Periodic Properties of Elements 1 Effective nuclear charge










- Slides: 29

Periodic Properties of Elements 1 -Effective nuclear charge (Zeff) is the “positive charge” felt by an electron. Zeff = Z - s 0 < s < Z (s = shielding constant) Zeff Z – number of inner or core electrons Z Core Zeff Radius (pm) Na 11 10 1 186 Mg 12 10 2 160 Al 13 10 3 143 Si 14 10 4 132

Effective Nuclear Charge (Zeff) increasing Zeff

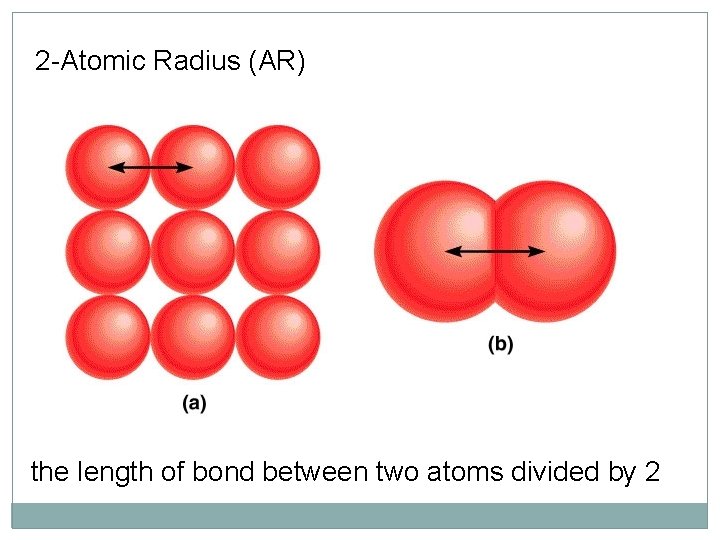
2 -Atomic Radius (AR) the length of bond between two atoms divided by 2


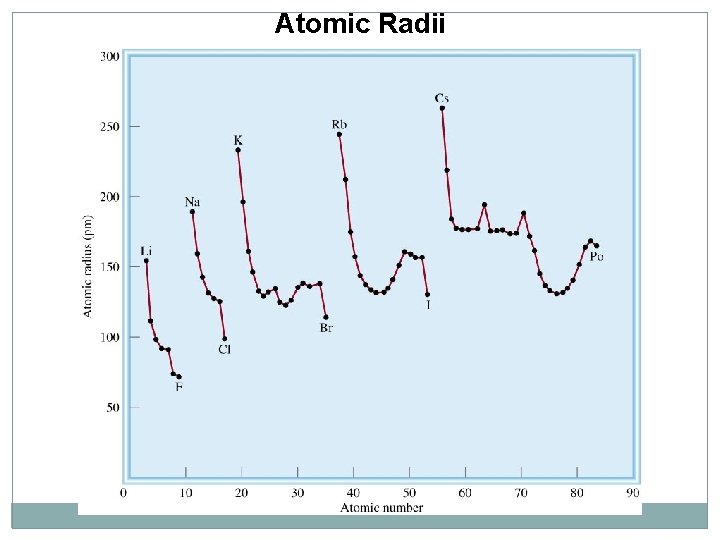
Atomic Radii

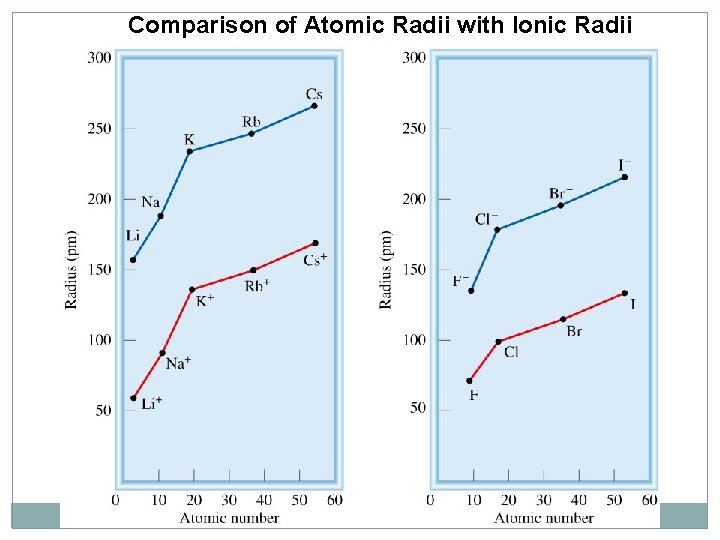
Comparison of Atomic Radii with Ionic Radii

