Periodic Table Families The Periodic Table All of

Periodic Table Families
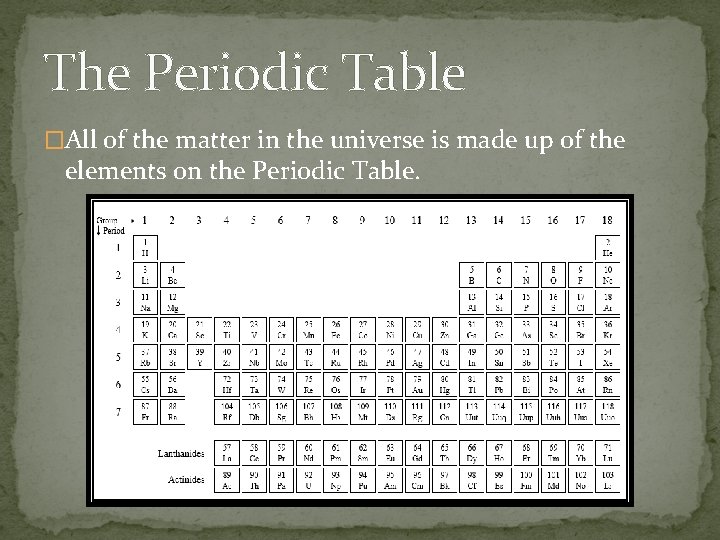
The Periodic Table �All of the matter in the universe is made up of the elements on the Periodic Table.
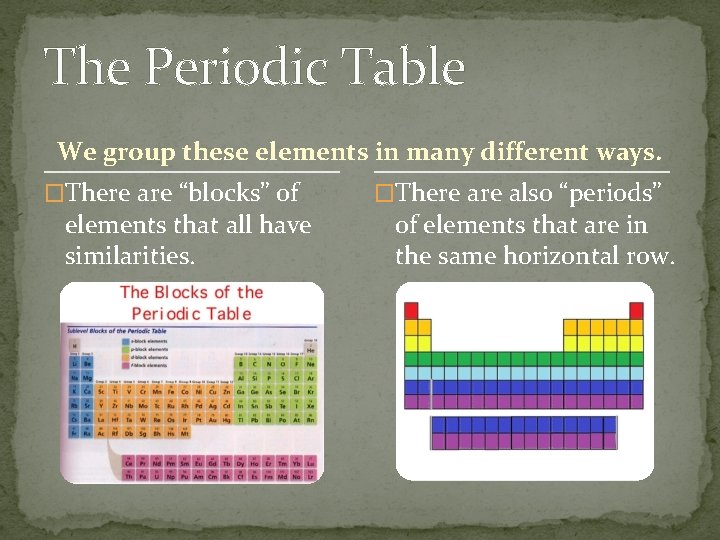
The Periodic Table We group these elements in many different ways. �There are “blocks” of elements that all have similarities. �There also “periods” of elements that are in the same horizontal row.
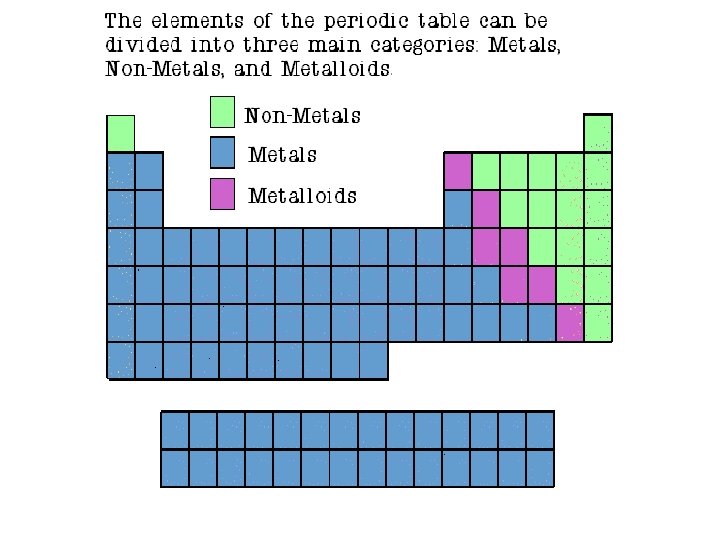

Properties of Metals � Metals are: � Good conductors of heat and electricity � Shiny � Ductile (can be stretched into thin wires) � Malleable (can be pounded into thin sheets). � A chemical property of metal is its reaction with water which results in corrosion.

