Periodic Table Electron Configurations The horizontal rows of

Periodic Table Electron Configurations
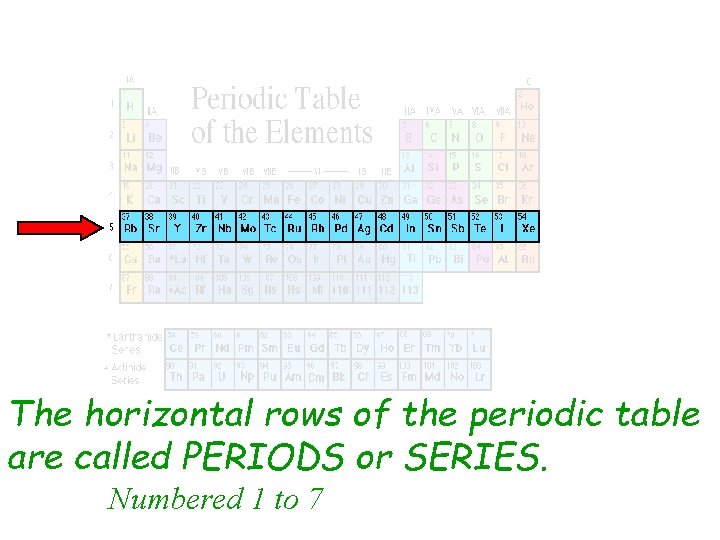
The horizontal rows of the periodic table are called PERIODS or SERIES. Numbered 1 to 7
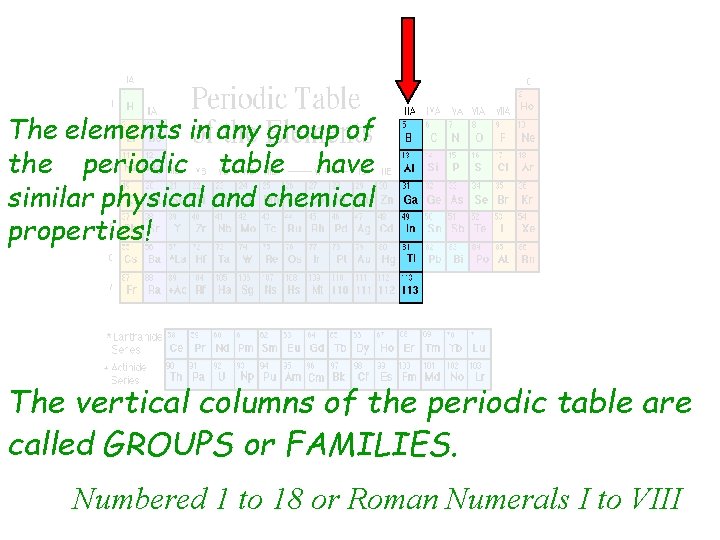
The elements in any group of the periodic table have similar physical and chemical properties! The vertical columns of the periodic table are called GROUPS or FAMILIES. Numbered 1 to 18 or Roman Numerals I to VIII

More on groups • Element properties relate to electronic structures. • Outer electron configurations are the same for the elements in the same group. • Some groups have special names.

H Li 1 3 Na 11 K 19 Rb 37 Cs 55 Fr 87 1 s 1 1 s 22 s 1 Group 1 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 10 5 p 66 s 1 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 66 s 24 f 145 d 106 p 67 s 1
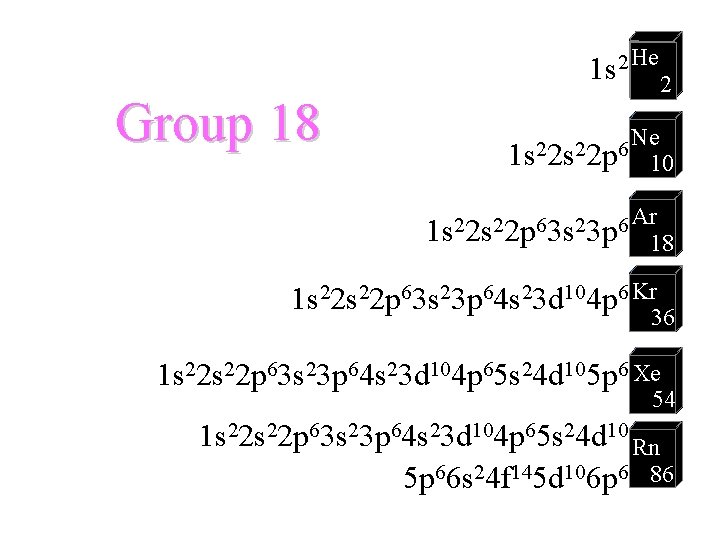
Group 18 1 s 2 He 2 1 s 22 p 6 Ne 10 Ar 2 2 6 1 s 2 s 2 p 3 s 3 p 18 1 s 22 p 63 s 23 p 64 s 23 d 104 p 6 Kr 36 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 6 Xe 54 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 10 Rn 5 p 66 s 24 f 145 d 106 p 6 86

