Periodic Table Chemical Families Families on the Periodic

Periodic Table Chemical Families
Families on the Periodic Table • Elements on the periodic table can be grouped into families based on their chemical properties. • Each family has a specific name to differentiate it from the other families in the periodic table. • Elements in each family react differently with other elements.
![Hydrogen: Atom: H Ion: [H]+ * Oils + Hydrogen Fats * Metal + Acid Hydrogen: Atom: H Ion: [H]+ * Oils + Hydrogen Fats * Metal + Acid](http://slidetodoc.com/presentation_image_h2/9ee3825e698a49603e5165592562dc52/image-3.jpg)
Hydrogen: Atom: H Ion: [H]+ * Oils + Hydrogen Fats * Metal + Acid Hydrogen - popping test
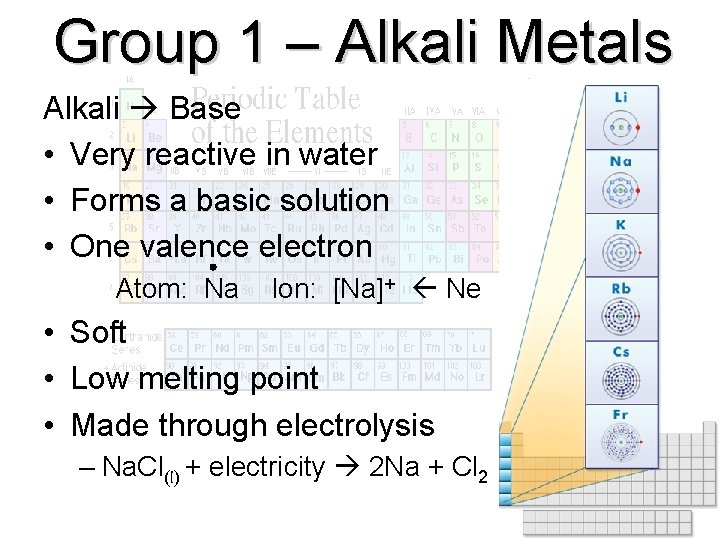
Group 1 – Alkali Metals Alkali Base • Very reactive in water • Forms a basic solution • One valence electron Atom: Na Ion: [Na]+ Ne • Soft • Low melting point • Made through electrolysis – Na. Cl(l) + electricity 2 Na + Cl 2

Group 1 - Alkali Metals • Flame Test • Na yellow • K violet • Li red Complete Questions #6. 1 -6. 5 on p. 205
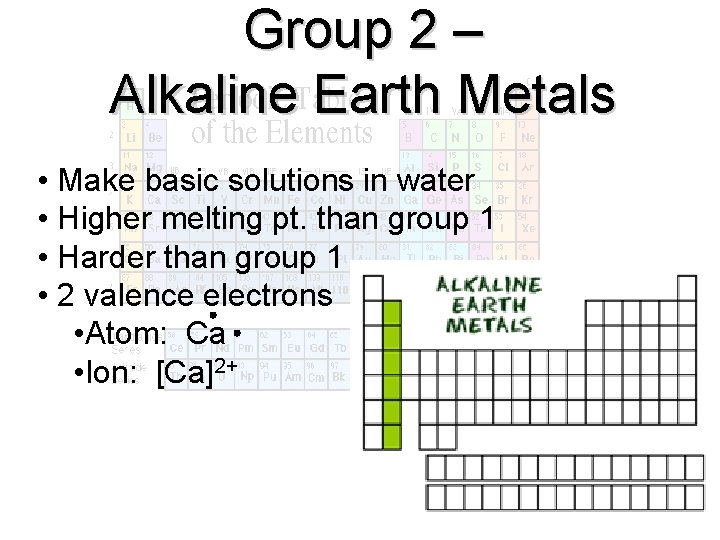
Group 2 – Alkaline Earth Metals • Make basic solutions in water • Higher melting pt. than group 1 • Harder than group 1 • 2 valence electrons • Atom: Ca • Ion: [Ca]2+

Group 2 – Alkaline Earth Metals Flame Test: Ca orange red Sr intense red Ba yellow-green

