PERIODIC TABLE I History of Periodic Table 1790s

PERIODIC TABLE

I. History of Periodic Table

1790’s Antoine Lavoisier n French chemist n compiled a list of 23 elements.

Early-Mid 1800’s n. Invention of electricity allowed breakdown of compounds, causing drastic increase in number of identified elements.

n. Industrial Revolution fueled development of chemistry-based industry

n In 1860, chemists agreed on a method of accurately determining atomic mass, so elements could be identified by this method.

n. By 1860’s, about 70 elements had been discovered.

John Newlands n English chemist n Arranged elements by increasing mass in 1864 n Found properties th repeated every 8 element n “Law of Octaves”

Dimitri Mendeleev § § § Russian chemist First to publish PT. His version published in 1869 Used vertical columns to list by increasing atomic mass, and horizontal rows to arrange by similar traits (opposite of today).

n Predicted that new elements would later be discovered that would fit into the “blank spaces” in his periodic table.


Henry Moseley n British physicist. n Discovered nuclear charge (protons). n Arranged P. T. by increasing atomic number.

The Modern Table

Arranged in horizontal rows by increasing atomic number.

n. Periodic Law: The physical and chemical properties of the elements are a periodic function of their atomic numbers (periodic = repeating).

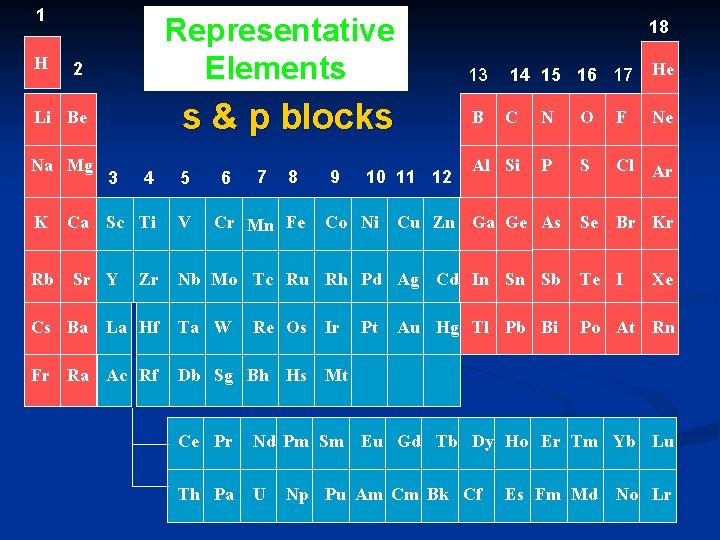
II. Organization of the Periodic Table
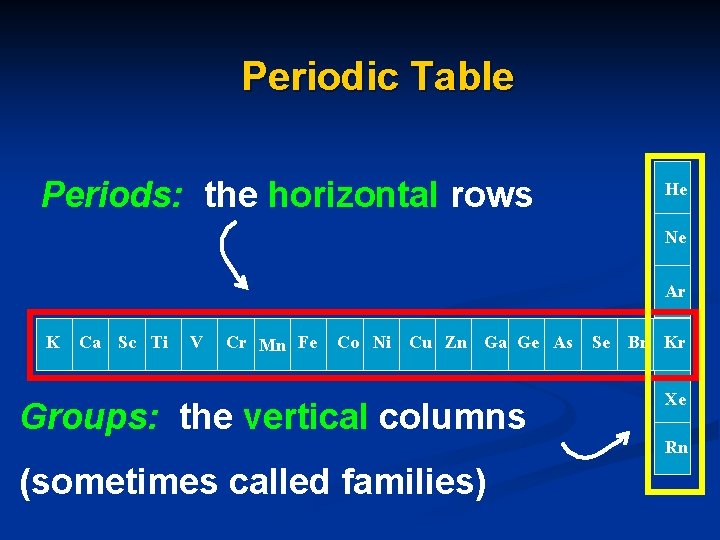
Periodic Table Periods: the horizontal rows He Ne Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Groups: the vertical columns Se Br Kr Xe Rn (sometimes called families)

