PERIODIC TABLE DIMITRI MENDELEEV Mendeleev categorized the elements

PERIODIC TABLE

DIMITRI MENDELEEV Mendeleev categorized the elements in order of relative atomic mass which he noticed was related to their chemical and physical properties, and was able to predict the atomic mass of the asyet-undiscovered elements which belonged in the gaps of the table. http: //www. privatehand. com/flash/elements. html http: //www. cmu. edu/news/stories/archives/ 2016/january/images/periodic_table_large. png

http: //www. youtube. com/watch? v=a. Pq 3 SEte. EJc

ELEMENTS ARE ARRANGED: Vertically into Group or Family Why? Horizontally Into Periods

IF YOU LOOKED AT ONE ATOM OF EVERY ELEMENT IN A GROUP YOU WOULD SEE… Each atom has the same number of electrons in it’s outermost shell. http: //www. youtube. com/watch? v=0 RRVV 4 Diomg
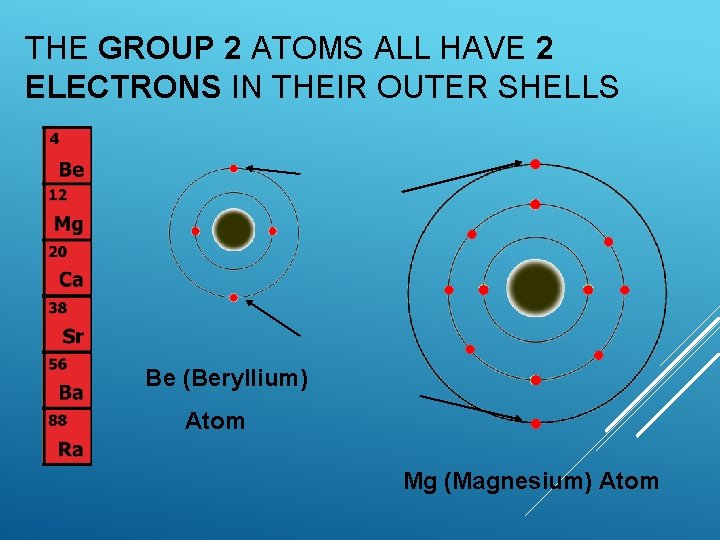
THE GROUP 2 ATOMS ALL HAVE 2 ELECTRONS IN THEIR OUTER SHELLS Be (Beryllium) Atom Mg (Magnesium) Atom