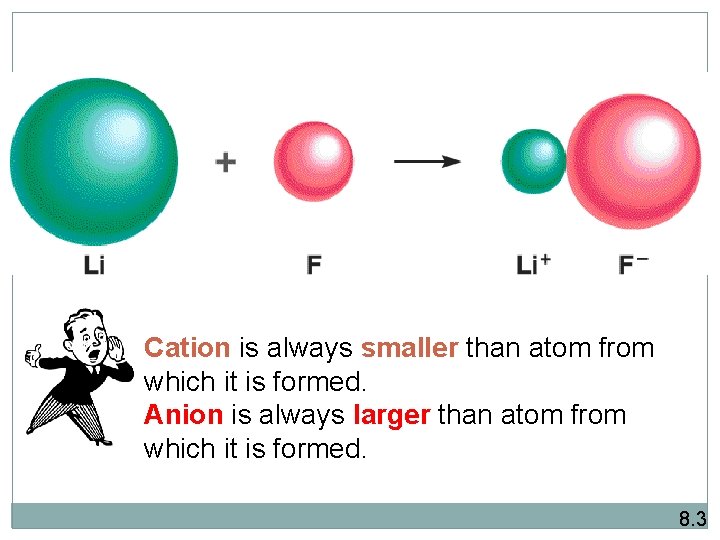
Cation is always smaller than atom from which it is formed. Anion is always larger than atom from which it is formed. 8. 3

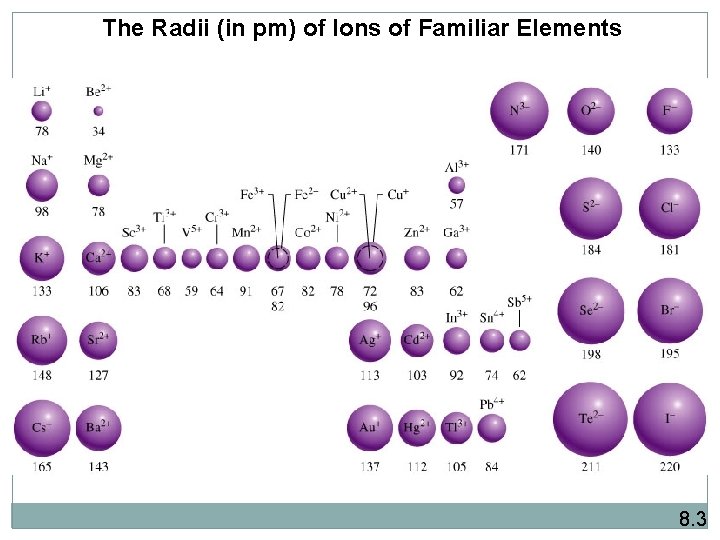
The Radii (in pm) of Ions of Familiar Elements 8. 3

