Periodic Table Families on the Periodic Table The
Periodic Table

Families on the Periodic Table • The families are the columns. • Elements can be grouped into families based: – chemical properties – Valance electron number • Each family has a certain valence electron number. How do you solve for the valence number? – Count the column it is in excluding the Transition Metals and that is the valence number.

What information can you find in each row? • As you go across the row from left to right the elements become less reactive. • As you go down the rows from top to bottom the atom becomes bigger. Why? – The atomic number increases having more protons so therefore the number of electrons increases as does the size of the atom.

Three Types of Matter are found on the Periodic Table. . . • Solids – Ex: Gold, Copper, Iron, Sodium, Meitnerium, Einstinium, Carbon • Liquids – Ex: Mercury, Bromine • Gas – Radon, Chlorine, Nitrogen, Helium, Hydrogen

Understanding the Periodic Table • Metals – Are found on the left side of the staircase Physical Properties: Chemical Properties: Luster Ductile Malleable Conducts heat Conducts electricity corrosive

Nonmetals • Location: to the right of the staircase Physical Properties: Chemical Properties: no luster high electron affinity dull in appearance brittle not ductile and malleable low density values low melting points less reactive than metals

Metalloids • Location: found on both sides of the staircase • Include: B, Si, Ge, As, Sb, Te, Po and At. • Properties: they have properties of both metals and nonmetals. • Metalloid means metallike. Physical Properties: solid can be shiny or dull can conduct heat and electricity ductile and malleable
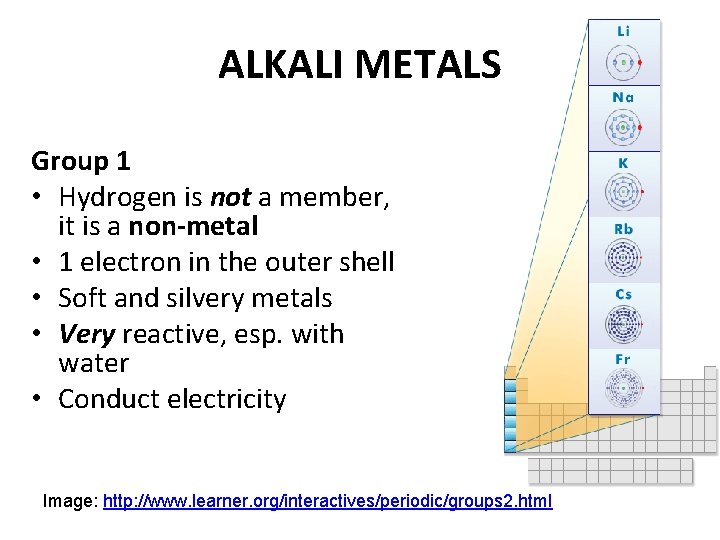