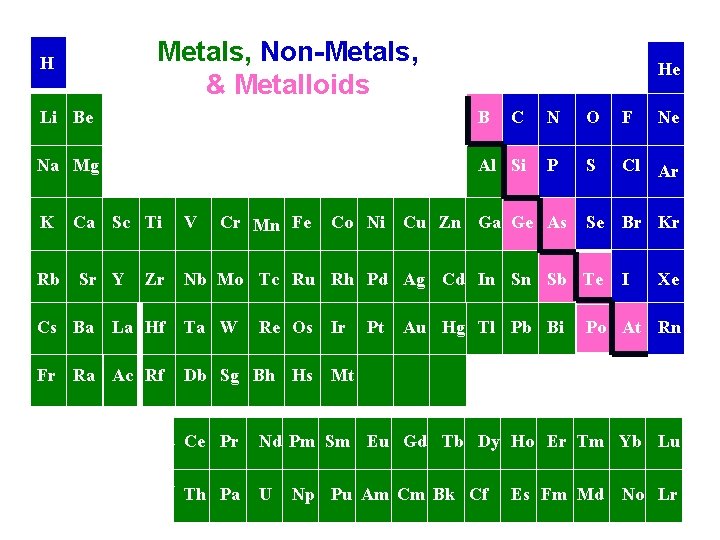
Physical State (phase) at Room Temperature *On classroom periodic table: BLACK = solid BLUE = liquid RED = gas CLEAR = man-made
1 A H Metals, Non-Metals, & Metalloids 2 A Li Be Na Mg K Rb 3 A B 3 B 4 B 5 B 6 B 7 B 8 B Ca Sc Ti Sr Y Cs Ba 8 A Zr La Hf Fr Ra Ac Rf V Cr Mn Fe 8 B 8 B 1 B 2 B Co Ni 4 A 5 A 6 A 7 A He C N O F Ne Al Si P S Cl Ar Se Br Kr Cu Zn Ga Ge As Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Ta W Re Os Db Sg Bh Hs Ir Pt Au Hg Tl Pb Bi I Xe Po At Rn Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

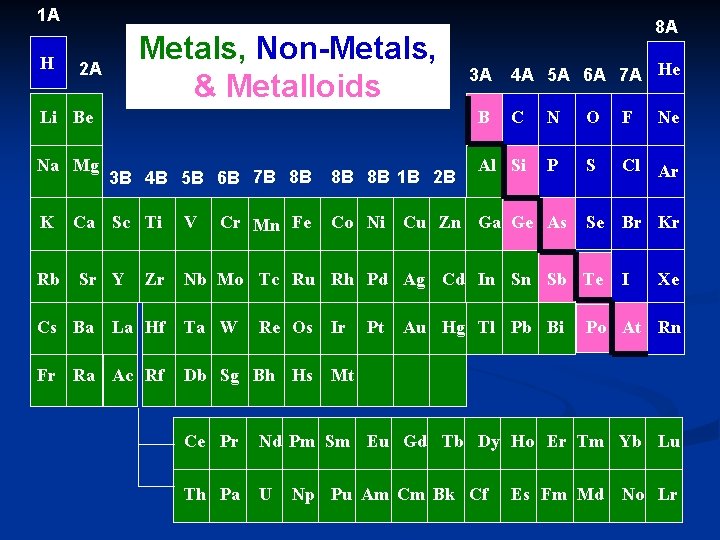
Metals n All elements to the left of the “stairstep” (except Hydrogen) n The most important characteristic of metals is the tendency to lose electrons and form positive ions.

Non-Metals n All elements to the right of the “stairstep” (and hydrogen) n Most important characteristic of nonmetals is the tendency to gain electrons and form negative ions.

