Periodic Table Group 1 What is this also
Periodic Table

Group 1 What is this also known as? What do Group 1 metals all have in common in terms of their electronic structure? What properties must they all have as they are metals?

Group 1 metals and oxygen Metal Lithium Symbol Observations Na Potassium What is the general equation for this reaction? What is the trend in the group?
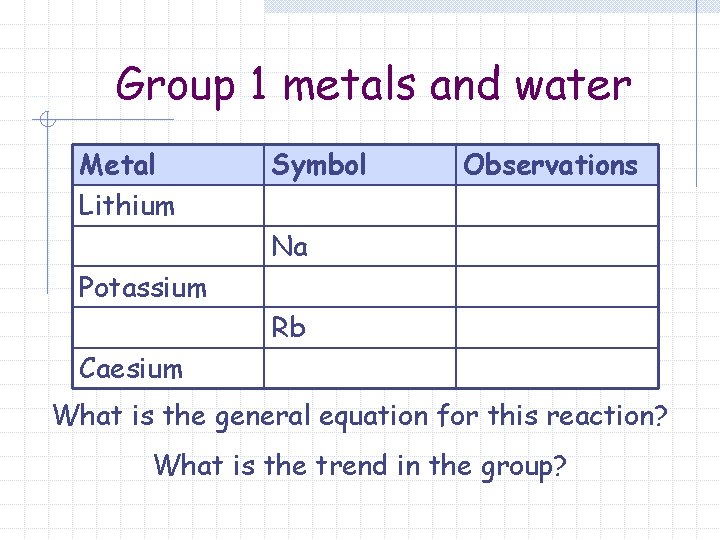
Group 1 metals and water Metal Lithium Symbol Observations Na Potassium Rb Caesium What is the general equation for this reaction? What is the trend in the group?

Are these statements true or false? Metals are on the right of the periodic table. Metal hydroxide is made when metals react with acid. Group 1 metals are kept without air. Hydrogen is produced when group 1 metals react with water. Hydrogen is tested with a glowing splint. Sodium is the most reactive group 1 metal. All group 1 metals have 6 electrons in their outer shells.

Copy and complete Group 1 metals, also known as ………… metals, they are highly reactive as they all have ………. electron(s) in their outer shell. They make ………. cations when they bond ………. . Their compounds are often ………. . solids which dissolve in water to make ………………. solutions. The alkali metals are not very ……………, which can be seen by the fact that they …………… on water. They are kept under oil as they react ……………. . with …………… and …………. in the air.

These are the definitions, but what are the key words? This is the other name for all the group 7 elements. These particles are what controls the chemistry. An atom that has gained electrons. The temperature where a chemical turns from a solid to a liquid, or a liquid to a solid. When a gas turns to a solid or a solid to a gas without turning into a liquid.
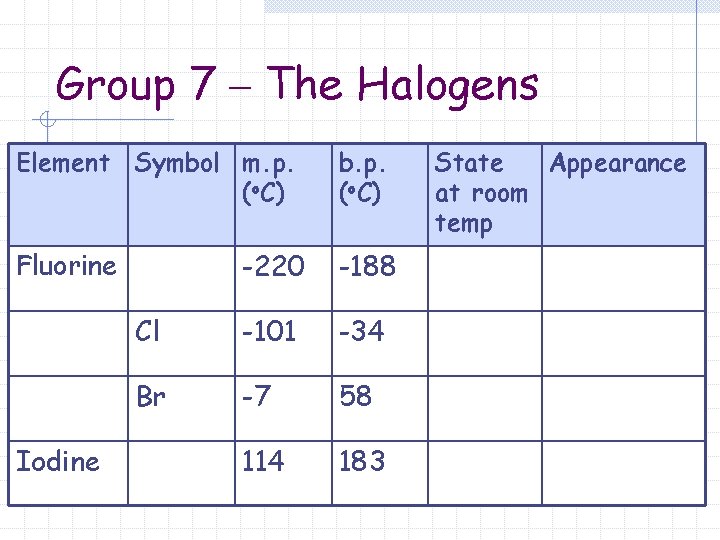
Group 7 – The Halogens Element Symbol m. p. (o. C) -220 -188 Cl -101 -34 Br -7 58 114 183 Fluorine Iodine b. p. (o. C) State Appearance at room temp

