PERIODIC TABLE FAMILIES Characteristics Hydrogen Unique Can act

PERIODIC TABLE FAMILIES Characteristics

Hydrogen ■ Unique – Can act as metal or nonmetal so family of its own ■ Gas ■ 1 Valence Shell electron ■ Oxidation # is + / - 1
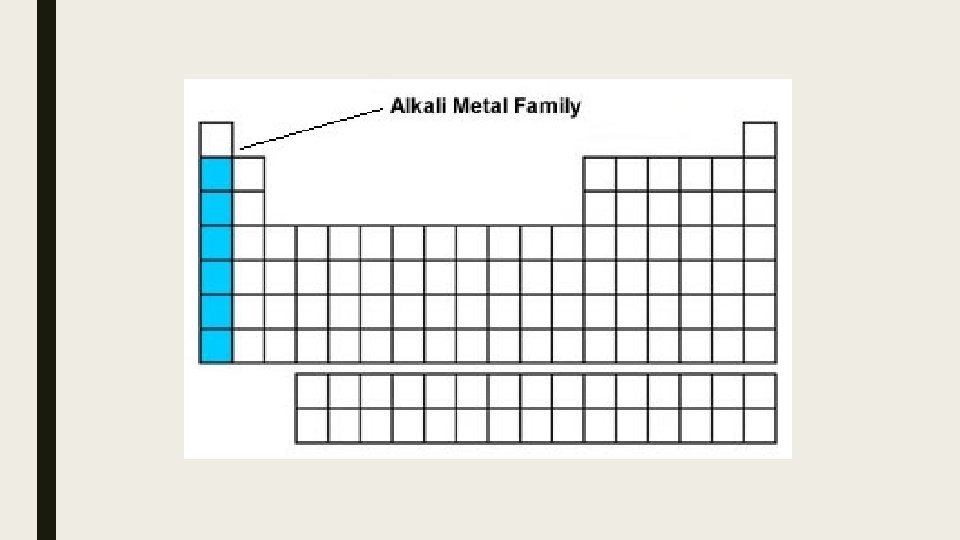

Alkali metals: • • • Most reactive metal family on table Not found as elements in nature – are always part of compounds Solid Silver Shiny Francium most metallic metal (most shiny, most silver) Francium most reactive of all metals Valence Shell electrons = 1 Oxidation Number = 1+ (give their outer most electron away)
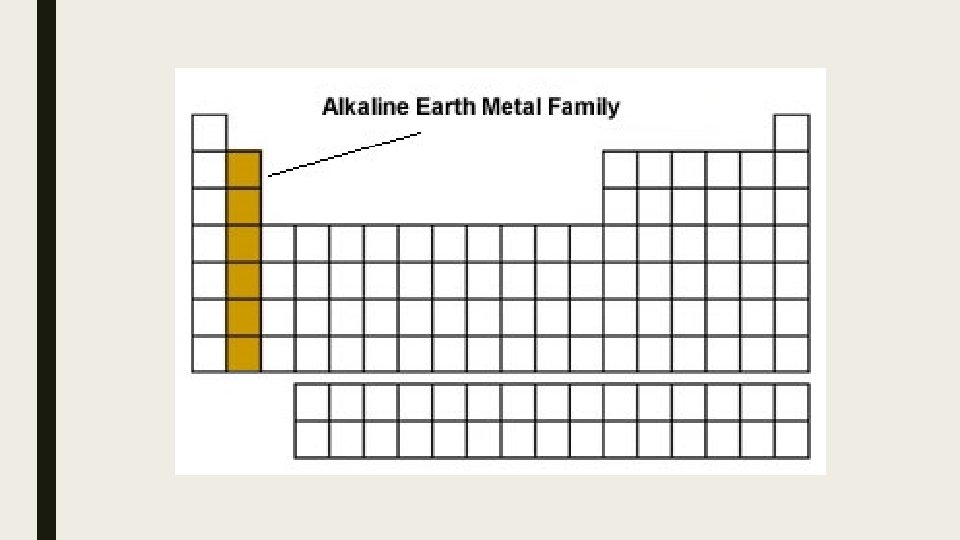