Group 2 – Alkaline Earth Metals Hard Water: • Ca 2+ or Mg 2+ • reacts with soap to make scum • reacts with Na 2 CO 3 – washing soda to make precipitate Ca 2+ + CO 32 - Ca. CO 3(s) Complete Questions #6. 6 -6. 9 on p. 211

Group 13 - Aluminum • Malleable • Ductile • Less dense than iron • Conducts heat and electricity • Makes an oxide coating for protection
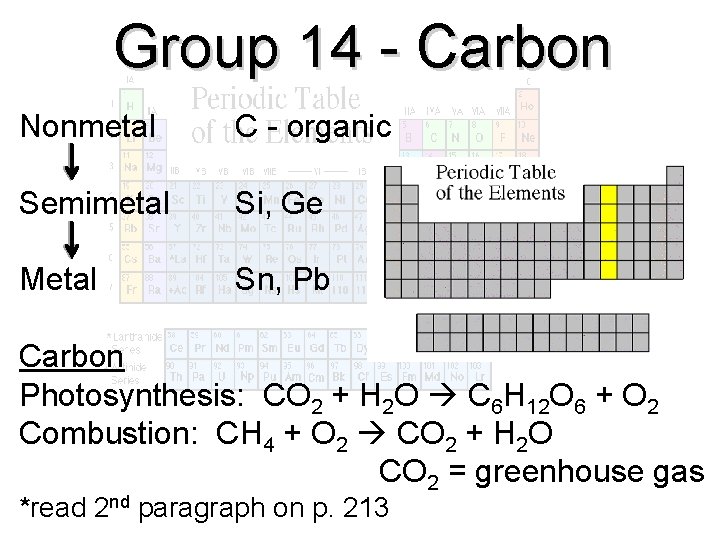
Group 14 - Carbon Nonmetal C - organic Semimetal Si, Ge Metal Sn, Pb Carbon Photosynthesis: CO 2 + H 2 O C 6 H 12 O 6 + O 2 Combustion: CH 4 + O 2 CO 2 + H 2 O CO 2 = greenhouse gas *read 2 nd paragraph on p. 213

Group 14 - Allotropes: • Same element but different structures • Carbon graphite, diamonds, fullerene • Each have different chemical + physical properties