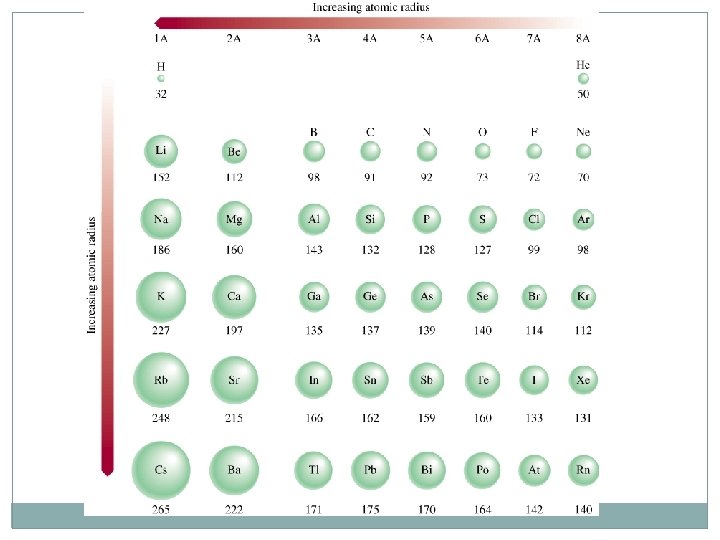
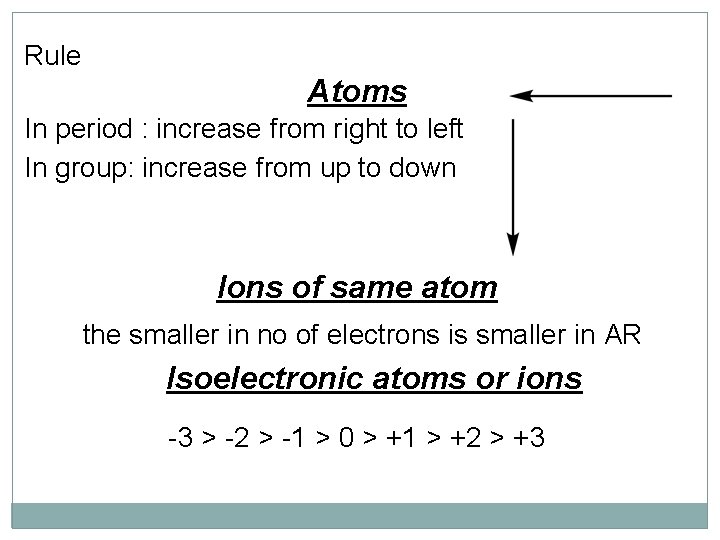
Rule Atoms In period : increase from right to left In group: increase from up to down Ions of same atom the smaller in no of electrons is smaller in AR Isoelectronic atoms or ions -3 > -2 > -1 > 0 > +1 > +2 > +3

Example 8. 2 p 332: Solution: N & P are in the same group. 1 Atomic radius increases Referring to a periodic table, arrange the following atoms in order of increasing atomic radius: P, Si, N. Atomic radius decreases N is smaller than P → 2. P & Si are in the same period P is smaller than Si → ﺍﻟﺼﻐﻴﺮ ﺍﻟﻰ ﺍﻟﻜﺒﻴﺮ The arrangement of increasing atomic radius is: N < P < Si

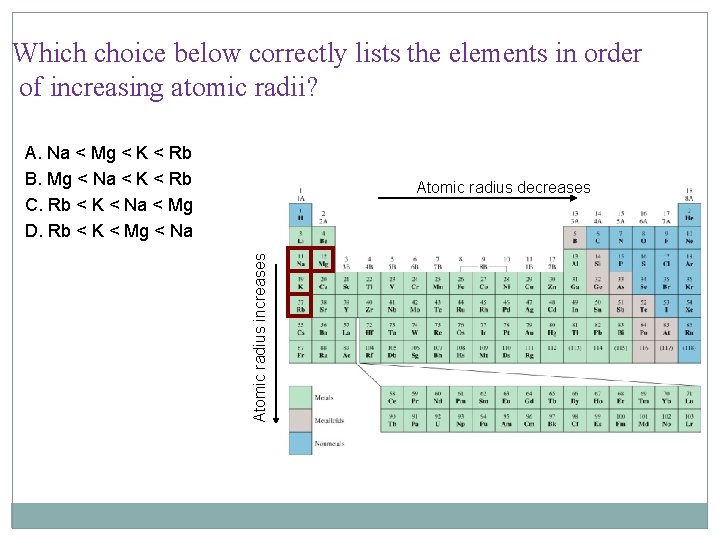
Which choice below correctly lists the elements in order of increasing atomic radii? A. Na < Mg < K < Rb B. Mg < Na < K < Rb C. Rb < K < Na < Mg D. Rb < K < Mg < Na Atomic radius increases Atomic radius decreases

Example 8. 3 p 335: For each of the following pairs, indicate which one is larger: N 3 - or F-. a Mg 2+ or Ca 2+. b Fe 2+ or Fe 3+. c F- < N 3 - isolelctronic anions → → Mg 2+ < Ca 2+ → in the same group → → cations → Fe 3+ < Fe 2+