FAMILY OF ELEMENTS The number of outer or “valence” electrons in an atom affects the way an atom bonds. The way an atom bonds determines many properties of the element. This is why elements within a group usually have similar properties.
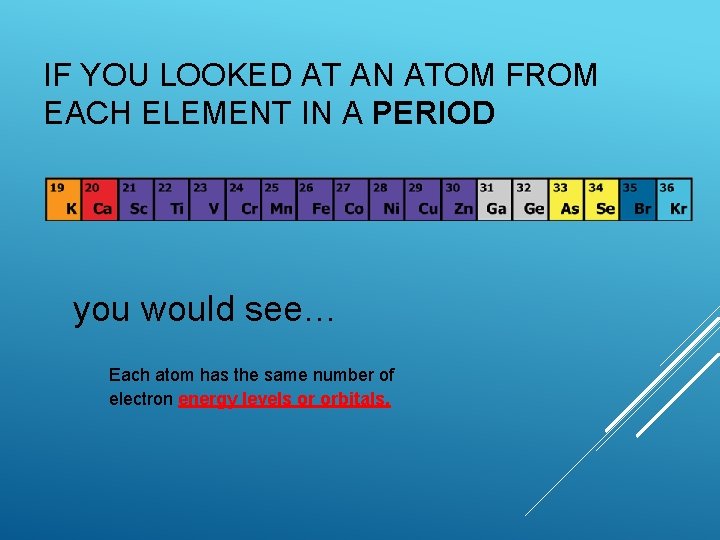
IF YOU LOOKED AT AN ATOM FROM EACH ELEMENT IN A PERIOD you would see… Each atom has the same number of electron energy levels or orbitals.
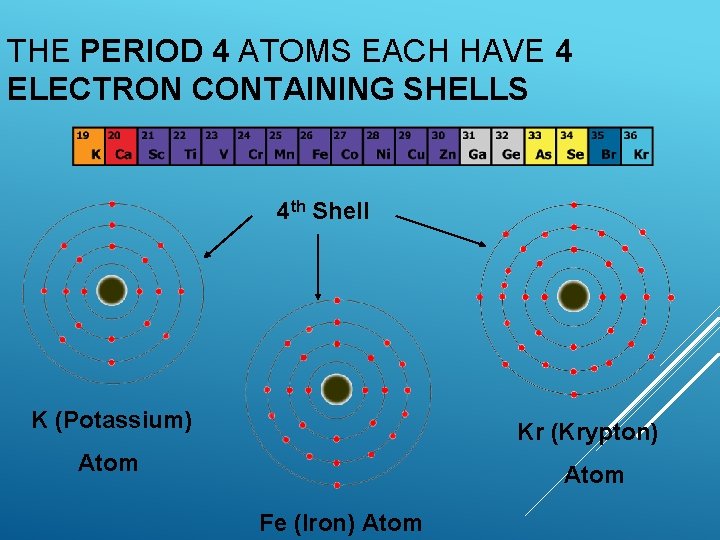
THE PERIOD 4 ATOMS EACH HAVE 4 ELECTRON CONTAINING SHELLS 4 th Shell K (Potassium) Kr (Krypton) Atom Fe (Iron) Atom

The periodic Table is divided into several groups based on the properties of different atoms. EACH GROUP HAS DISTINCT PROPERTIES -Metal-Explosionhttp: //www. dnatube. com/video/1837/Mythbusters-Mc-Guyver-Special. Sodium-

Alkali Metals ALKALI METALS Soft, silvery colored metals Very reactive!!! http: //www. youtube. com/watch? v=m 55 kgy. Ap. Yr. Y
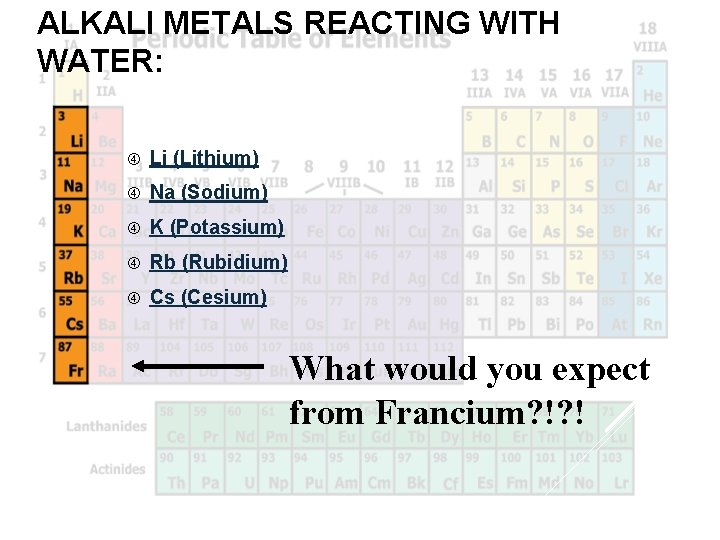
ALKALI METALS REACTING WITH WATER: Li (Lithium) Na (Sodium) K (Potassium) Rb (Rubidium) Cs (Cesium) What would you expect from Francium? !? !