Electron Configurations repeat • The shape of the periodic table is a representation of this repetition. • When we get to the end of the row the outermost energy level is full. • This is the basis for our shorthand electron configurations.
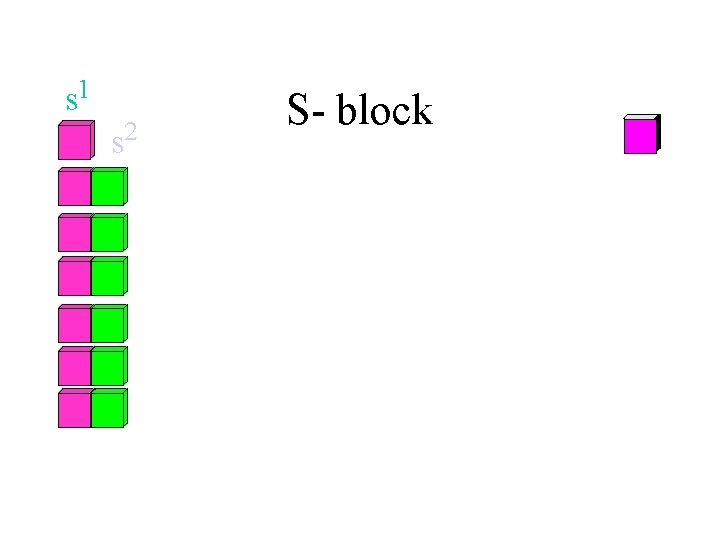
s 1 s 2 S- block
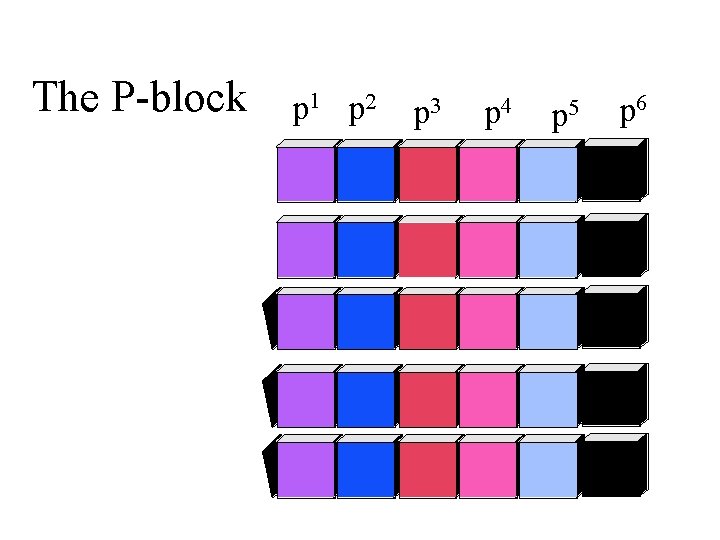
The P-block p 1 p 2 p 3 p 4 p 5 p 6
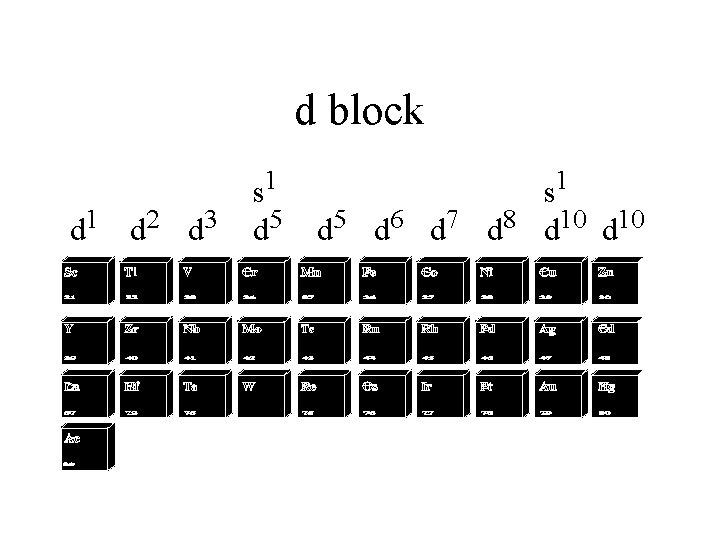
d block 1 d 2 d 3 d s 1 5 6 7 8 10 10 d d d
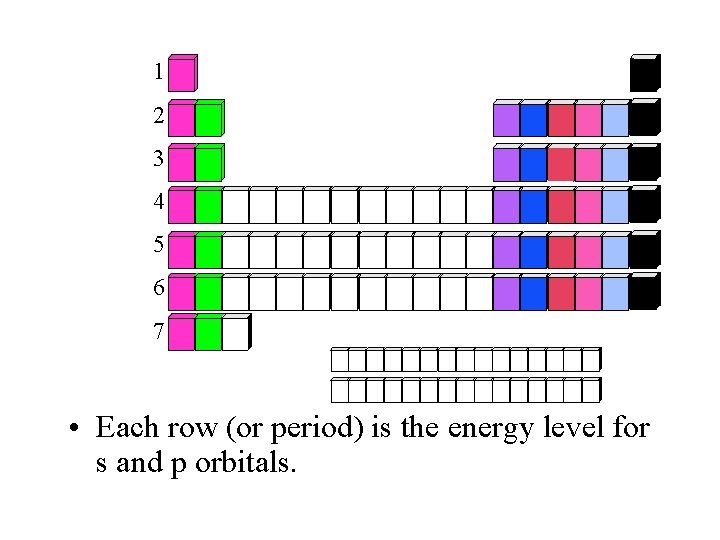
1 2 3 4 5 6 7 • Each row (or period) is the energy level for s and p orbitals.