Properties of Metalloids � Metalloids (metal-like) have properties of both metals and non-metals. � They are solids that can be shiny or dull. � They conduct heat and electricity better than nonmetals but not as well as metals. � Ductile and malleable. Silicon

Properties of Non-Metals � Non-metals are: � Poor conductors of heat and electricity � Non-metals are not ductile or malleable � Dull in luster � Solid non-metals are brittle and break easily. � Many non-metals are Sulfur gases.
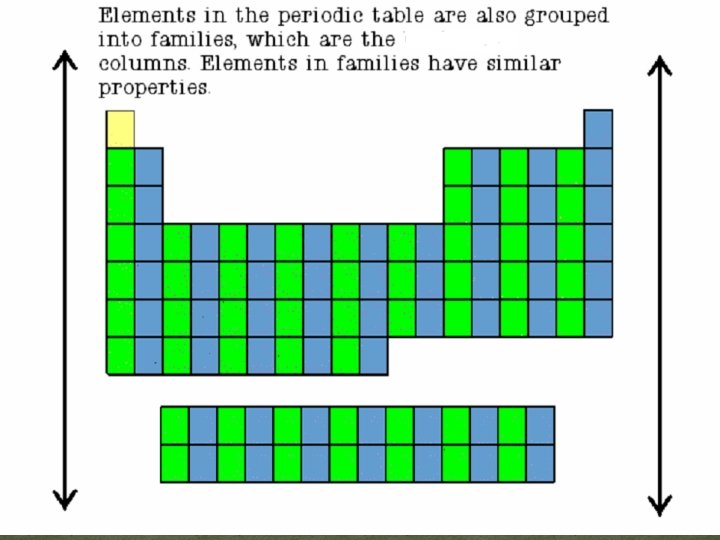
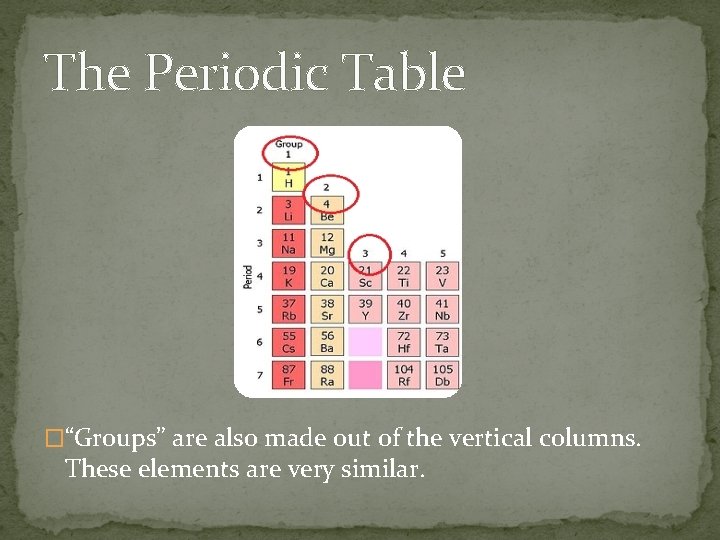
The Periodic Table �“Groups” are also made out of the vertical columns. These elements are very similar.
The Periodic Table �The last way we can group the elements is in to families. ! y l i m a F Just like the mem bers of a have ma fam ny simil arities, t ily element he so Periodic f a family on th e Table ha ve lots i n commo n! �We are going to focus on these families because they group together the elements with the most similar characteristics. Looking at the family will tell you the most information about an element.

