PERIODIC TABLE Arrangement Mendeleev arranged the first periodic
PERIODIC TABLE

Arrangement • Mendeleev arranged the first periodic table. • Arranged by increasing atomic mass. • Periodic Law: arranged in order of increasing atomic number. • Repetition of properties.
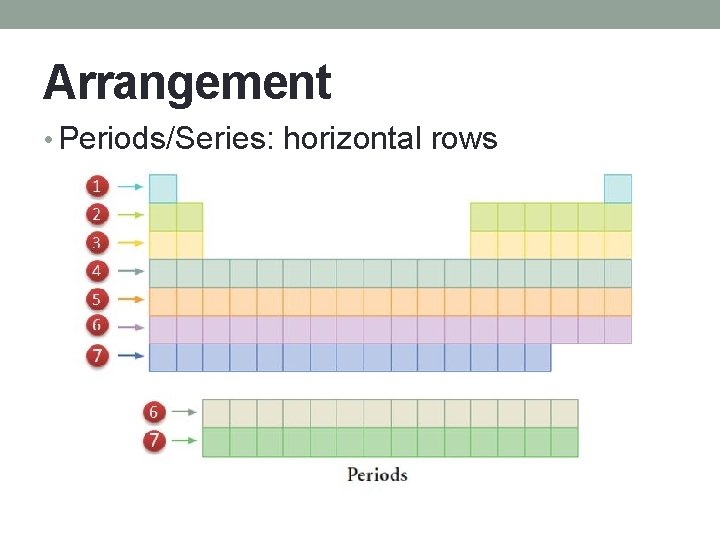
Arrangement • Periods/Series: horizontal rows
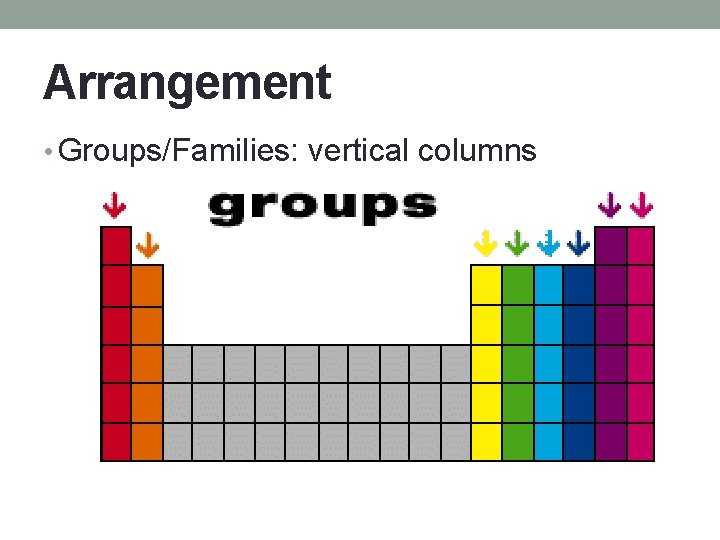
Arrangement • Groups/Families: vertical columns
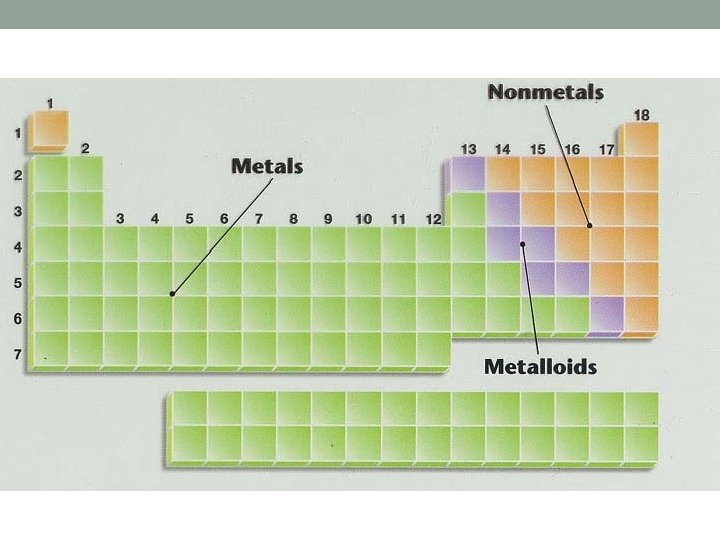

Metals Physical Properties • Good electrical • Solids at room conductors • Good heat conductors • Lustrous • Ductile • Malleable temperature • High melting point • High density • High boiling point

Metals Chemical Properties • Lose valence e- easily • Low Electronegativity • Corrode easily

Alkali Metals • React with water • Not found in nature • Soft • 1 valence electron • Oxidation of +1 • Loses 1 electron to form bonds • Lower melting and boiling points

Alkaline Earth Metals • React with Halogens to form salts • Not found in nature • Low density • Found in molecules in earth’s crust • 2 valence electrons • Oxidation of +2 • Loses 2 electrons to form bonds
Transition Metals • Vary in oxidation states • Different number of valence electrons • Mercury
Inner Transition Metals • Lanthanides • Actinides • “Pull out” rows in periods 6 and 7 • Trans-uranium: man-made (nuclear chemistry)

Nonmetals Physical Properties • Mostly gas at room temperature. • Poor conductors of heat • Poor electrical conductors • Brittle • Non-lustrous • Low melting and boiling points

Non-metals Chemical Properties • Gain or share valence electrons

Halogens (Nonmetals) • Form salts when bonded to alkaline earth metals • Form acids when bonded to Hydrogen • 7 valence electrons • Oxidation of -1 • Readily reactive
Noble Gases (Nonmetals) • Unreactive/inert • Odorless • Colorless • 8 valence electrons (2 for He) • Full octet (outer shell)

Metalloids • Similar to both metals and nonmetals. • Different conditions change properties. • Form alloys with metals. • “Stair-step” elements

VALENCE AND TRENDS

Valence Electrons • Valence electrons – Electrons in an atom’s highest-numbered energy level. • How many are in each of the examples?

