Periodic Table 3 15 18 Due Periodic Table

Periodic Table: 3. 15. 18 Due: Periodic Table Wksht. -qsts. 1 -6 Objectives: I can identify elements on the table based on what group and period they are in. I identify important groups on the table. I can locate metals, non-metals, and metalloids on the table. I can distinguish between representative and transitional metals. I can predict chemical reactivity of an element based on its location on the table.
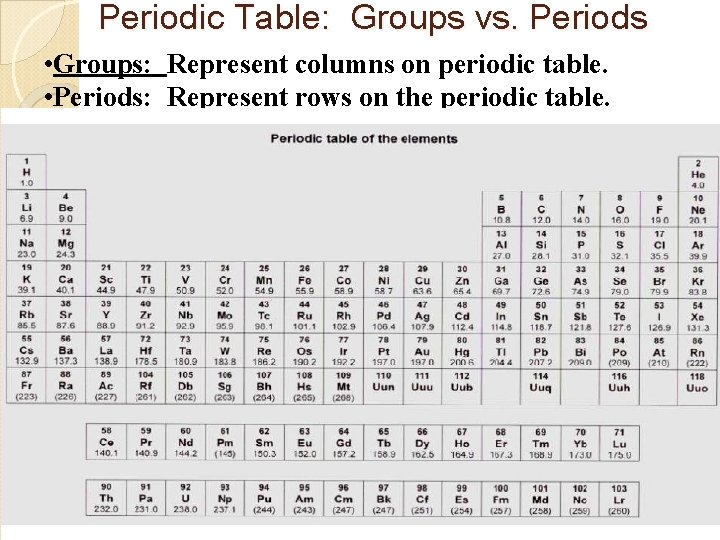
Periodic Table: Groups vs. Periods • Groups: Represent columns on periodic table. • Periods: Represent rows on the periodic table.
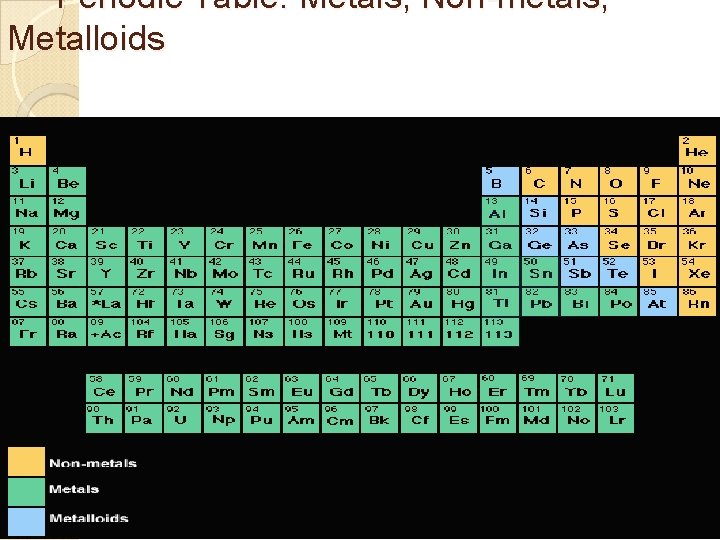
Periodic Table: Metals, Non-metals, Metalloids
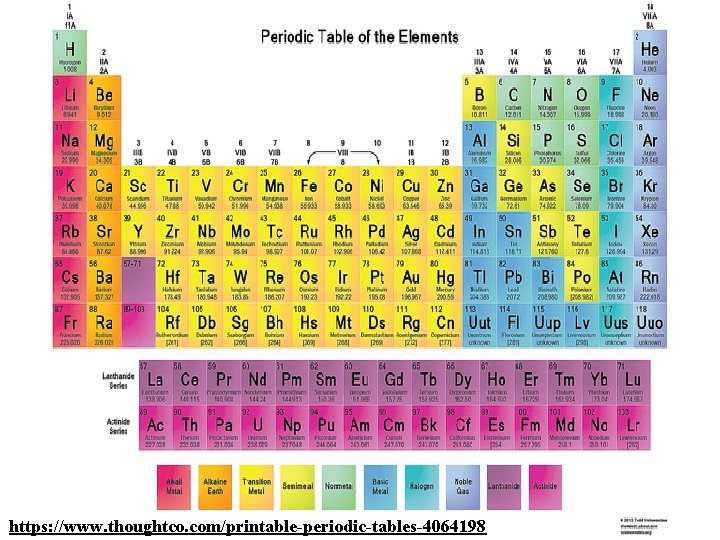
Periodic Table: Groups https: //www. thoughtco. com/printable-periodic-tables-4064198
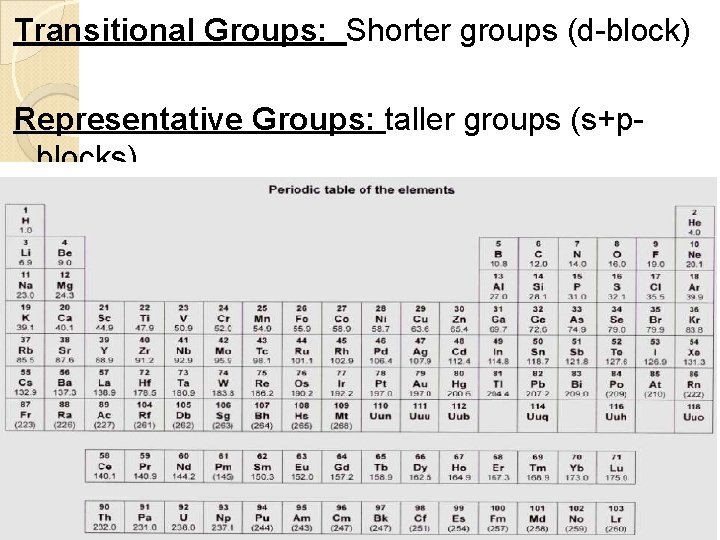
Transitional Groups: Shorter groups (d-block) Representative Groups: taller groups (s+pblocks)

