Periodic Table Chapter 6 History o Dimitri Mendeleev

Periodic Table Chapter 6

History o Dimitri Mendeleev (a Russian scientist) usually gets credit for arranging the elements on the periodic table according to increasing atomic mass. o His periodic law stated that the properties of elements were periodic functions of their increasing atomic mass.

Dimitri Mendeleev

Groups or families of elements have similar properties. o This is due to similar outer electron arrangement. o Outer electrons are called valence electrons. o They are vertical columns on the periodic chart.

Families and Groups

Periods o Elements are also arranged in horizontal rows called periods. o The row or period signifies the number of energy levels being occupied by the electrons of a particular element.

Henry Mosely o A young, English chemist who used X-ray diffraction to determine that the number of protons was responsible for the identity of an element. o Today’s periodic chart is arranged according to increasing atomic number. o Modern periodic law states that the properties of the elements are periodic functions of their increasing atomic numbers.

Henry Moseley

Trends on the Periodic Table

Ionization Energy o Energy required to remove the outermost electron from an atom. o Measured in k. J/mol. o Generally increases from left to right across a period. o Decreases as you move down a column.

Electron Affinity o the attraction of an atom for an electron. o Same unit and same trend as ionization energy. o Metals want to get rid of electrons; nonmetals WANT electrons!

Electronegativity o Relative tendency of an atom to attract electrons to itself when bound with another atom. o Same trend, NO UNIT!
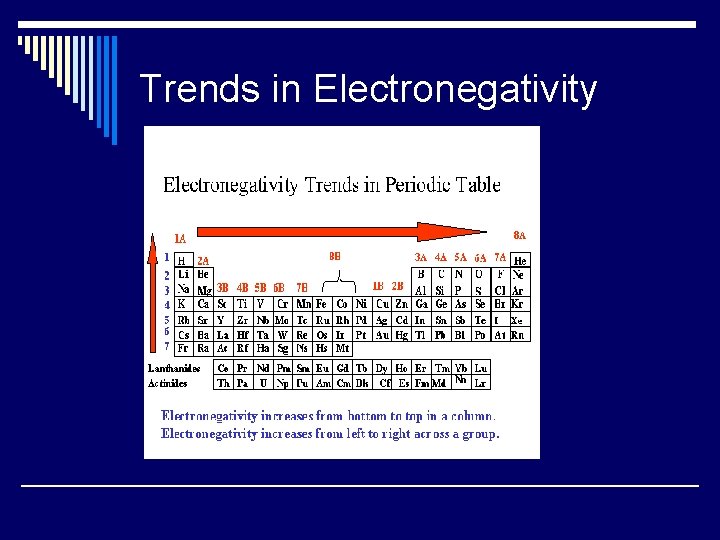
Trends in Electronegativity
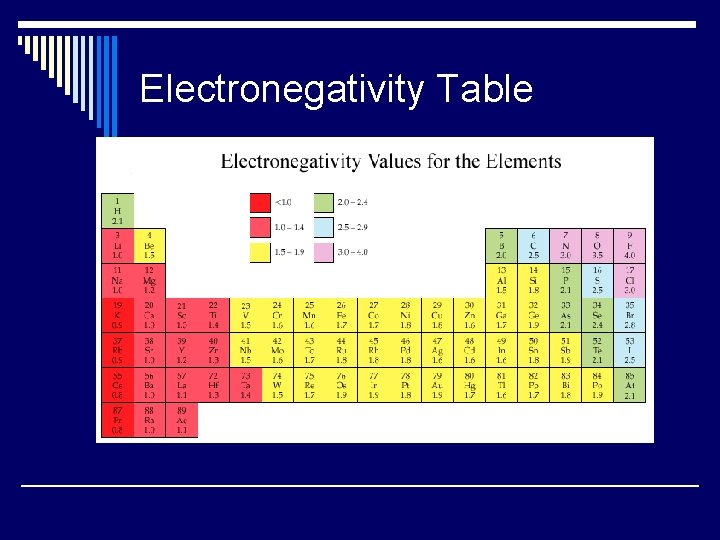
Electronegativity Table

Atomic Radius o The distance from the center of an atom’s nucleus to its outermost electron. o Radius size tends to decrease across a period due to increasing nuclear charge (this attraction pulls the electrons in closer, making the “circle” get smaller). o Size increases as you move down a group due to the addition of another energy level (like adding a story to a house)

Atomic Radius

Ionic Radius o Metallic ions lose electrons and are smaller compared to the atoms from which they come from. o For ex. , Na+ is smaller than Na since it loses an electron! o Nonmetallic ions gain electrons and are LARGER compared to the atoms they come from. Cl- is larger than Cl

Ionic Radius
- Slides: 18