Color and Label Activity Please take out your copy of The Periodic Table. Label and Color along as we talk about each part. After you have colored and labeled the family on your Table, fill in the notes about each family’s characteristics on your “Families of the Periodic Table” worksheet.
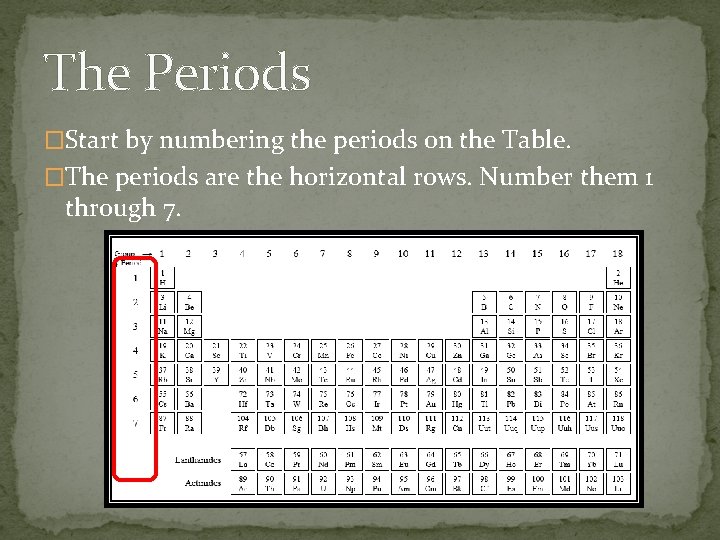
The Periods �Start by numbering the periods on the Table. �The periods are the horizontal rows. Number them 1 through 7.
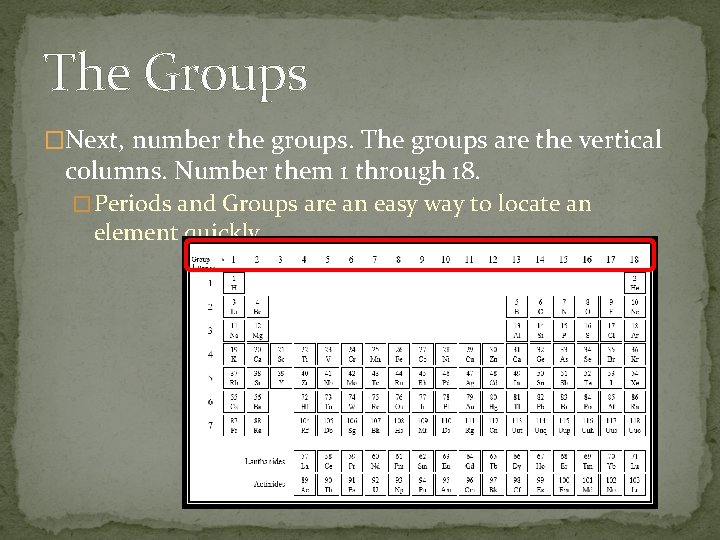
The Groups �Next, number the groups. The groups are the vertical columns. Number them 1 through 18. � Periods and Groups are an easy way to locate an element quickly.
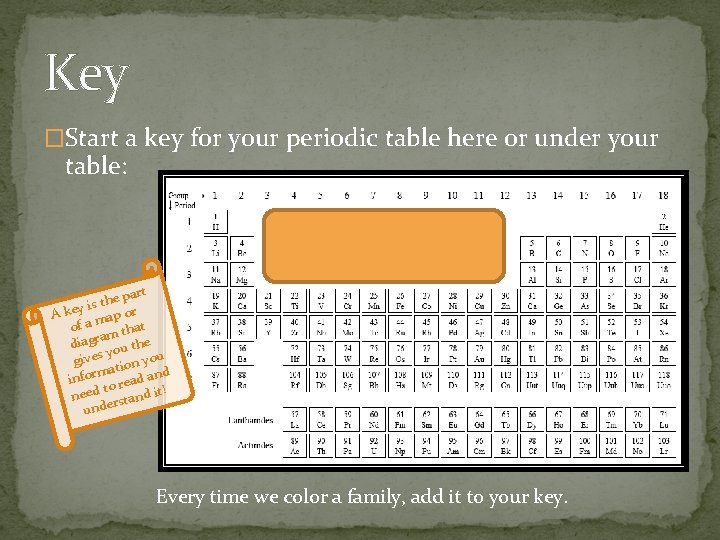
Key �Start a key for your periodic table here or under your table: part e h t is A key map or of a that m a r diag ou the y gives tion you ma d infor o read an t need rstand it! unde Every time we color a family, add it to your key.
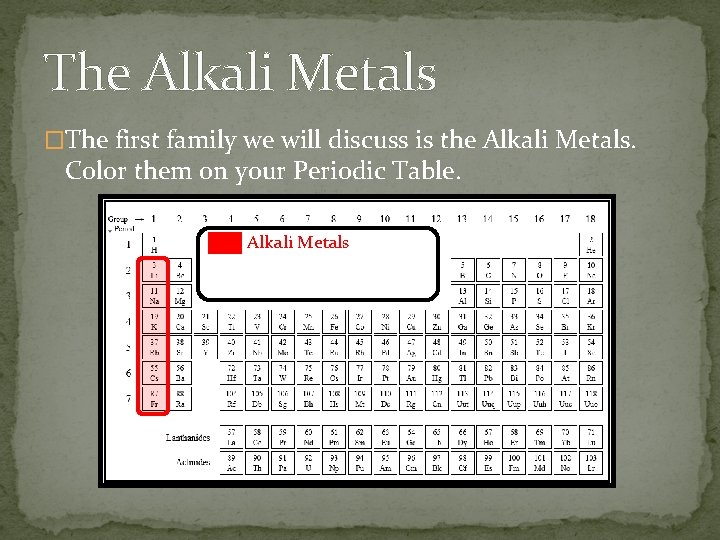
The Alkali Metals �The first family we will discuss is the Alkali Metals. Color them on your Periodic Table. Alkali Metals