ALKALI METALS Group 1 • Hydrogen is not a member, it is a non-metal • 1 electron in the outer shell • Soft and silvery metals • Very reactive, esp. with water • Conduct electricity Image: http: //www. learner. org/interactives/periodic/groups 2. html
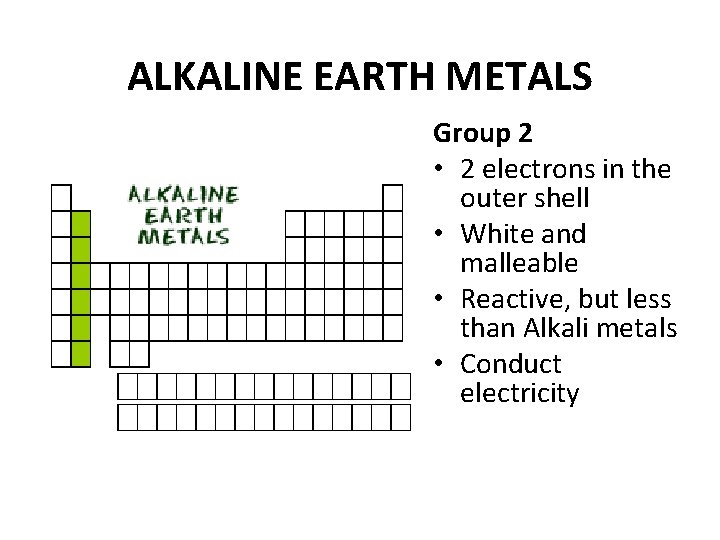
ALKALINE EARTH METALS Group 2 • 2 electrons in the outer shell • White and malleable • Reactive, but less than Alkali metals • Conduct electricity
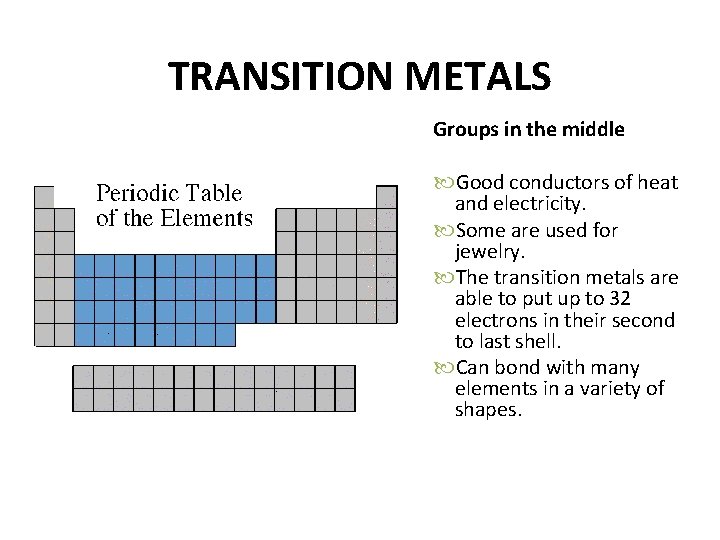
TRANSITION METALS Groups in the middle Good conductors of heat and electricity. Some are used for jewelry. The transition metals are able to put up to 32 electrons in their second to last shell. Can bond with many elements in a variety of shapes.
BORON FAMILY Group 3 • 3 electrons in the outer shell • Most are metals • Boron is a metalloid
CARBON FAMILY Group 4 • 4 electrons in the outer shell • Contains metals, metalloids, and a non-metal Carbon (C)
NITROGEN FAMILY Group 5 • 5 electrons in the outer shell • Can share electrons to form compounds • Contains metals, metalloids, and non-metals
OXYGEN FAMILY Group 6 • 6 electrons in the outer shell • Contains metals, metalloids, and non-metals • Reactive
Halogens Group 7 • 7 electrons in the outer shell • All are nonmetals • Very reactive are often bonded with elements from Group 1
Noble Gases Group 8 Exist as gases Non-metals 8 electrons in the outer shell = Full Helium (He) has only 2 electrons in the outer shell = Full Not reactive with other elements
Rare Earth Metals • Some are Radioactive • Includes the Lanthanoid Series and Actinoid Series • The rare earths are silver, silvery-white, or gray metals. • Conduct electricity
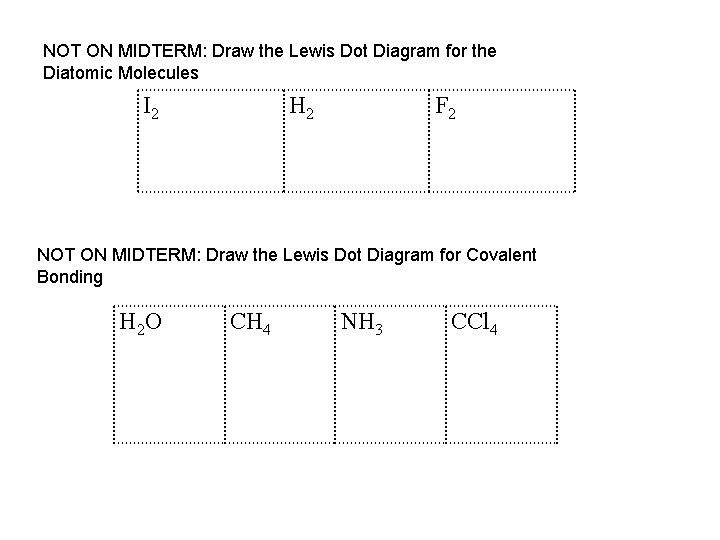
NOT ON MIDTERM: Draw the Lewis Dot Diagram for the Diatomic Molecules I 2 H 2 F 2 NOT ON MIDTERM: Draw the Lewis Dot Diagram for Covalent Bonding H 2 O CH 4 NH 3 CCl 4
- Slides: 18