Alkaline Earth Metals ALKALINE EARTH METALS Silvery-White Metals Fairly reactive Many are found in rocks in the earth’s crust
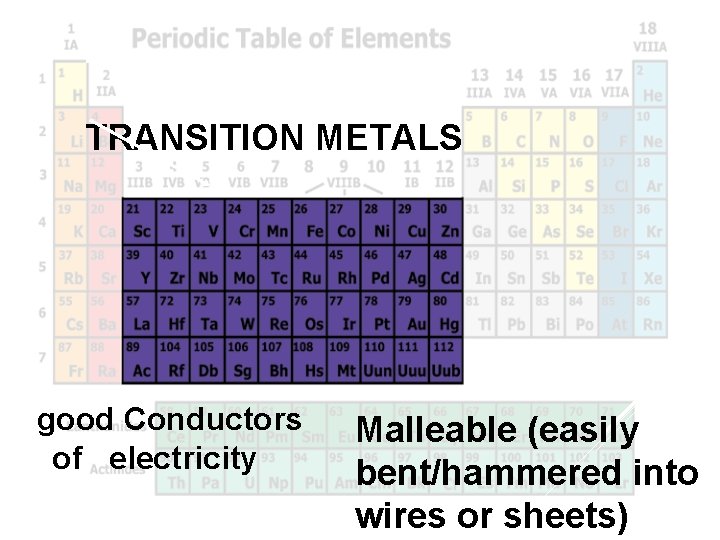
Transition Metals TRANSITION METALS Mgood Conductors Malleable (easily of electricity bent/hammered into wires or sheets)

Metalloids lie on either side of these “stairsteps” They share properties with both metals and non-metals Si (Silicon) and Ge (Germanium) are very important “semi-conductors” http: //www. youtube. com/watch? v=1 PSz. STilu_s
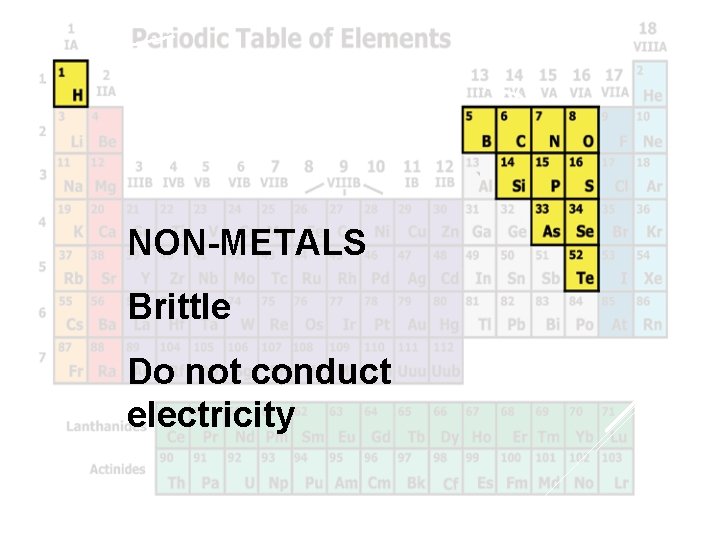
Nonmetals NON-METALS Brittle Do not conduct electricity
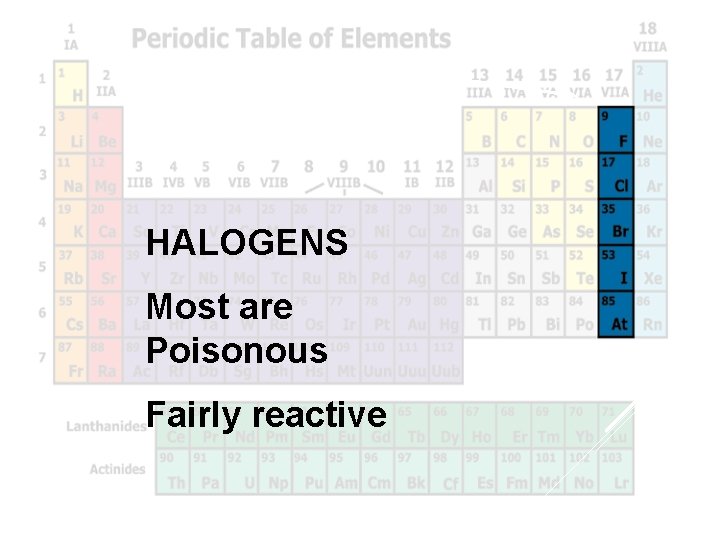
Halogens HALOGENS Most are Poisonous Fairly reactive

