it is not a member of that family. Hydrogen is in a class of its own. • Hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. • Stars in the main sequence are mainly composed of hydrogen in its plasma state. • Naturally occurring elemental hydrogen is relatively rare on Earth. • It’s a diatomic gas at room temperature (H 2), colorless, does not conduct heat and electricity well. • Hydrogen does react explosively with oxygen to produce water: 2 H 2 + O 2 → 2 H 2 O • It has one proton and one electron in its one and only energy level. • Hydrogen only needs 2 electrons to fill up its valence shell.

Hydrogen • The commercial preparation of hydrogen is from natural gas (methane) and steam. • At high temperatures (1000– 1400 K, 700– 1100 °C or 1300– 2000 °F), steam (water vapor) reacts with methane to yield carbon monoxide and H 2. CH 4 + H 2 O → CO + 3 H 2 • Additional hydrogen can be recovered from the steam by use of carbon monoxide through the water gas shift reaction, especially with an iron oxide catalyst (substance which speeds up a chemical reaction without itself being used up). This reaction is also a common industrial source of carbon dioxide. catalyst CO + H 2 O • • a) CO 2 + H 2 The common laboratory preparation of H 2 is by reacting an active metal with strong acid Zn + H 2 SO 4 Zn. SO 4 + H 2 Zink Sulfide Hydrogen Sulfuric Acid Uses of Hydrogen : Making ammonia by Haber process. The ammonia is then used to make fertilizers. N 2 + 3 H 2 b) 900⁰C catalyst 2 NH 3 Hydrogenation – Making solid fats from liquid oils by adding hydrogen to double bonds of certain hydrocarbons. c) Oxy-hydrogen welding d) Possible future use for energy from use in fuel cells or fusion

Sodium • Sodium is the most abundant alkali metal and was discovered in 1807 by Sir Humphry Davy when he passed an electric current through molten sodium chloride. • The main source of sodium is from sodium chloride (Na. Cl) from the oceans or underground brine and salt deposits. • Sodium is a soft, silvery-white solid with a low density as metals go. It is so soft it can be cut with a knife. • Sodium with a symbol Na and atomic number 11 is highly reactive metal that it must be stored under kerosene (paraffin) to keep it away from moisture in the air. • Sodium react violently with water to produce sodium hydroxide and hydrogen gas as well as heat. 2 Na + 2 H 2 O → 2 Na. OH + H 2 • Uses of Sodium: a) Dehydrating agent to remove water from other liquids b) Vapor lamps c) Sodium mixes with other metal like Hg, K, Sn and Sb to make important alloys d) The petroleum industry uses sodium to make certain antiknock additives for gasoline e) Sodium is also used by nuclear industry as a liquid coolant for power plants like FFTF facility in Hanford. The Fast Flux Test Facility is a 400 MW nuclear test reactor owned by the U. S. Department of Energy, sodium-cooled Fast neutron reactor.

Chlorine • Chlorine is the chemical element with atomic number 17 and symbol Cl. • It is a halogen, yellow-green diatomic gas (Cl 2), which is denser then air • As the chloride ion, which is part of common salt and other compounds, one of the element with which both hydrogen and sodium react. • It is abundant in nature and necessary to most forms of life, including humans. • Chlorine is a powerful oxidant and is used in bleaching and disinfectants, as well as an essential reagent in the chemical industry • Chlorine is very reactive and poisonous, it will react with water in your lungs to make HCl, hydrochloric acid. • In nature, chlorine is found primarily as the chloride ion, a component of the salt that is deposited in the earth or dissolved in the oceans— about 1. 9% of the mass of seawater is chloride ions. Even higher concentrations of chloride are found in the Dead Sea and in underground brine deposits • Chlorine was discovered in 1774 by Swedish chemist Carl Wilhelm Scheele and name in 1810 by Sir Humphrey Davy. • In industry, elemental chlorine is usually produced by the electrolysis of sodium chloride dissolved in water: 2 Na. Cl + 2 H 2 O → Cl 2 + H 2 + 2 Na. OH • Chlorine is an important chemical for water purification (such as water treatment plants), in disinfectants, and in bleach • Chlorine is usually used (in the form of hypochlorous acid) to kill bacteria and other microbes in drinking water supplies and public swimming pools. • Chlorine is using in chemical industry to make pesticides, plastics, refrigerants (freon) Salt from Death Valley.

Silicon • Silicon is the most common metalloid. • It is a chemical element, which has the symbol Si and atomic number 14. • It is less reactive than its chemical analog carbon. • Silicon is the eighth most common element in the universe by mass, but very rarely occurs as the pure free element in nature. • It is more widely distributed in dusts, sands, planetoids and planets as various forms of silicon dioxide (silica) or silicates. • In Earth's crust, silicon is the second most abundant element after oxygen, making up 27. 7% of the crust by mass. • Silicon has many industrial uses. It is the principal component of most semiconductor devices • In its crystalline form, pure silicon has a gray color and a metallic luster. • Silicon is one of the few substances (like water, bismuth, and gallium) whose density is higher in liquid than in solid state, so it expands when it freezes. • Silicon was discovered by Berzelius in 1824. • Silica occurs in minerals consisting of (practically) pure silicon dioxide in different crystalline forms. Amethyst, agate, quartz, rock crystal, chalcedony, flint, jasper, and opal are some of the forms in which silicon dioxide appears. • Silicates (silicon-oxygen compounds) are used in making clays, cement, glass. Very pure silicon itselfs is used to making computer chips, transistors and integrated circuits.

• Known as Alkali Metals Group 1 A • Very reactive • Never found free in nature • React readily with water • Known as Alkaline earth elements Group 2 A • All are metals • Occur only in compounds • React with oxygen in the general formula EO (where O is oxygen and E is Group 2 A element) • Metalloids Group 3 A • Includes Aluminum (the most abundant metal in the earth) • Forms oxygen compounds with a X 2 O 3 formula • Includes metals and nonmetals Group 4 A • Go from nonmetals at the top of the column to metals at the bottom • All oxygen form compounds with a XO 2 formula Group 5 A Group 6 A • All elements form an oxygen or sulfur compound with E 2 O 3 or E 2 S 3 formulas • Includes oxygen, one of the most abundant elements. • Generally, oxygen compound formulas within this group are EO 2 and EO 3 • Elements combine violently with alkali metals to form salts Group 7 A • Called halogens, which mean "salt forming" • Are all highly reactive • Least reactive group Group 8 A • All elements are gases • Not very abundant on earth • Given the name noble gas because they are not very reactive

More information about Periodic Table, Groups, Periods and Elements: http: //chemistry. about. com/od/elementgroups/Element_Groups. htm http: //www. chemicalelements. com/index. html http: //www. lenntech. com/periodic/elements/bi. htm http: //www. uky. edu/Projects/Chemcomics/
- Slides: 43