Periodic Table Demtri Mendeleev first arranged the periodic
Periodic Table

Demtri Mendeleev first arranged the periodic table arranged them by atomic mass he noticed that they had similar chemical and physical properties but some elements were out of order

Henry Mosley Arranged the elements by atomic number modern table is arranged this way periodic law: the properties of the elements are periodic functions of their atomic number Patterns
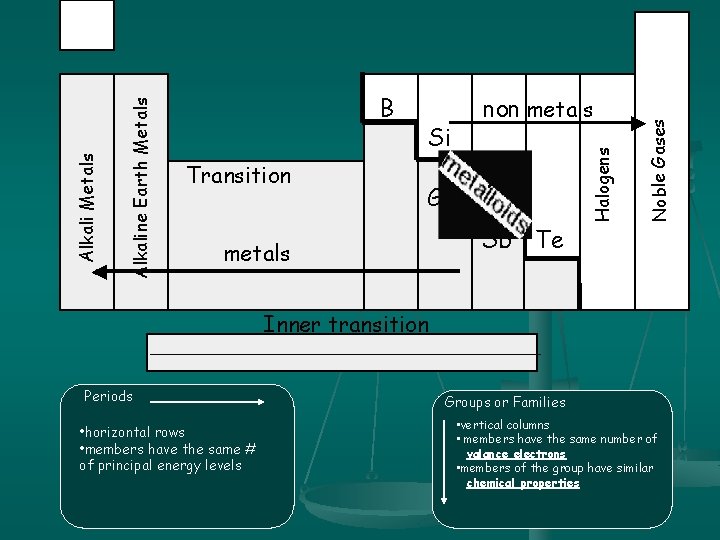
Si Ge metals As Sb Te Noble Gases Transition non metals Halogens Alkaline Earth Metals Alkali Metals B Inner transition Periods • horizontal rows • members have the same # of principal energy levels Groups or Families • vertical columns • members have the same number of valance electrons • members of the group have similar chemical properties

Metals Metalloids Nonmetals • Luster • semi-metals • dull • ductile • touch a side of the staircase • brittle solids • malleable • good conductors of heat and electricity • high melting and boiling pt. Has properties of both metals and nonmetals B, Si, As, Ge, Sb, Te ONLY!!! • poor conductors of heat and electricity • low melting and boiling pt.

Group 1 - Alkali Metals valance electron(s) 1 +1 ion • most active metals • not found in nature by themselves • form stable compounds • can be obtained by electrolysis of fused salt.

Group 2 - Alkaline Earth Metals valance electrons 2 +2 ion • second most active metal • some are not found in nature by themselves

Group 17 - Halogens • valance electrons 7 • most common ion -1 • have all states of matter at room temp. F 2, Cl 2 gases Br 2, liquid I 2, solids • are the most reactive non metals

Group 18 - Noble Gases valance electrons 8 • Inert gases (non reactive) • have the most stable electron configuration- 8 e in the valence shell • Kr and Xe can react with fluorine-they have an oxidation number other than 0

Transition Elements • many positive oxidation numbers • valance electrons are in various energy levels --can lose electrons from more than one energy level • form colored ions in solutions

Diatomic Elements I Bring Clay From Our New Home I 2, Br 2, Cl 2, F 2, O 2, N 2 , H 2 all other elements are monatomic Allotropes same element in various forms Different physical and chemical properties O 2 O 3 C- graphite diamond Liquids Br nonmetal Hg metal Radioactive All elements over #82 have no stable isotopes

Ionization Energy • amount of energy needed to remove an electron NOTE: Leave Room in each box. We will revisit in detail Electronegativity • the ability of an atom to attract an electron TABLE S Periodic Trends patterns on the periodic table Atomic Radius • distance from the nucleus to the outer most energy level Reactivity • the ability of a substance to undergo a change

Ionization Energy • lower the energy -easier to remove • across periods value increases -harder to remove e-’s from non-metals they need more to be stable; easier to remove from metals- they want to get rid of them. • move down a family (group) decrease -harder to remove e-’s closer to the (+) nucleus -(Opposites attract!); easier to remove e-’s farther away shielding by other e-’s

Electronegativity • 0 -4. 0 range (4. 0 is highest) • move across a period increases -non metals want e-’s so they will be attracted to them; metals want to get rid of e-’s so they will not be attracted to them. • move down a family decreases -Harder to attract e-’s when there are other e-’s blocking the pull from the nucleus and repelling the e-’s.

Atomic Radius • period: decrease as you move across because increase in nuclear charge(+) • group: increase as we go down because increasing number of energy levels in an atom

Reactivity • metals: left and down • most active metal Fr • nonmetals; right and up • most active non metal F

IE increases EN increases Radius decrease IE decreases EN decreases Radius increase nonmetal Reactivity

Atomic vs. Ionic Size Metals lose electrons become positive ion is smaller than atom Na Na+ + 1 e Nonmetals gain electrons become negative ions Ion is larger than atom Cl + 1 e Cl-

- Slides: 19