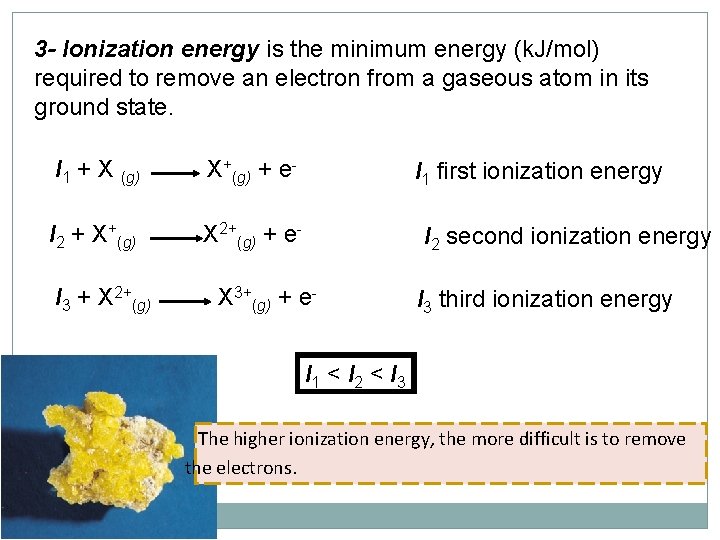
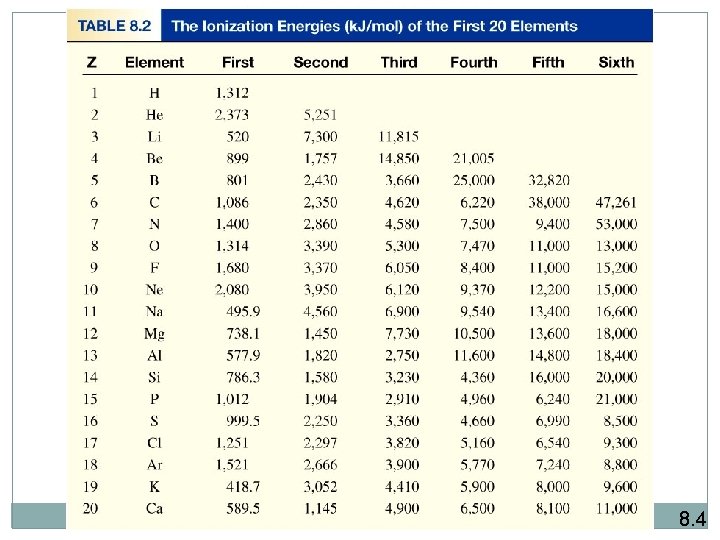
3 - Ionization energy is the minimum energy (k. J/mol) required to remove an electron from a gaseous atom in its ground state. I 1 + X (g) X+(g) + e- I 2 + X+(g) X 2+(g) + e- I 3 + X 2+(g) I 1 first ionization energy I 2 second ionization energy X 3+(g) + e- I 3 third ionization energy I 1 < I 2 < I 3 The higher ionization energy, the more difficult is to remove the electrons.

8. 4

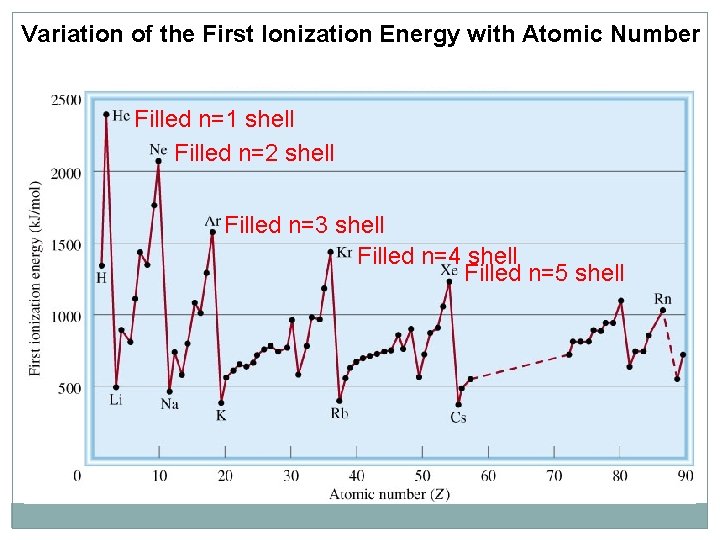
Variation of the First Ionization Energy with Atomic Number Filled n=1 shell Filled n=2 shell Filled n=3 shell Filled n=4 shell Filled n=5 shell