Alkaline Earth metals: • • Second most reactive metal group on table Solid Silver Shiny Not found as elements in nature – found as compounds Valence Shell electrons = 2 Oxidation Number = 2+ (give their outer 2 electrons away)
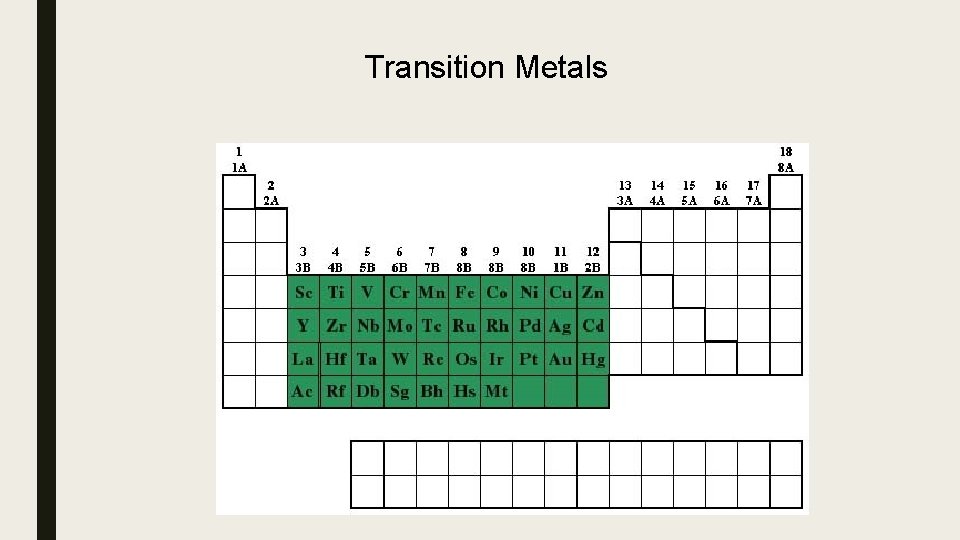
Transition Metals

Transition metals (D- Block) • • 3 rd most reactive metals on table Most are Silver (some exceptions: gold, copper) Shiny Solid (all but one – Mercury is liquid at room temp) Conductive – heat and electricity Malleable – can hammer into a flat sheet Ductile – can stretch into a thin wire Valence shell electrons and oxidation number vary – no pattern

Post Transition Metals (elements in green)

Post transition metals: • (triangle of metals (part of p block)) • • Al, Ga, In, Tl, Sn, Pb, Bi Weakest group of metals Weakest conductors Least metallic – Furthest metals from Francium
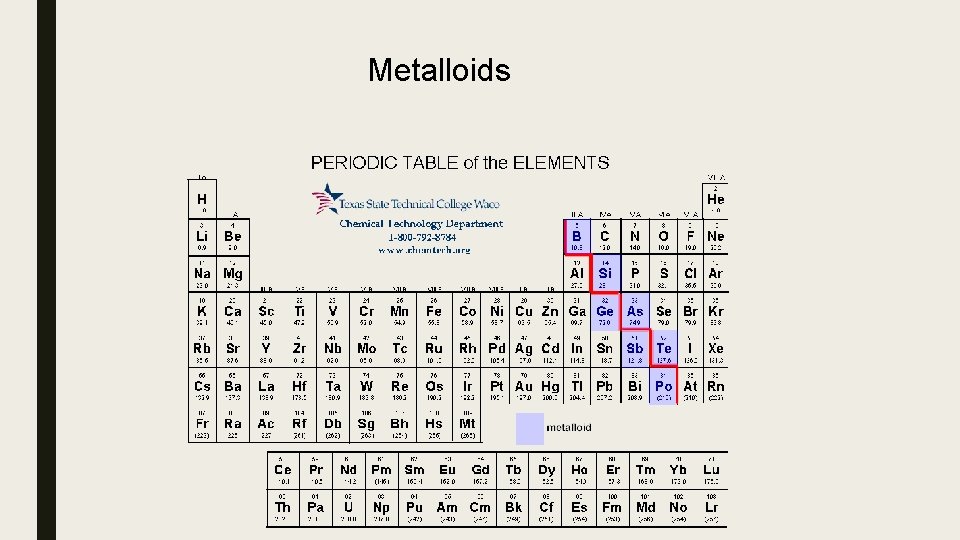
Metalloids

Metalloids – • elements on top and bottom of stair step line. • aluminum (Al) touches line but is part of the post transition metals. • B, Si, Ge, As, Sb, Te ( sometimes Po and At) • mix of metal and nonmetal properties • Boron family has 3 valence electrons and an oxidation # =3
Other Non metals – (white boxes)

Other nonmetals: • C, N, P, O, S, Se • • Not shiny…they are dull Not silver…typically they have color Not malleable or ductile - they are brittle Not conductive • • • Carbon family valence shell electron = 4, oxidation # = + or – 4 Nitrogen family valence shell electrons = 5 oxidation # = -3 Oxygen family valence shell electrons = 6 oxidation # = -2
Halogens

Halogens: • • • • Most reactive nonmetal family Contains all 3 states of matter: F, Cl are gases Br is liquid I , At are solid Most nonmetallic element is Fluorine Most reactive nonmetal is Fluorine Found as compounds in nature as they are very reactive Halogen family valence shell electrons = 7 oxidation # = -1
Noble gases

Noble gases: • All gases at room temp • Very stable • Very Unreactive – found as elements in nature • Noble gas family valence shell electrons = 8 • Exception • Helium only has 2 valence shell electrons oxidation # = 0
Inner Transition Metals

Inner Transition metals (F – Block) • 4 F = lanthanids (lanthanoids) • same properties as transition metals • one is manmade. . symbol is only outlined on periodic table (Pm) • • 5 F = actinids (actinoids) same properties as transition metals many are manmade all are radioactive
- Slides: 20