Periodic Table Check-up 1. a. What period and group is sulfur on? b. What element is in period 6; group 2 ? 2. Give an example of an element from the following groups: a. Noble Gases b. Transitional c. Alkali d. Halogens, e. Alkaline Earth 3. Circle the elements below that are representative: , Na, Cr, Ag, Kr, C, Sn, Au, Pb

Periodic Table: 3. 16. 18 Due: Periodic Table Wksheet completed Metallic vs. Non-metallic Venn Diagram Objectives: I can locate elements by the period and group they are in. Identify the name of certain groups. I can classify elements as representative or transitional. I can classify elements as metals, non-metals, or metalloids. I can distinguish between metallic and nonmetallic properties. I can predict chemical reactivity for an element based on its location on the table.
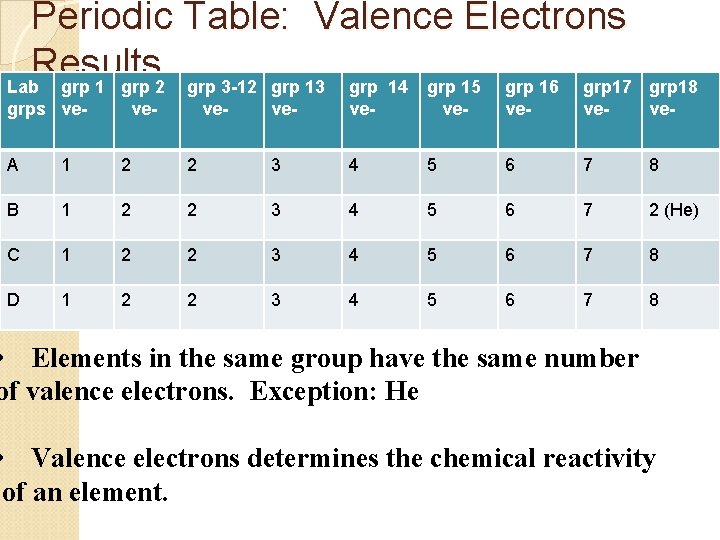
Periodic Table: Valence Electrons Results Lab grp 1 grp 2 grp 3 -12 grp 13 grp 14 grp 15 grp 16 grp 17 grps ve- ve- grp 18 ve- A 1 2 2 3 4 5 6 7 8 B 1 2 2 3 4 5 6 7 2 (He) C 1 2 2 3 4 5 6 7 8 D 1 2 2 3 4 5 6 7 8 • Elements in the same group have the same number of valence electrons. Exception: He • Valence electrons determines the chemical reactivity of an element.