Valence Electrons Shortcut • You can tell how many valence electrons any atom should have by its position in the periodic table. 1 8 2 34 5 67 2 variable

Octet Rule • 8 electrons in outer shell is most stable (one exception) • Atoms will combine to make the octet full, giving them the same electron configuration as the closest noble gas. • How many electrons do each element (from the example) want to lose or gain?

Atomic Radii (AR) • Center of the nucleus to edge of the electron cloud.

Atomic Radii (AR) • Increases down a group • Each new energy level adds radius • Electrons have more room to move around

Atomic Radii (AR) • Decreases across a period • More protons and electrons in the same energy level (same amount of space) • Increased attraction pulls the e- cloud closer to the nucleus (like magnets)

Electronegativity (EN) • Ability to gain or hold electrons • Forms a negative ion (anion) • A + e- A- + E • Does not include Noble Gases
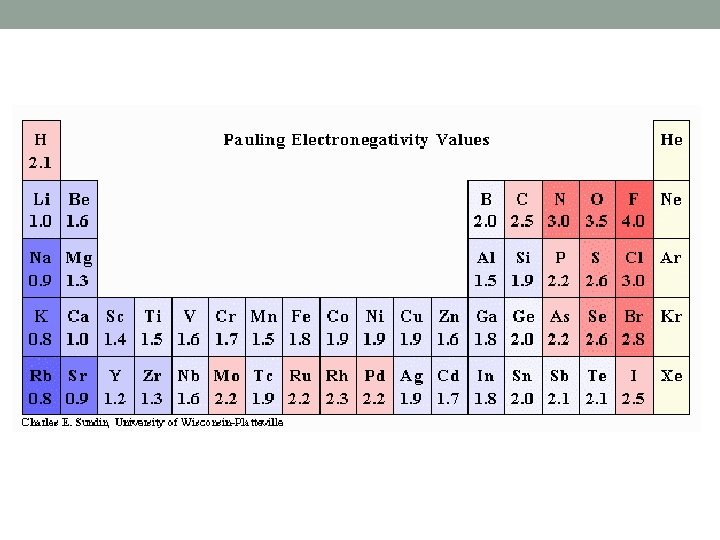

Electronegativity (EN) • Decreases down a group • As you move down, space is added between e- and nucleus • Lessens attractive force between p+ and e • Easier for e- to be removed from atom
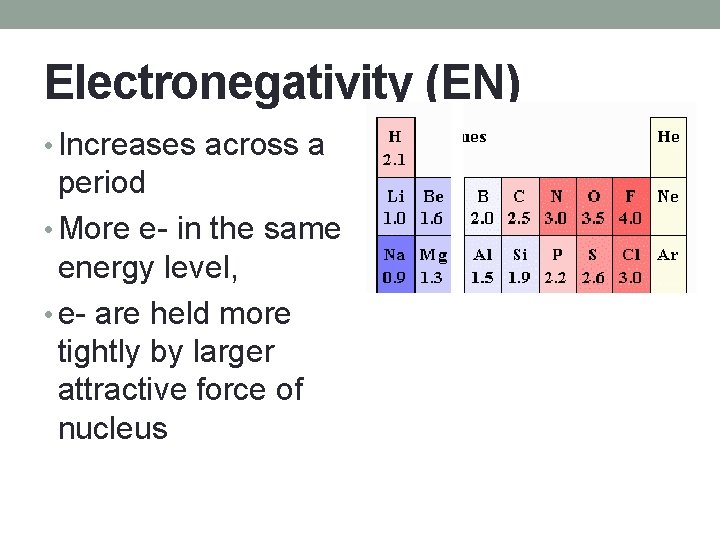
Electronegativity (EN) • Increases across a period • More e- in the same energy level, • e- are held more tightly by larger attractive force of nucleus

Ionization Energy (IE) • Energy put into molecule to remove outer shell electrons (valence electrons) • Forms a positive ion (cation) • A + E A+ + e-
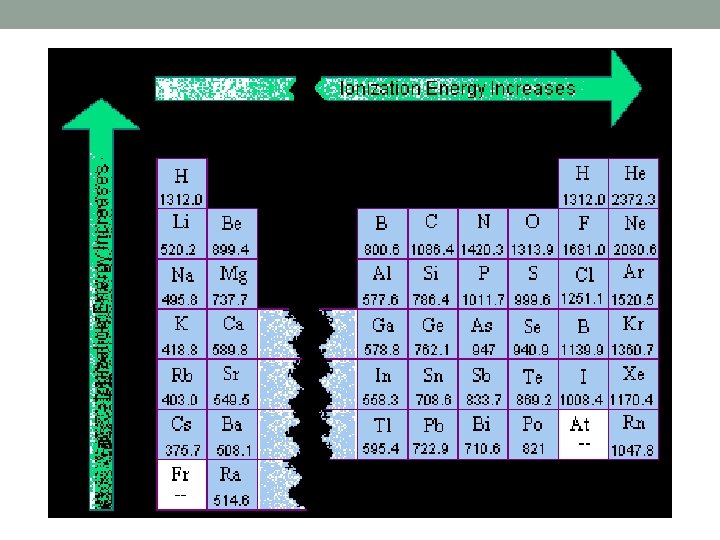

Ionization Energy (IE) • Decreases down a group • More space between nucleus and electrons • Less energy to remove e- due to less attractive force

Ionization Energy (IE) • Increases across a period • More e- in the same energy level • Held tighter because of higher attractive force from nucleus • More energy to remove e-
- Slides: 31