Metalloids (Semi-metals) n Have at least one side touching “stairstep” n Exception: Aluminum n Properties of both metals and non-metals
1 A H Metals, Non-Metals, & Metalloids 2 A Li Be Na Mg K Rb 3 A B 3 B 4 B 5 B 6 B 7 B 8 B Ca Sc Ti Sr Y Cs Ba 8 A Zr La Hf Fr Ra Ac Rf V Cr Mn Fe 8 B 8 B 1 B 2 B Co Ni 4 A 5 A 6 A 7 A He C N O F Ne Al Si P S Cl Ar Se Br Kr Cu Zn Ga Ge As Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Ta W Re Os Db Sg Bh Hs Ir Pt Au Hg Tl Pb Bi I Xe Po At Rn Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
1 H Representative Elements 2 s & p blocks Li Be Na Mg K Rb 4 5 Ca Sc Ti V 3 Sr Y Cs Ba 18 Zr La Hf Fr Ra Ac Rf 6 7 8 Cr Mn Fe 9 10 11 12 Co Ni 13 14 15 16 17 He B C N O F Ne Al Si P S Cl Ar Se Br Kr Cu Zn Ga Ge As Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Ta W Po At Rn Re Os Db Sg Bh Hs Ir Pt Au Hg Tl Pb Bi Xe Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
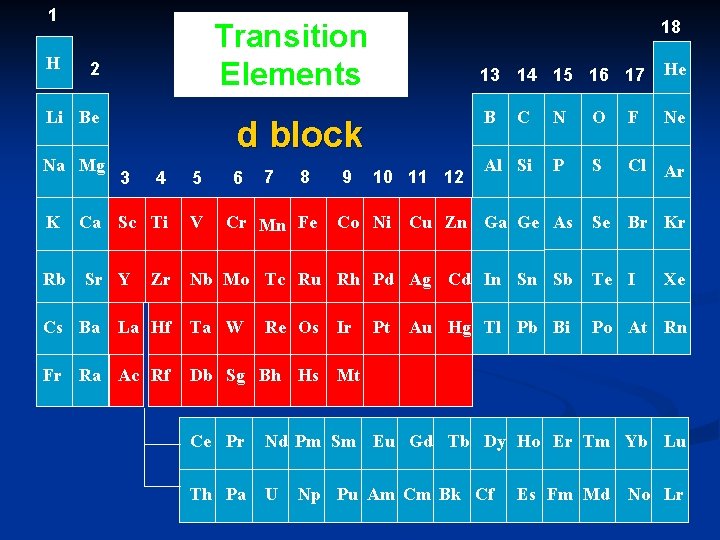
1 H 2 Li Be Na Mg K Rb d block 4 5 Ca Sc Ti V 3 Sr Y Cs Ba 18 Transition Elements Zr La Hf Fr Ra Ac Rf 6 7 8 Cr Mn Fe 9 10 11 12 Co Ni 13 14 15 16 17 He B C N O F Ne Al Si P S Cl Ar Se Br Kr Cu Zn Ga Ge As Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Ta W Po At Rn Re Os Db Sg Bh Hs Ir Pt Au Hg Tl Pb Bi Xe Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
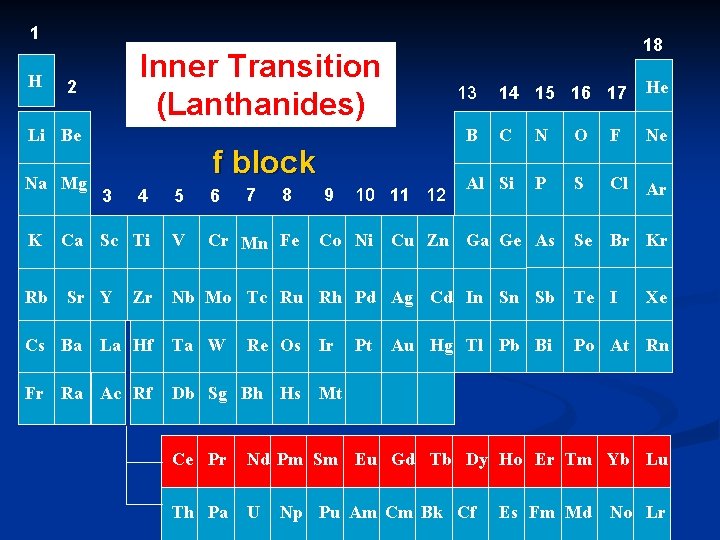
1 H Inner Transition (Lanthanides) 2 Li Be Na Mg K Rb 3 7 8 5 6 Ca Sc Ti V Cr Mn Fe Sr Y Zr La Hf Fr Ra Ac Rf 14 15 16 17 He C N O F Ne Al Si P S Cl Ar Se Br Kr 13 B f block 4 Cs Ba 18 9 10 11 12 Co Ni Cu Zn Ga Ge As Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Ta W Po At Rn Re Os Db Sg Bh Hs Ir Pt Au Hg Tl Pb Bi Xe Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
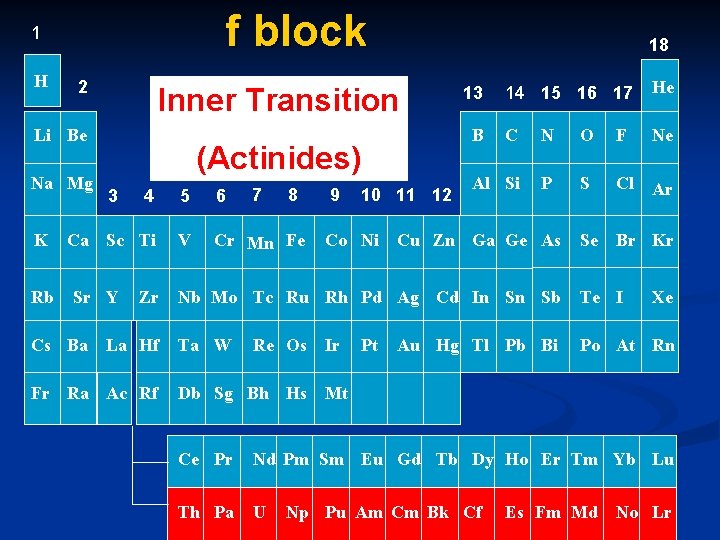
f block 1 H 2 Inner Transition Li Be Na Mg K Rb 3 7 8 5 6 Ca Sc Ti V Cr Mn Fe Sr Y Zr La Hf Fr Ra Ac Rf 9 10 11 12 Co Ni 14 15 16 17 He C N O F Ne Al Si P S Cl Ar Se Br Kr 13 B (Actinides) 4 Cs Ba 18 Cu Zn Ga Ge As Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Ta W Po At Rn Re Os Db Sg Bh Hs Ir Pt Au Hg Tl Pb Bi Xe Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

and do today’s HW
- Slides: 28