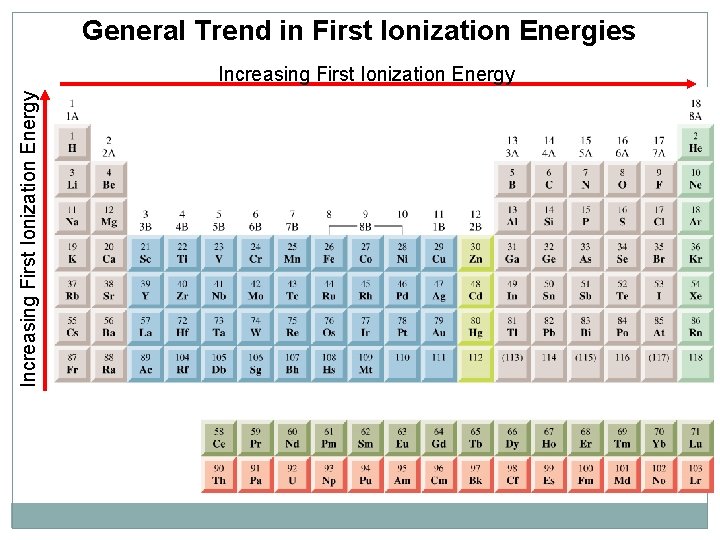
General Trend in First Ionization Energies Increasing First Ionization Energy

Ionization Energy (IE) In period : increase from left to right In group: increase from down to up Some Notes IE of the transition elements don't increase across period as rapidly as the main elements IE of inner transition elements remain almost constant Metals have low IE due to it tends to lose electron in chemical reaction Non metal have high IE due to it tends to accept electrons

Noble gases have the highest IE due to they are stable IE of atoms Be, Mg, Zn, Cd, Hg have higher values than elements that follow them because they have filled s subshell (ns 2) IE of atoms of group 5 A have higher value than elements that follow them because they have half filled p subshell (ns 2 np 3) PT IE value generally depends on if the loss of electron are from filled or half filled

Some irregularities (Exceptions) of Ionization Energy Group 2 A (ns 2 ) higher than 3 A (ns 2 np 1) in the same period. Ø Example (Be & B) Group 5 A (ns 2 np 3) higher than 6 A (ns 2 np 4) in the same Ø period. Example (N & O) (3 A < 2 A) (6 A < 5 A) 1 A < 2 A < 3 A < 4 A < 5 A < 6 A < 7 A < 8 A X 1 A < 3 A < 2 A < 4 A < 6 A < 5 A < 7 A < 8 A √ Decreasing First Ionization Energy Increasing First Ionization Energy

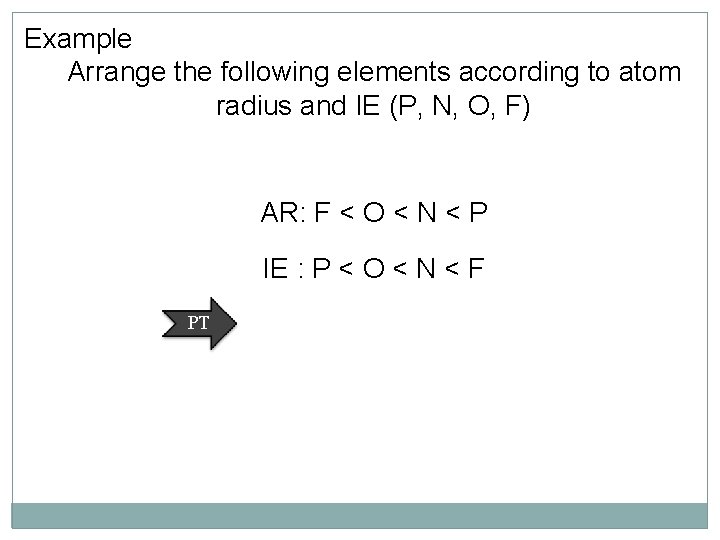
Example Arrange the following elements according to atom radius and IE (P, N, O, F) AR: F < O < N < P IE : P < O < N < F PT