Periodic Table: 3. 16. 18 Due: Periodic Table Wksheet completed Metallic vs. Non-metallic Venn Diagram Objectives: I can locate elements by the period and group they are in. Identify the name of certain groups. I can classify elements as representative or transitional. I can classify elements as metals, non-metals, or metalloids. I can distinguish between metallic and nonmetallic properties. I can predict chemical reactivity for an element based on its location on the table.

Chemical Stability Octet Rule: Atoms will gain, lose, or share valence electrons to reach maximum stability. What is stability for most atoms? 8 valence electrons (ve-) Exceptions: H and He max. stability = 2 ve. How do atoms achieve stability or 8 ve- ? Atoms chemically bond with other atoms. Formation of diverse compounds in nature.
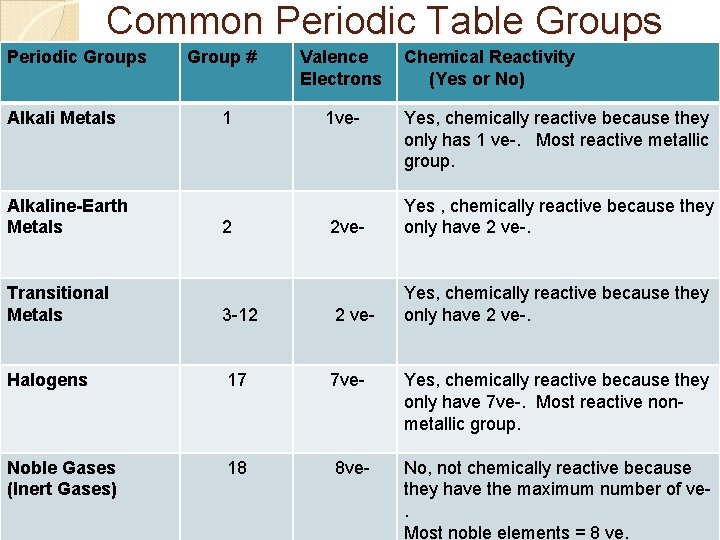
Common Periodic Table Groups Periodic Groups Alkali Metals Alkaline-Earth Metals Group # 1 2 Valence Electrons 1 ve- Chemical Reactivity (Yes or No) Yes, chemically reactive because they only has 1 ve-. Most reactive metallic group. 2 ve- Yes , chemically reactive because they only have 2 ve-. Yes, chemically reactive because they only have 2 ve-. Transitional Metals 3 -12 2 ve- Halogens 17 7 ve- Yes, chemically reactive because they only have 7 ve-. Most reactive nonmetallic group. Noble Gases (Inert Gases) 18 8 ve- No, not chemically reactive because they have the maximum number of ve. Most noble elements = 8 ve.

Chemical Stability: Ions Metals will lose ve- to reach stability. Form a cation (+ charged) www. google. com/url? sa=i& Non-metals will gain ve- to reach stability. Form an anion (- charged) http: //www. green-planet-solar-energy. com/the-element-chlorine. html

Chemical Stability: Ions Will metals tend to gain or lose valence electrons to reach stability? *Metals will lose the few valence Electrons they have to become stable. They become cations. 1. www. google. com/url? sa=i& Will non-metals tend to gain or lose valence electrons to reach stability? Non-metals will gain the few they need to become stable. They become anions. http: //www. green-planet-solar-energy. com/the-element-chlorine. html

Chemical Stability-Key Determine what type of charge each element below would form to reach maximum stability. a. b. c. d. Sodium Na 1+ Oxygen O 2 Argon Ar Phosphorus p 3 e. Chromium Cr 2+

Periodic Table: 3. 19. 18 Periodic Table Wksheet completed Metallic vs. Non-metallic Venn Diagram Objectives: I can locate elements by the period and group they are in. Identify the name of certain groups. I can classify elements as representative or transitional. I can classify elements as metals, non-metals, or metalloids. I can distinguish between metallic and nonmetallic properties. I can predict chemical reactivity for an element based on its location on the table.