The Alkali Metals �All of the alkali metals: �Are shiny and soft. �Are highly reactive with other elements (explosively with water) �Have 1 valence electron, they lose easily. �Can be cut easily. �In nature, are only found in salts - never by themselves. � The most abundant of which is Sodium, which is in the salt we add to our food! Did you know that sodium is a metal?

The Alkaline Earth Metals �Next, color and label the Alkaline Earth Metals and color them in. Alkaline Earth Metals

The Alkaline Earth Metals �All of the Alkaline Earth Metals: �Are shiny and silvery-white. �Are somewhat reactive with other elements. �Easily lose their two outermost electrons. �Have low density, melting point, and boiling point.
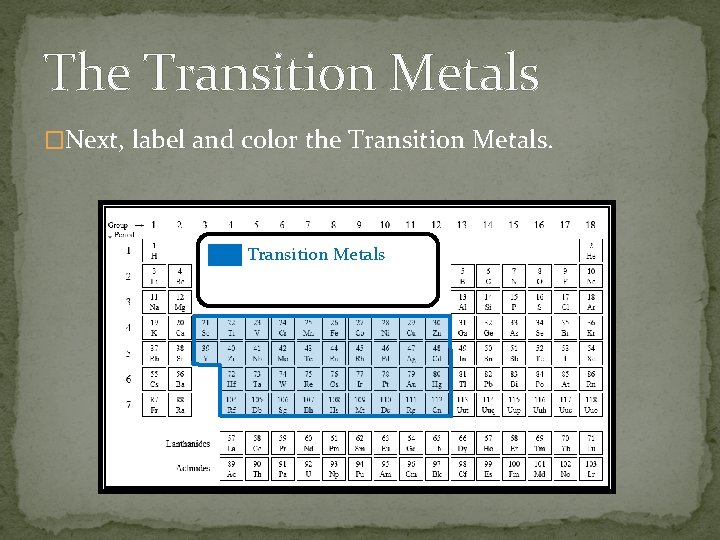
The Transition Metals �Next, label and color the Transition Metals

The Transition Metals �All of the Transition Metals: �Are malleable and ductile. �Are very good conductors of heat and electricity �Are very dense. �Have a high boiling point and a high melting point. �Other than the metals in group 12 – they have a lower boiling and melting point than the others. HG = liquid at room temperature.