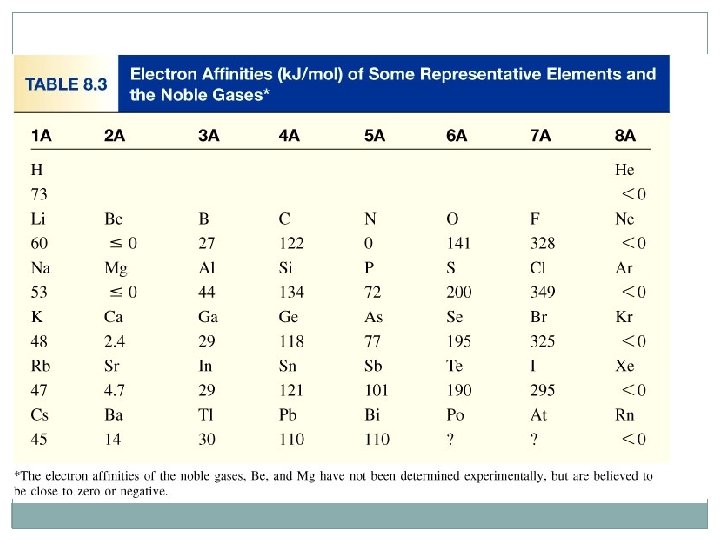
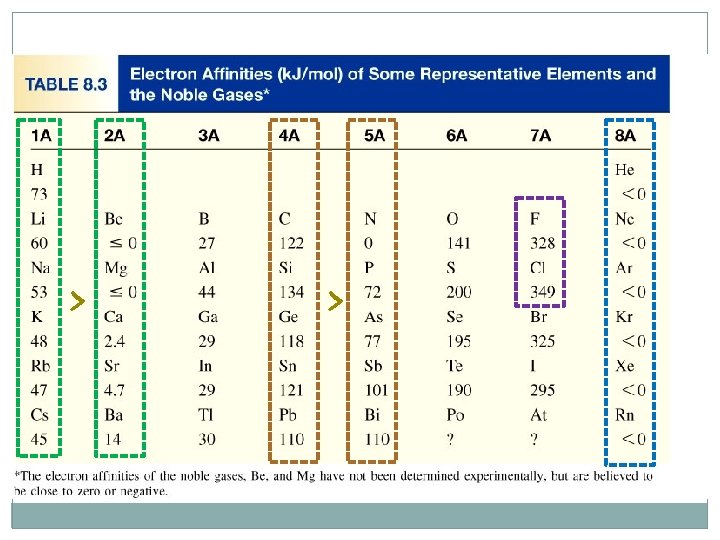
4 -Electron affinity is the negative of the energy change that occurs when an electron is accepted by an atom in the gaseous state to form an anion. X (g) + e- X-(g) F (g) + e- X-(g) O (g) + e- O-(g) DH = -141 k. J/mol EA = +141 k. J/mol DH = -328 k. J/mol EA = +328 k. J/mol


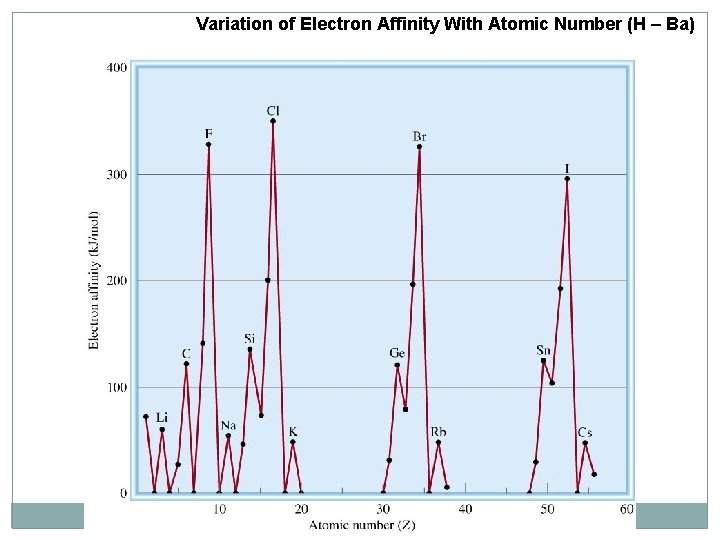
Variation of Electron Affinity With Atomic Number (H – Ba)