Chemical Stability: Ions Will metals tend to gain or lose valence electrons to reach stability? *Metals will lose the few valence Electrons they have to become stable. They become cations. 1. www. google. com/url? sa=i& Will non-metals tend to gain or lose valence electrons to reach stability? Non-metals will gain the few they need to become stable. They become anions. http: //www. green-planet-solar-energy. com/the-element-chlorine. html

Periodic Table: 3. 21. 18 Due: Periodic Table Review Objectives: I can predict chemical reactivity for an element based on its location on the table. I can predict the charge for an element when it becomes stable. I can distinguish between metallic and nonmetallic properties. I can use the periodic table to predict trends for atomic radius, ionization energy and electronegativity.

Metallic vs. Non-metallic Properties. Key Low luster (NM) High luster(M) low density (NM) High density(M) High melting point(M) Low melting point(NM) Ductility(M) Primary gases at room temp. (NM) Primary solids at room temp. (M) Alkaline earth group(M) Stability(both) Brittle(NM) Malleable(M) Conductors of heat and electricity(M) Insulators of heat and electricity(NM) Transitional group(M) Noble gases(NM) Halogens(NM) Valence electrons(both) Alkali group(M)
Metallic vs. Non-metallic Properties

Periodic Table: Quiz Give an example of a metal, non-metal, and metalloid from the periodic table. 2. Classify the following as either metallic (M) or non-metallic (NM) insulator of heat high density ductility anion high melting point high luster conductors of e 2. Determine if the following neutral elements needs to become ions to reach stability. If so, predict what charge they would have to reach stability. 1.

Periodic Table: 3. 21. 18 Due: Periodic Trends Worksheet Objectives: Periodic Table Quiz I can use the periodic table to predict trends for atomic radius, ionization energy and electronegativity. I can identify the scientist responsible for the organization of the periodic table. I know how the current periodic table is arranged.

Periodic Trends Atomic Radius Ionization Energy Electronegativity

Periodic Trends Atomic Radius • Size of atom • Distance from nucleus to outer energy level • Measure in picometers (pm) 1 pm = 1 x 10 -12 m Ionization Energy • Energy needed to remove a valence electron from an atom when it becomes a cation. • Metals have a lower ionization energy than nonmetals, why the become cations to achieve stability. Electronegativity • The degree of attraction one atom’s nucleus has towards another atom’s ve- when they bond with one another to become stable.
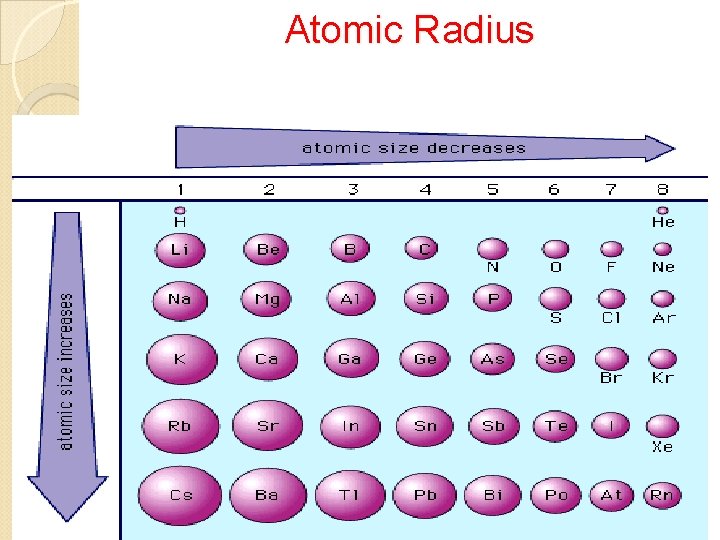
Atomic Radius
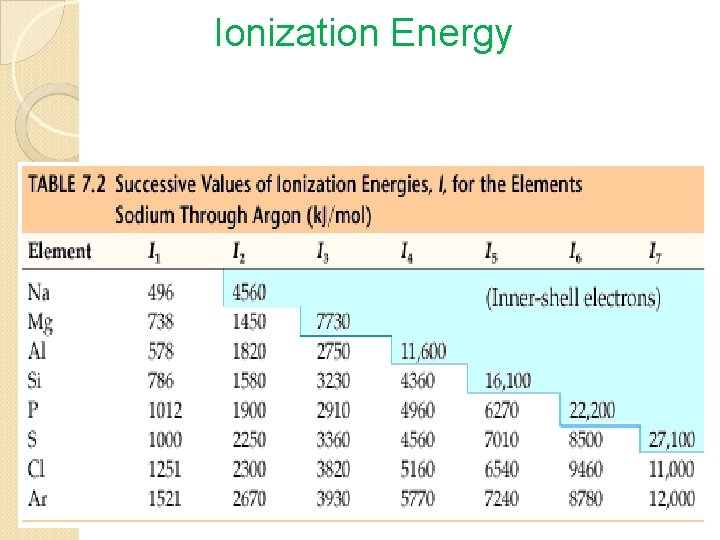
Ionization Energy
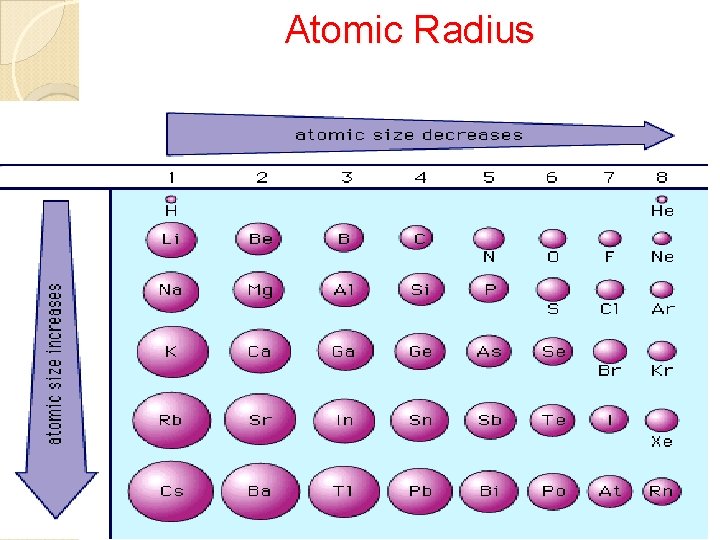
Atomic Radius