Group 14 - Silicon • Si. C – silicon carbide – very hard – network solid • 2 nd most abundant element on earth
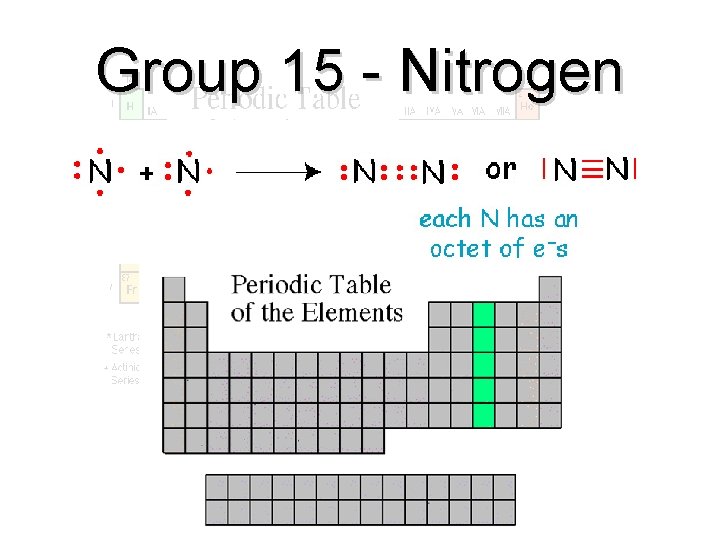
Group 15 - Nitrogen
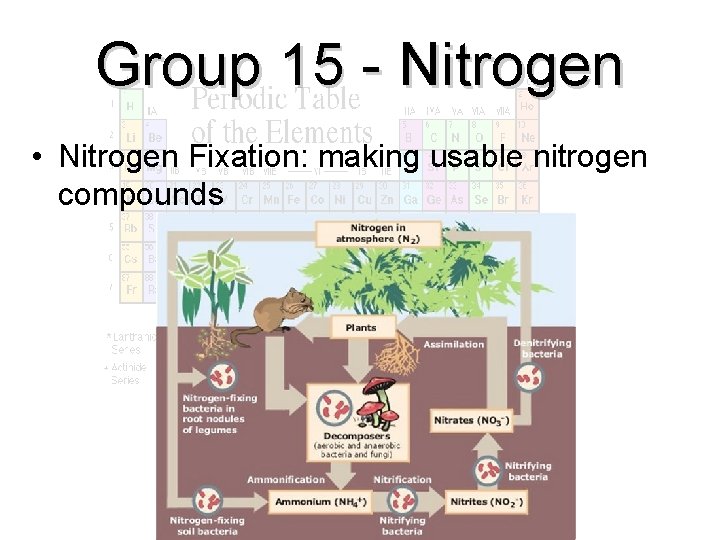
Group 15 - Nitrogen • Nitrogen Fixation: making usable nitrogen compounds
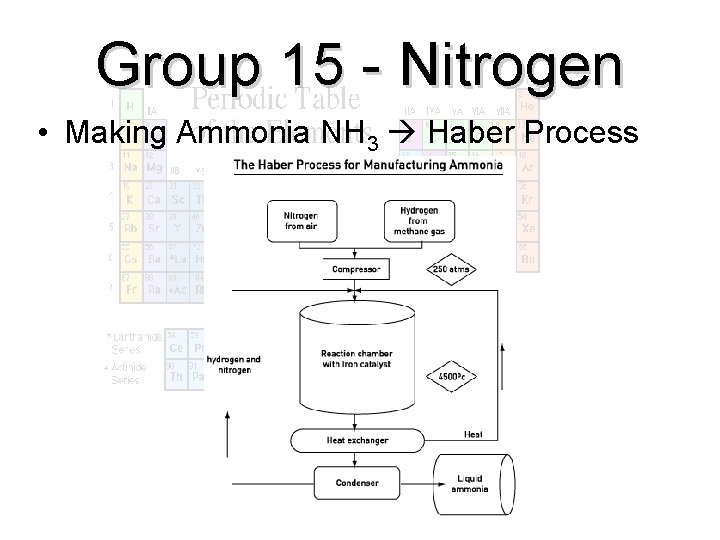
Group 15 - Nitrogen • Making Ammonia NH 3 Haber Process

Group 15 - Nitrogen Allotropes – Phosporous White Red Violet Black

Group 16 - Oxygen – 20% of air – Allotrope O 2 vs O 3 (ozone) Sulfur – Yellow – Allotrope: rhombic, monoclinic, amorphous – S 8 – Acid rain • Burning things with sulfur dioxide SO 2 + H 2 O H 2 SO 4
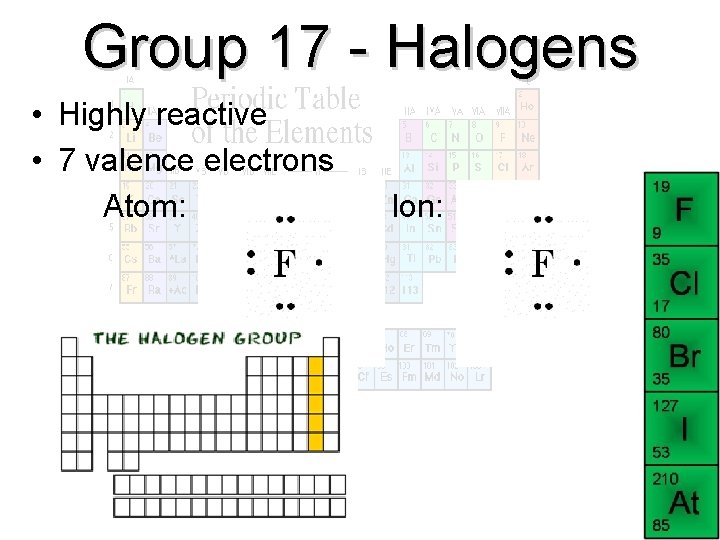
Group 17 - Halogens • Highly reactive • 7 valence electrons Atom: Ion:

Group 18 – Noble Gases • Inert • Stable octet • Monatomic (He) vs Diatomic (H 2, O 2, N 2, Cl 2, F 2, Br 2, I 2) • As you go down group, more attraction, higher Boiling Point

Complete Questions #6. 14 -6. 17 on p. 231 Review Video
- Slides: 20