Electron Affinity (EA) In period : increase (-ve value) from left to right In group: increase from down to up Noble gases has +ve EA For cations it is the same as IE of neutral atom but with –ve sign EA of atoms of group 5 A have lower value than elements that befor them because they have half filled p subshell (ns 2 np 3)

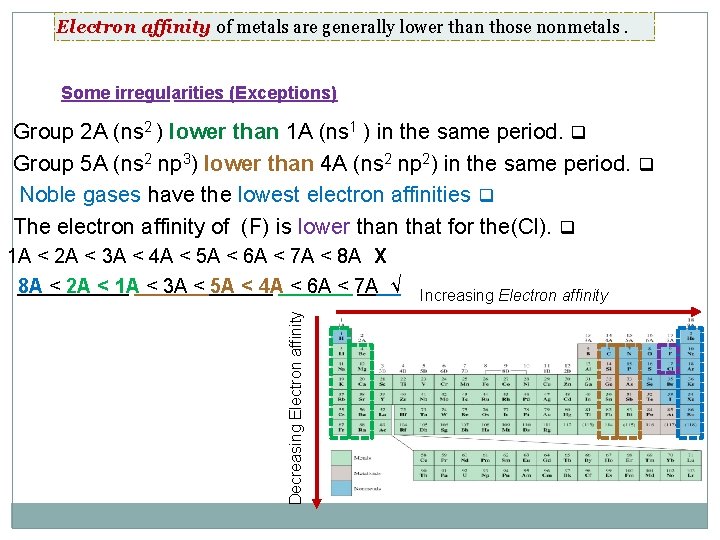
Electron affinity of metals are generally lower than those nonmetals. Some irregularities (Exceptions) Group 2 A (ns 2 ) lower than 1 A (ns 1 ) in the same period. q Group 5 A (ns 2 np 3) lower than 4 A (ns 2 np 2) in the same period. q Noble gases have the lowest electron affinities q The electron affinity of (F) is lower than that for the(Cl). q Decreasing Electron affinity 1 A < 2 A < 3 A < 4 A < 5 A < 6 A < 7 A < 8 A X 8 A < 2 A < 1 A < 3 A < 5 A < 4 A < 6 A < 7 A √ Increasing Electron affinity


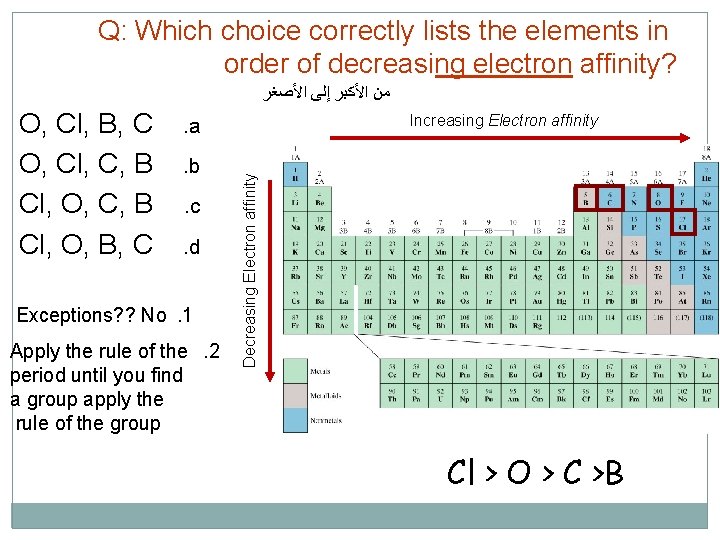
Q: Which choice correctly lists the elements in order of decreasing electron affinity? ﻣﻦ ﺍﻷﻜﺒﺮ ﺇﻟﻰ ﺍﻷﺼﻐﺮ Increasing Electron affinity . a. b. c. d Exceptions? ? No. 1 Apply the rule of the. 2 period until you find a group apply the rule of the group Decreasing Electron affinity O, Cl, B, C O, Cl, C, B Cl, O, B, C Cl > O > C >B

Arrange the following Li, Be, B, C, Si, N , S, Ar in the order of increasing: a) Effective nuclear charge Li < Be < B , C < Si < N < S < Ar b) Atomic radius Ar < S < N < C < Si < Be < Li c) Ionization energy Li < Be < Si < C < S < N < Ar d) Electron affinity Ar < Be < Li < B < N < Si < C < S