Chlorine Gas was used as a chemical weapon during World War I. It was used by the Nazis in World War II. http: //www. youtube. com/watch? v=d 0 z. ION 8 xjb. M

Noble Gases NOBLE GASES Unreactive Gases at room temperature http: //www. youtube. com/watch? v=O-K 1 V 45 Kz 6 I http: //www. youtube. com/watch? v=q. Na. BMv. JXd. J 4 http: //www. youtube. com/watch? v=33 oi. A 4 -fxp 4

THE NOBLE GASES GLOW IN DISTINCTIVE COLORS WHEN USED INSIDE GAS-DISCHARGE LAMPS, SUCH AS NEON LIGHTS. XENON IS COMMONLY USED IN XENON ARC LAMPS, WHICH ARE PRESENT IN FILM PROJECTORS AND AUTOMOBILE HEADLIGHTS DUE TO THEIR NEARLY CONTINUOUS SPECTRUM THAT RESEMBLES DAYLIGHT.
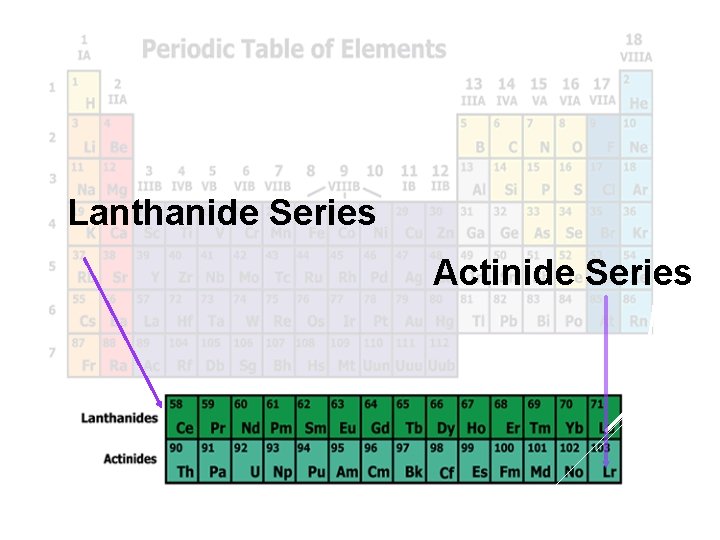
Lanthanide Series Actinide Series