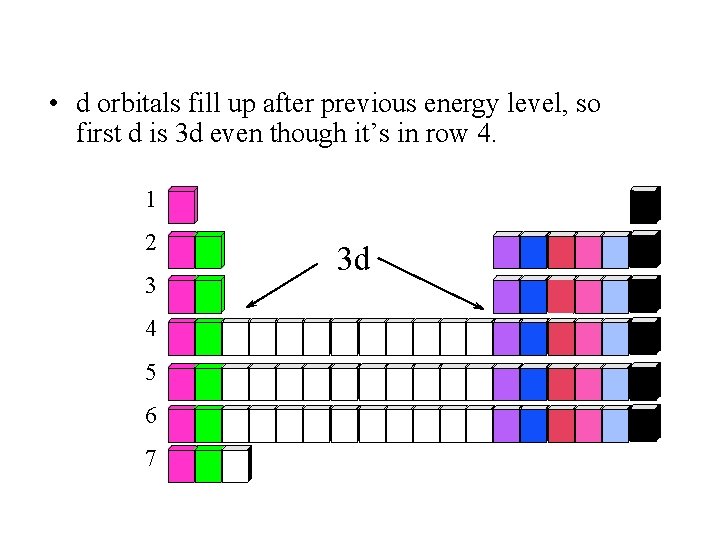
• d orbitals fill up after previous energy level, so first d is 3 d even though it’s in row 4. 1 2 3 4 5 6 7 3 d
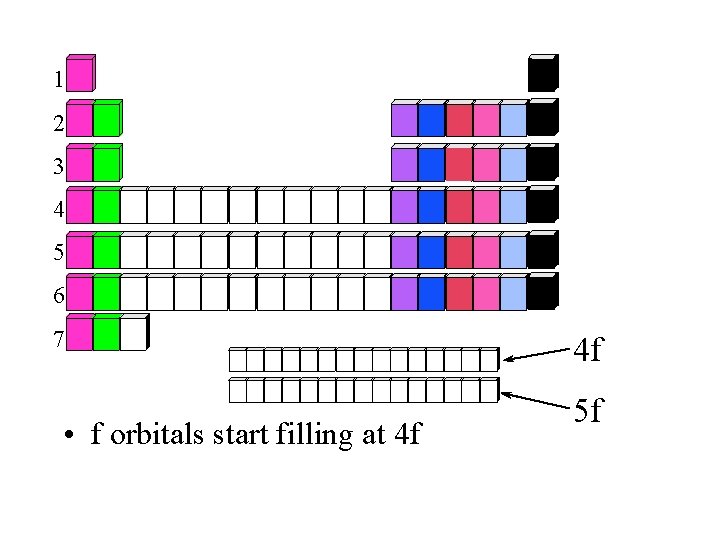
1 2 3 4 5 6 7 • f orbitals start filling at 4 f 4 f 5 f

Summary on groups • Outer electrons are called valence electrons. • Octet Rule – All atoms need 8 valence e-s to be stable • Inner electrons are called core electrons. • Abbreviated electron configurations called Kernel Structures (Noble Gas config): Ex) Calcium is [Ar] 4 s 2 • Note: use only group 18 elements in brackets.
- Slides: 14