Periodic Table Lesson 3 Atomic Number vs Atomic

Periodic Table - Lesson 3 Atomic Number vs. Atomic Mass

Warm-up: • A Bohr model of a helium atom and a beryllium atom are given below. Helium, He Berylium, Be • List three similarities and three differences. Unit 1 • Investigation III

Notes – Atomic number is the number of protons in the nucleus of an atom. – Mass number is the mass of an individual atom. Unit 1 • Investigation III

Activity • 1. Label the protons, neutrons and electrons on the Bohr models. Electron Proton Neutron Beryllium Atom Fluorine Atom Carbon Atom 2. Each atom is neutral. Explain why. 3. Is the nucleus of the atoms positive, negative or neutral? How do you know? (cont. ) Unit 1 • Investigation III

• 4. How did you know which were protons and which were neutrons? • 5. Use your periodic table to fill in the data table. Unit 1 • Investigation III
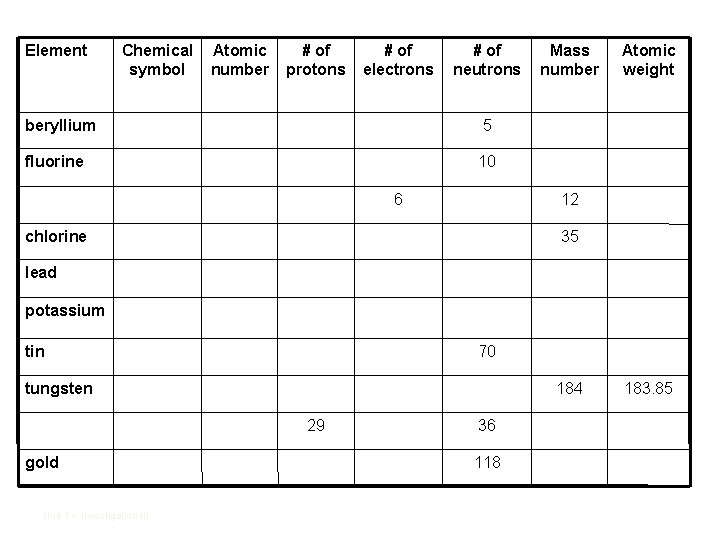
Element Chemical symbol Atomic number # of protons # of electrons # of neutrons beryllium 5 fluorine 10 6 Mass number Atomic weight 12 chlorine 35 lead potassium tin 70 tungsten 184 29 gold Unit 1 • Investigation III 36 118 183. 85

• 6. How did you figure out the number of electrons for each atom? • 7. How did you figure out the number of protons for each atom? • 8. How did you figure out the number of neutrons for each atom? • 9. How does the mass number compare to the atomic weight on the periodic table? Unit 1 • Investigation III

10. If you know the atomic number of an element, what other information can you figure out about the atoms of that element? 11. If you know the atomic number of an element, can you figure out how many neutrons an atom of that element has? Can you come up with a close guess? Explain. Unit 1 • Investigation III

Notes – Mass number is the number of protons plus the number of neutrons. – Atomic mass is the “weight” or mass of a single atom. – Atomic weight is the decimal number on the periodic table. Unit 1 • Investigation III

Wrap-Up – Each successive element has one more proton than the element preceding it. – The atomic number is equal to the number of protons. (cont. ) Unit 1 • Investigation III

(cont. ) – The number of electrons is equal to the number of protons (as long as the atom is neutral). – The mass number is equal to the number of protons plus the number of neutrons (most of the mass is found in the nucleus). Unit 1 • Investigation III

Ticket-Out • Use your periodic table to identify the following elements: • a) Atomic number 18 • b) Has three electrons • c) Atomic mass of 16. 0 Unit 1 • Investigation III
- Slides: 12