PERIODIC TABLE SUMMARY 1. Elements are arranged by increasing atomic number. 2. FAMILIES/GROUPS ARE COLUMNS, 1 -18 FAMILY NAMES: 1 -ALKALI METALS, 2 - ALKALINE EARTH METALS, 3 -12 TRANSITION METALS, ZIG-ZAG LINE-METALLOIDS, 17 -HALOGENS, 18 -NOBLE GASES 3. PERIODS ARE ROWS, 1 -7 4. For elements in columns 1, 2, and 13 -18, the elements have the same number of outer level electrons. 1 2 13 14 15 16 17 18 Number of electrons in outer level 1 2 3 4 5 6 7 8 (except He) 5. Trends: ionization energy increases across and decreases down. Electronegativity increases across and decreases down. Atomic radius decrease across and increases down. Metallic properties decrease across and increase down. 6. Periodic law states that the chemical properties of an element are a function of the atomic number. 7. The oxidation number for columns 1 2 13 14 15 16 17 18 are +1 +2 +3 +/-4 -3 -2 -1 0 8. The number of the row is the same as the valence electron level. 9. Most elements are metals and are on the left of the zig-zag line. Non-metals are to the right of the zig zag line. 10. Informtion in each box: Name, symbol, atomic number, atomic mass.
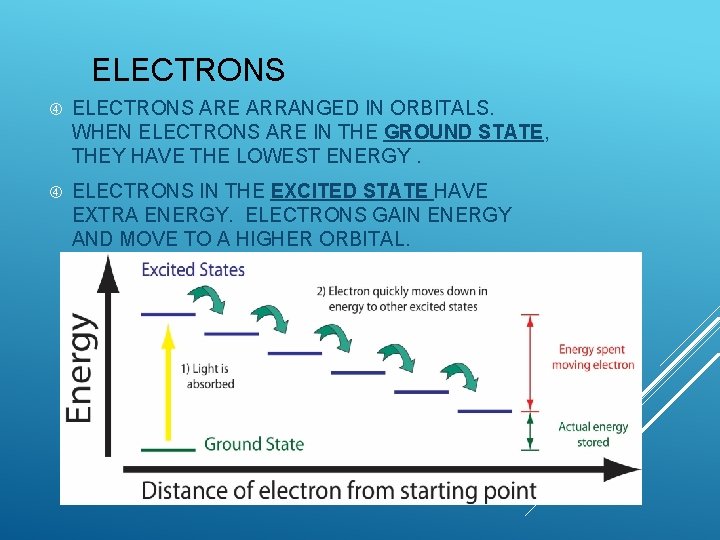
ELECTRONS ARE ARRANGED IN ORBITALS. WHEN ELECTRONS ARE IN THE GROUND STATE, THEY HAVE THE LOWEST ENERGY. ELECTRONS IN THE EXCITED STATE HAVE EXTRA ENERGY. ELECTRONS GAIN ENERGY AND MOVE TO A HIGHER ORBITAL.

ELECTRONS GAIN ENERGY BY ABSORBING LIGHT OR HEAT. ELECTRONS MUST RELEASE THIS EXTRA ENERGY BY GIVING OFF PHOTONS ARE ENERGY IN THE FORM OF LIGHT.

ELECTRONS EVERY ELEMENT HAS IT OWN “LIGHT FINGERPRINT”. EACH ELEMENT WILL RELEASE A PHOTON AT A PARTICULAR FREQUENCY WHICH CORRESPONDS TO A COLOR OF LIGHT.

ELECTRONS FIREWORKS http: //www. youtube. com/watch? v=N 7 ss. CM 3 q. M 3 U https: //www. youtube. com/watch? v=N 7 ss. CM 3 q. M 3 U http: //www. youtube. com/watch? v=vpz. EWQK 7 p. OU

FLAME TESTS WHEN AN ELEMENT’S ELECTRONS GAIN ENERGY BY ABSORBING LIGHT OR HEAT, THE ELECTRONS MOVE TO A HIGHER ENERGY LEVEL. THE EXCITED ELECTRON MUST RELEASE THAT ENERGY BECAUSE IT IS UNSTABLE. THE RELEASED ENERGY IS IN THE FORM OF A PHOTON OF LIGHT. EACH ELEMENT WILL DISPLAY A SPECIFIC COLOR OF LIGHT WHEN HEATED. http: //www. youtube. com/watch? v=d 8 hp. Ut. Rns. Yc https: //vimeo. com/6482824 http: //www. youtube. com/watch? v=j. Jv. S 4 uc 4 Tb. U https: //www. youtube. com/watch? v=W 3 x. HWzm. SOf. U

ELECTRON SHIELDING: DECREASE IN ATTRACTION IN ELECTRONS IN OUTER LEVELS Electron shielding due to the inner electrons “shielding” the outer electrons from the nucleus. Only seen down a column, no effect across a row. Further away electron is from nucleus, the greater the shielding. JUST LIKE A LINEMAN BLOCKS, THE INNER ELECTRONS BLOCK FOR THE NUCLEUS https: //www. youtube. com/watch? v=phkc. Jo-94 p. Q
- Slides: 28