Electronegativity
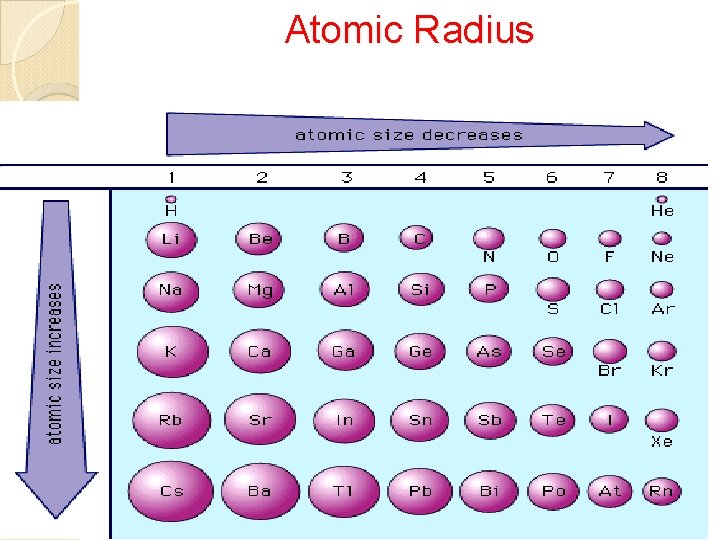
Atomic Radius

Atomic Radius of Atoms/Ions file: ///I: /Atomic_&_ionic_radii%20(1). svg

Size of Ions Circle the atom that is larger in size. a. Ca or Ca 2+ 1. b. S or S 2 - Circle the atom that is smaller in size. a. Al or Al 3+ 2. 3 -

Size of Ions Circle the atom that is larger in size. a. Ca or Ca 2+ Larger because has an extra energy level. a. S or S 2 Larger because more ve- in energy level so repulsion force causes the outer energy level to swell. (ve- push away from one another more) 2. Circle the atom that is smaller in size. a. Al or Al 3+ 1. b. N or N 3 -

History of the Periodic Table Dmitri Mendeleev: • Russian chemist and teacher • When organized elements into groups by similar chemical properties, he observed the periods increasing in atomic mass. (1869) • His organization system was successful at predicting undiscovered elements. • Do you observe any inconsistencies with his organization system?

History of Periodic Table Henry Moseley: • British Physicists • Tweaked Mendeleev’s periodic table. • When elements were placed in groups by chemical properties, the periods consistently increased by atomic number. (1913) • Current organization of elements on the periodic table.
- Slides: 33