The Other Metals �Now, label and color the Other Metals. The Other Metals

The Other Metals �The Other Metals: �Are the metals that don’t share characteristics with any other family of metals. �Are also malleable and ductile. �Have a semi-high density. �Are opaque. �Are solid at room temperature.
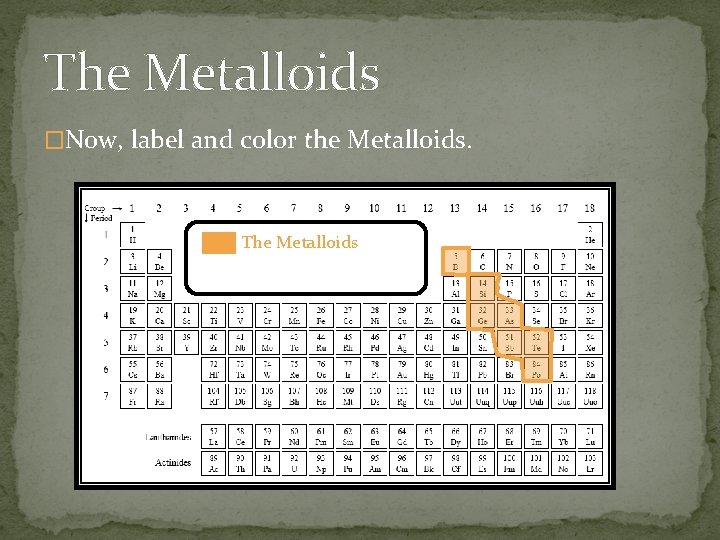
The Metalloids �Now, label and color the Metalloids. The Metalloids

The Metalloids �The Metalloids: �Are the elements in between the metals and the non-metals. � Share similarities with both metals and nonmetals. �Most are semi-conductors.
The Non-Metals �Now, label and color the Non-Metals. The Non-Metals

The Non-Metals �All of the Non-Metals: �Do not conduct heat or electricity well. �Some are gases, like Oxygen. �Some are solids (Carbon) and are very brittle. �Have no luster and reflect no light.

The Halogens �Next, label and color the Halogens. The Halogens

The Halogens �All of the Halogens: �Are non-metallic. �Are toxic. �Are highly reactive - have 7 valence electrons. �Occur in solids, liquids, and gases at room temperature. �They are never found free in nature.

The Noble Gases �Next, label and color the Noble Gases. The Noble Gases

The Noble Gases �All of the Noble Gases: �Almost never react with any other element. �Have full outer shells. �Are very stable. �Are all gases. �Are odorless and colorless. �Have low melting and boiling points.

The Rare Earth Elements � Lastly, label and color the Rare Earth Elements. The Rare Earth Elements

The Rare Earth Elements �All of the Rare Earth Elements: �Are referred to as the lanthanide and actinide series. �Are mostly synthetic (man-made). �Are a part of group 3 on the Periodic Table. �Are metals. �And are sometimes called “Rare Earth Metals” instead of Rare Earth Elements.

Hydrogen �Sits atop Family AI, but is not a member of that family. �A gas at room temperature. �Has one proton and one electron in its one and only energy level. �Only needs 2 electrons to fill up its valence shell.

Boron Family � Named after the first element in the family. � Have 3 valence electrons. � Includes a metalloid (boron), and the rest are metals. � Includes the most abundant metal in the earth’s crust (aluminum).

Carbon Family �Have 4 valence electrons. �Includes a non-metal (carbon), metalloids, and metals. �Carbon is called the “basis of life. ” �An entire branch of chemistry devoted to carbon compounds called organic chemistry.

Nitrogen Family � Named after the element that makes up 78% of our atmosphere. � Includes non-metals, metalloids, and metals. � Have 5 valence electrons. They tend to share electrons when they bond.

Oxygen Family � Have 6 valence electrons. � Share electrons when forming compounds. � Oxygen is the most abundant element in the earth’s crust. It is extremely active and combines with almost all elements.
The End!
- Slides: 38