Group 7 names In the elemental form they are known as Halogens and their name ends in –ine. In compounds they change their name to end in –ide.
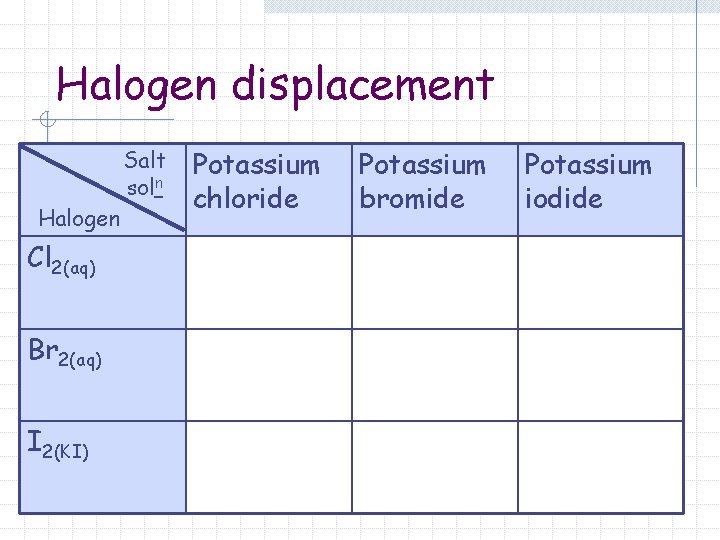
Halogen displacement Salt soln Halogen Cl 2(aq) Br 2(aq) I 2(KI) Potassium chloride Potassium bromide Potassium iodide

Copy and complete All halogens are in group …………… of the Periodic Table. They all have …………… electrons in their outer shell and gain ………. to make …………… in ionic compounds. The halogens will also bond ………… with other non-metals, by …………. . electrons. In the elemental form, they make …………… molecules, which are bonded ……………. . e. g. …………All halogens have coloured gases. There are three trends in group 7, as you go down the group: • • •
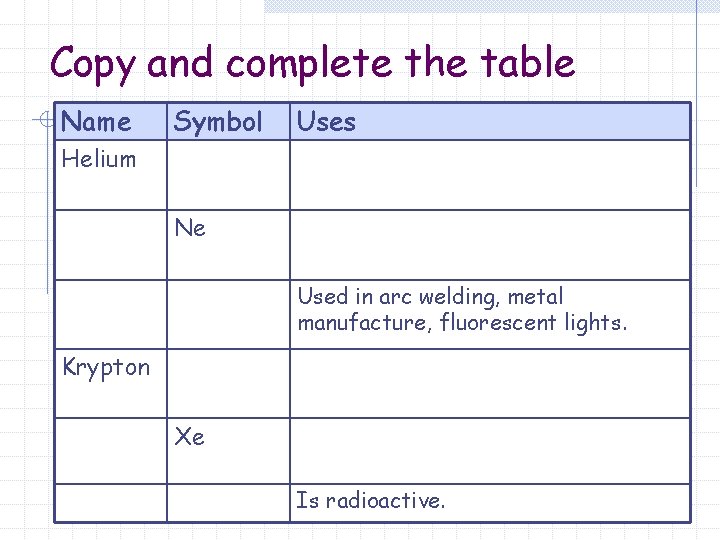
Copy and complete the table Name Symbol Uses Helium Ne Used in arc welding, metal manufacture, fluorescent lights. Krypton Xe Is radioactive.

Physical Properties of Transition Metals Malleable Ductile Sonorous Lustrous Contain the only magnetic elements (Fe, Ni and Co) Thermal and electrical conductors

Chemical Properties of Transition Metals Do they react with air? – think about copper roofs. Do they react with water? What can the metals be used for? Think about catalysts, also mention Fe, Cu, W. What are their compounds like? n Uses of their compounds.
- Slides: 14